Abstract
Vascular aging and essential hypertension cause similar structural and molecular modifications in the vasculature. The 12-lipoxygenase (LO) pathway of arachidonic acid metabolism is linked to cell growth and the pathology of hypertension. Thus, elevated expression of 12-LO has been observed in vascular smooth muscle cells (VSMCs) from spontaneously hypertensive rats (SHR). In the present study, we investigated the differences in 12-LO expression and activity between VSMCs from old normotensive Wistar-Kyoto rats (old WKY, 90-week old) and SHR (13-week old). The protein and mRNA expression of basal or angiotensin II (Ang II)-induced 12-LO in old WKY VSMCs were higher than those in SHR VSMCs. The degradation rate of 12-LO mRNA in old WKY VSMCs was slower than that in SHR VSMCs. However, basal or Ang II-induced 12-LO mRNAs in both old WKY and SHR VSMCs decayed more rapidly than that in young WKY (13-week old) VSMCs. Higher expression of 12-LO in old WKY VSMCs than in SHR VSMCs was correlated with the expression level of Ang II subtype 1 receptor (AT1R). The reduced levels of nitric oxide (NO) in old WKY and SHR VSMCs compared with young WKY VSMCs were similar, and there was no significant difference in NO production between old WKY and SHR VSMCs transfected with 12-LO siRNA. In addition, in contrast to the proliferation of SHR VSMCs, the proliferation of old WKY VSMCs was not dependent on 12-LO activation. These results suggest that the potential role of 12-LO in normotensive aging vasculature may be different from that in SHR vasculature.
Similar content being viewed by others
Introduction
With increasing age, the vasculature goes through structural and molecular modifications, and changes in the expression and activation of vasoactive mediators occur naturally.1 The arterial walls of individuals with essential hypertension undergo similar functional and structural changes to those of normotensive aging individuals. However, hypertension accelerates aging-associated modifications of the arterial walls.2, 3 It is not known which molecular mechanism has a critical role in the vascular changes observed during the aging process.
Age-associated arterial proinflammation might be the foundation for the initiation and progression of vascular hypertension in old age, and this proinflammation is associated with various downstream molecules of angiotensin II (Ang II) signaling networks, including monocyte chemoattractant protein-1, calpain-1, matrix metalloproteinase type II, reactive oxygen species (ROS) and nitric oxide (NO) bioavailability.2, 4, 5, 6, 7, 8, 9 In addition, elevated production of Ang II-induced signaling molecules have been observed in spontaneously hypertensive rats (SHR) relative to normotensive Wistar–Kyoto rats (WKY) with aging.10, 11, 12 These findings suggest that the Ang II pathway has an important role in the development of hypertension with aging.
The lipoxygenase (LO) pathway is associated with the pathogenesis of hypertension.12, 13, 14, 15 LOs are classified as 5-,8-,12- and 15-LO according to the carbon atom of the arachidonic acid at which the oxygen is inserted. Three isoforms of platelet, leukocyte and epidermal type 12-LO have been cloned. Leukocyte type 12-LO and human 15-LO have high homology and are classified as 12/15-LO because they produce 12(S)-hydroxyeicosatetraenoic acid (12(S)-HETE) and 15(S)-HETE from arachidonic acid.16, 17 Leukocyte type 12-LO shows a wide tissue distribution, including vascular smooth muscle cells (VSMCs).13, 18 Ang II is a potent positive regulator of 12-LO activation and expression in porcine and human VSMCs.18, 19 In addition, an elevated level of 12-LO activity in SHR plasma and decreased blood pressure by inhibitors of 12-LO pathway have been reported in SHR.12
Most of the studies that have examined the molecular and cellular mechanisms of the interaction of aging and hypertension have focused on the effect of monocyte chemoattractant protein-1, matrix metalloproteinase type II, ET-1, ROS or NO bioavailability on endothelial function, and biological roles for 12-LO in the vascular aging process remain largely undefined. Thus, in the present study, we investigated 12-LO expression and activity in old WKY and SHR VSMCs and compared them with young WKY VSMCs.
Methods
Reagent
Trizol reagent for total RNA isolation was purchased from Invitrogen (Carlsbad, CA, USA). Dulbecco′s phosphate-buffered saline (PBS), Dulbecco′s modified Eagle′s medium (DMEM), penicillin–streptomycin and fetal bovine serum (FBS) were purchased from Gibco/BRL (Life Technologies, Gaithersburg, MD, USA). Ang II was obtained from Calbiochem (San Diego, CA, USA). CCL5 was obtained from R&D systems (Minneapolis, MN, USA). LightCycler FastStart DNA SYBR Green I Mix was obtained from Roche (Mannheim, Germany). Baicalein was obtained from Sigma-Aldrich (St Louis, MO, USA). 12(S)-HETE was purchased from Cayman Chemical (Ann Arbor, MI, USA). Actinomycin D was obtained from Sigma-Aldrich. The rabbit anti-human Ang II subtype 1 receptor (AT1R) and subtype 2 receptor (AT2R) antibodies were purchased from Abcam (Cambridge, UK). Goat anti-human 12-LO and ET-1 polyclonal antibody was purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA). The primer sequences for 12-LO, AT1R and β-actin were purchased from Bionics (Seoul, South Korea). The rat AT1R and 12-LO small interfering RNA sequences were purchased from Bioneer technology (Daejon, South Korea). All other reagents were pure-grade commercial preparations.
Experimental animals
Specific pathogen-free male inbred SHR or normotensive WKY were purchased from Japan SLC (Shizuka, Japan). All experimental animals received autoclaved food and bedding to minimize exposure to viral and/or microbial pathogens. The rats were cared for in accordance with the Guide for the Care and Use of Experimental Animals of Yeungnam Medical Center.
VSMCs preparation
VSMCs were obtained from the thoracic aortas of 90-week-old male WKY (W90 or old WKY), 13-week-old male SHR (S13) and WKY (W13 or young WKY) using the explant method as described by Griendling et al.20 VSMCs were cultured in DMEM, which was supplemented with 10% FBS and 1% penicillin–streptomycin. Cells were detached with 0.25% trypsin/EDTA and seeded onto 75-cm2 tissue culture flasks at a density of 105 cells per ml. All experiments were conducted between cell passages 3 to 7. Before stimulation, 95%-confluent VSMCs were serum-starved overnight by incubating in DMEM containing 0.1% FBS. Cell cultures were incubated in a humidified incubator at 37 °C and 5% CO2 in the presence or absence of stimuli for the indicated times.
Preparation of total RNA, RT–PCR and real-time PCR
Total RNA was extracted using the Trizol reagent according to the manufacturer′s instructions. The quantity of total RNA obtained was determined by measuring the optical density at 260 and 280 nm.
One μg of total RNA per sample was reverse-transcribed using Moloney murine leukemia virus reverse transcriptase (Perkin Elmer, Norwalk, CT, USA) and oligo dT priming at 42 °C for 15 min, according to the manufacturer′s instruction. Amplification with specific primers was performed in a Gene Amp PCR system 9600 (Perkin Elmer) for 35 cycles with a 20 s/94 °C denaturation, 10 s/57 °C annealing, 40 s/72 °C extension profile in the case of 12-LO; for 35 cycles with a 20 s/94 °C denaturation, 10 s/55 °C annealing, 40 s/72 °C extension profile in the case of AT1R; for 30 cycles with a 20 s/95 °C denaturation, 30 s/60 °C annealing, 30 s/72 °C extension profile in the case of β-actin. The mRNA of the housekeeping gene, β-actin, was amplified and used as an internal quality standard. Amplified products were electrophoresed on 1.5–2% agarose gel stained with 0.5 μg ml−1 ethidium bromide. The primer sequences were as follows: AT1R (445 bp) sense, 5′-cacctatgtaagatcgcttc-3′, antisense, 5′-gcacaatcgccataattatcc-3′; 12-LO (312 bp) sense, 5′-tggggcaactggaagg-3′, antisense, 5′-agagcgcttcagcaccat-3′; β-actin (101 bp) sense, 5′-tactgccctggctcctagca-3′, antisense, 5′-tggacagtgaggccaggatag-3′.
12-LO or ET-1 was amplified by real-time PCR using the LightCycler (Roche). RNA was reverse-transcribed to cDNA from 1 μg of total RNA and then subjected to real-time PCR. PCR was performed in triplicate. The total PCR volume was 20 μl and the PCR consisted of LightCycler FastStart DNA SYBR Green I mix (Roche), primer and 2 μl of cDNA. Before PCR amplification, the mixture was incubated at 95 °C for 10 min, and the amplification step consisted of 45 cycles of denaturation (10 s at 95 °C), annealing (5 s at the primer-appropriate temperature), and extension (10 s at 72 °C) with fluorescence detection at 72 °C after each cycle. After the final cycle, melting point analyses of all samples were performed over a temperature range of 65 to 95 °C with continuous fluorescence detection. β-actin expression levels were used for sample normalization. Results for each gene were expressed as the relative expression level compared with β-actin. The primers of 12-LO and β-actin used for PCR were the same as the primers described above and the primer used for PCR of ET-1 was as follows: sense, 5′-ctcctccttgatggacaagg-3′, antisense, 5′-cttgatgctgttgctgatgg-3′. The level of 12-LO or ET-1 mRNA was determined by comparing experimental levels to standard curves and was expressed as relative fold expression.
Small interfering RNA (siRNA)
VSMCs were plated on 24-well plates and grown to 90% confluence. VSMCs were then transfected with AT1R or 12-LO siRNA oligomers (50 nmol l−1) using lipofectamine 2000 in accordance with the manufacturer’s instructions. After 24 h of incubation, VSMCs were placed in growth medium for 24 h before the experiments. Cells were then cultured in the presence or absence of stimuli for 4 h. The sense and antisense oligonucleotides used in these experiments were as follows: AT1R siRNA sense, 5′-gucacuguuacuacaccua-3′, antisense, 5′-uagguguaguaacagugac-3′; 12-LO siRNA sense, 5′-gugugugacuauguuccaa-3′, antisense, 5′-uuggaacauagucacacac-3′.
Western blotting
Total lysates were prepared in PRO-PREP buffer (iNtRON, Seongnam, South Korea). Protein concentrations were determined by the Bradford assay (Bio-Rad, Richmond, CA, USA) using bovine serum albumin as standard. Thirty-microgram protein samples were separated on 10% SDS–polyacrylamide gels, and then transferred to nitrocellulose membranes. The membranes were soaked in 5% nonfat dried milk in TBST (10 mmol l−1 Tris–HCl pH7.5, 150 mmol l−1 NaCl and 0.05% Tween-20) for 1 h and then incubated for 16–18 h with primary antibodies against 12-LO, ET-1, AT1R, AT2R and γ-tubulin at 4 °C. Membranes were washed three times with TBST for 10 min and then incubated with horseradish peroxidase-conjugated secondary antibody for 1 h at 4 °C. The membranes were rinsed three times with TBST for 10 min and the antigen-antibody complex was detected using the enhanced chemiluminescence detection system (LAS-3000, Fujifilm, Tokyo, Japan).
Nitric oxide production
Quantitative measurement of NO production was performed using a nitrate/nitrite colorimetric assay kit (Cayman, Ann Arbor, MI, USA), according to the manufacturer’s instructions. Culture media were centrifuged at 800 r.p.m. for 10 min, and isolated supernatant was then transferred to a 96-well plate in 40 μl aliquots. 10 μl of enzyme cofactor mixture and 10 μl of nitrate reductase mixture were added to each well, and the plate was covered and incubated at RT for 3 h. Next, 100 μl of Greiss reagent (0.5% sulfanilamide, 2.5% phosphoric acid, and 0.25% naphthylenediamine) was added to the wells for 10 min and the absorbance was read at 540 nm using a microplate reader (Sunrise, Tecan, Sydney, Australia).
Enzyme-linked immunosorbent assay (ELISA)
12(S)-HETE levels in cell supernatants with an ELISA kit that was obtained from Enzo Life Sciences (San Diego, CA, USA). All procedures were performed in accordance with the manufacturer’s instructions.
VSMCs proliferation
VSMCs were plated in 24-well plates with serum-free medium for 24 h and then exposed to the stimulant. [3H]-thymidine (1 μCi ml−1) (Perkin Elmer precisely, Boston, MA, USA) was added to the plates during the last 24 h of incubation. The cells were subsequently washed three times with cold PBS. [3H]-thymidine-labeled cells were collected with 0.1% SDS, and radioactivity was counted using a Packard scintillation counter (Packard Instrument Company, Meriden, CT, USA).
Statistical analysis
Results were expressed as means±s.e.m. from at least three or four independent experiments. The statistical significance was determined by the Mann–Whitney test (for comparison between two groups) or Kruskal–Wallis ANOVA (for comparison of three groups) followed by Bonferroni’s post-hoc tests. All statistical tests were performed with the use of SPSS version 18.0. A value of P less than 0.05 was considered statistically significant.
Results
Basal expression of 12-LO is higher in old WKY VSMCs than in SHR VSMCs
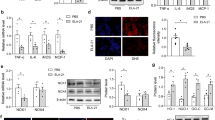
It is well known that both 12-LO and ET-1 activity are linked to the development of hypertension.21, 22 Thus, we compared the constitutive expression patterns of 12-LO at the mRNA and protein levels with those of ET-1 in WKY and SHR VSMCs. The expression of both 12-LO and ET-1 was higher in old WKY and SHR VSMCs than in young WKY VSMCs, and the expression of ET-1 in old WKY VSMCs was lower than in SHR VSMCs. However, the expression of 12-LO was significantly higher than in SHR VSMCs (Figure 1a). The effect of Ang II on 12-LO and ET-1 expression in old WKY VSMCs was also compared to that on SHR VSMCs. Ang II increased 12-LO and ET-1 mRNA expression in all three groups of VSMCs. In addition, the expression patterns of Ang II-induced 12-LO or ET-1 at the mRNA and protein levels were the same as the basal expression levels of 12-LO or ET-1. Although the expression of Ang II-induced 12-LO mRNA was remarkable in old WKY VSMCs, the protein level of 12-LO by Ang II was not remarkable compared with the expression level of Ang II-induced 12-LO mRNA (Figure 2a). This remarkably high expression of Ang II-induced 12-LO mRNA in old WKY VSMCs was consistent with the results of Ang II-reacted time course and dose response (Figure 2b). Moreover, we confirmed the production of 12(S)-HETE in response to Ang II-induced 12-LO in WKY and SHR VSMCs. Increased levels of 12(S)-HETE were detected in W90 and S13 VSMCs treated with Ang II. However, there was no significant difference in 12(S)-HETE production between old WKY VSMCs and SHR VSMCs (Figure 2d).
Expression of 12-LO and ET-1 in WKY and SHR VSMCs. (a) After total RNAs were isolated from WKY and SHR VSMCs, real-time PCR was performed. Bars represent means±s.e.m. from three independent experiments. There are statistical differences among three different characters. (b) VSMCs lysates were prepared and separated on 10% SDS–polyacrylamide gels and then immunoblotted with the 12-LO or ET-1 antibody. The data shown are representative of three independent experiments. The density of 12-LO or ET-1 was determined using Scnimage software (Scion corporation, Frederick, MD, USA). Results are shown as % of relative density over respective untreated cells. Bars represent means±s.e.m. from three independent experiments. There are statistical differences among three different characters.
Expression of Ang II-induced 12-LO in old WKY VSMCs is higher than that in SHR VSMCs. (a) WKY and SHR VSMCs were untreated or treated with Ang II (0.1 μmol l−1) for 1 h. After total RNA was isolated, real-time PCR was performed. The cell lysates were prepared and separated on 10% SDS–polyacrylamide gels and then immunoblotted with the 12-LO or ET-1 antibody. Bars represent means±s.e.m. from three independent experiments. *P<0.05 vs. untreated VSMCs. The data shown are representative of three independent experiments. (b) WKY and SHR VSMCs were treated with Ang II (0.1 μmol l−1) for the indicated times and the total RNA was analyzed by real-time PCR. Each point represents the relative 12-LO mRNA level (mean±s.e.m.) of three independent experiments, and there are statistical differences among three different characters. (c) WKY and SHR VSMCs were untreated or treated with 0.01, 0.1, 0.5 or 1 μmol l−1 of Ang II for 1 h. After total RNAs were isolated, real-time PCR was performed. Each point represents the relative 12-LO mRNA level (mean±s.e.m.) of three independent experiments, and there are statistical differences among three different characters. (d) WKY and SHR VSMCs were untreated or treated with 0.1 μmol l−1 of Ang II for 1 h. After cell supernatants were isolated, ELISA for 12(S)-HETE was performed. Bars represent means±s.e.m. from three independent experiments. *P<0.05 vs. untreated VSMCs.
The rate of 12-LO mRNA decay is slower in old WKY VSMCs than in SHR VSMCs
We next compared the basal stability of the 12-LO mRNA between WKY and SHR VSMCs, by treating the VSMCs with actinomycin D (Act. D) to prevent further transcription. After incubation up to 90 min, 12-LO mRNA levels were assessed and quantified based on the % remaining by real-time PCR. Although the basal expression of 12-LO mRNA was increased in both the old WKY and SHR VSMCs compared with that in young WKY VSMCs, the stability of 12-LO mRNA in old WKY and SHR VSMCs was lower than in the young WKY VSMCs. However, the degradation rate of 12-LO mRNA in old WKY VSMCs was slower than that in SHR VSMCs (Figure 3a).
Stability of 12-LO mRNA in WKY and SHR VSMCs. (a) WKY and SHR VSMCs were treated with actinomycin D (Act. D, 5 μg ml−1) for the indicated times before analysis of the 12-LO mRNA levels by real-time PCR. 12-LO mRNA levels were plotted as per cent mRNA remaining at each time point. The relative expression fold of 12-LO mRNA based on the real-time PCR data at 0 time point of Act. D treatment was fixed as 100% mRNA remaining. Each point represents the mean±s.e.m. from three independent experiments, and there are statistical differences among three different characters. (b) WKY and SHR VSMCs were untreated or treated with Ang II (0.1 μmol l−1) for 1 h. After additional incubation with actinomycin D (Act. D, 5 μg ml−1) for the indicated times, real-time PCR was performed. 12-LO mRNA levels were plotted as per cent mRNA remaining at each time point. The relative expression fold of 12-LO mRNA based on the real-time PCR data at 0 time point of Act. D treatment was fixed as 100% mRNA remaining. Each point represents the mean±s.e.m. from three independent experiments, *P<0.05 vs. untreated VSMCs.
To determine the effect of Ang II on the stability of 12-LO mRNA in VSMCs, WKY and SHR VSMCs were stimulated with Ang II for 1 h before treatment with Act. D. After additional incubation up to 90 min, 12-LO mRNA levels were assessed and quantified based on the % remaining by real-time PCR. Although Ang II upregulates 12-LO expression in VSMCs, the stability of Ang II-induced 12-LO mRNA in WKY and SHR VSMCs was lower than the stability of the basal 12-LO mRNA. The degradation rate of Ang II-induced 12-LO mRNA was almost the same after 30 min of exposure to Act. D in old WKY and SHR VSMCs; however, at longer times, the degradation rate in old WKY VSMCs was slower than that in SHR VSMCs (Figure 3b).
Expression of AT1R is higher in old WKY VSMCs than in SHR VSMCs
Expression of AT1R and AT2R are modulated by Ang II or 12-LO,23 and Ang II-induced 12-LO expression is mediated by these receptors.24 Thus, we hypothesized that the high expression of 12-LO mRNA in old WKY VSMCs may be influenced by AT1R or AT2R. To test this hypothesis, the basal expression levels of AT1R and AT2R protein in old WKY VSMCs were compared with those in SHR VSMCs. AT1R was increased in both the old WKY and SHR VSMCs when compared with young WKY VSMCs, and the expression of AT1R in old WKY VSMCs was remarkably higher than in SHR VSMCs. However, the level of AT2R in old WKY and SHR VSMCs was not high compared with the level of AT1R, and there was no significant difference in the expression level of AT2R between old WKY and SHR VSMCs (Figure 4a). We next examined the effect of AT1R activity on 12-LO expression in old WKY VSMCs compared with SHR VSMCs. After observing successful transfection of AT1R siRNA into old WKY and SHR VSMCs by RT–PCR, we performed real-time PCR on samples treated with AT1R-directed siRNA. The expression of 12-LO mRNA in old WKY VSMCs decreased significantly, and there was no significant difference in the expression level of 12-LO mRNA between old WKY and SHR VSMCs (Figure 4b).
High expression of 12-LO in old WKY VSMCs is associated with upregulated AT1R. (a) Cell lysates from WKY and SHR VSMCs were prepared and separated on 10% SDS–polyacrylamide gels and then immunoblotted with AT1R and AT2R antibody. The data shown are representative of three independent experiments. Bars represent means±s.e.m. from three independent experiments. There are statistical differences among three different characters. (b) Old WKY and SHR VSMCs were plated on 24-well plates, grown to a confluency of 90% and then transfected with AT1R siRNA oligomers (50 nmol l−1). Total RNAs from transfected or non-transfected VSMCs were analyzed by RT–PCR to confirm successful transfection, and then real-time PCR was performed. The data are representative of three independent experiments. Bars represent means±s.e.m. from three independent experiments. *P<0.05 vs. non-transfected VSMCs or non-transfected W90 VSMCs.
Effect of 12-LO on nitric oxide (NO) production and VSMCs proliferation of old WKY and SHR VSMCs
NO bioactivity is elevated in 12/15-LO knockout mice.25, 26 Thus, we hypothesized that the higher expression of 12-LO in old WKY VSMCs may inhibit NO production to a higher degree than observed in SHR VSMCs. Under the basal VSMCs condition, production levels of NO in old WKY and SHR VSMCs were lower than in young WKY VSMCs; however, there were no significant differences between old WKY and SHR VSMCs (Figure 5a). We also found that the 12-LO product, 12(S)-HETE, directly inhibited NO production in old WKY and SHR VSMCs. The inhibitory effect of 12(S)-HETE on NO production was weak in the both old WKY and SHR VSMCs, and there was no significant difference between old WKY and SHR VSMCs (Figure 5b). After transfection of 12-LO siRNA into VSMCs, NO production increased very weakly in both old WKY and SHR VSMCs, and there was also no significant difference in the NO level between old WKY and SHR VSMCs (Figure 5c).
There are no significant differences in the production levels of NO and inhibitory effect of 12-LO on NO production between old WKY and SHR VSMCs. (a) After 24 h of incubation, cell supernatants were isolated from cultures of WKY and SHR VSMCs, and nitrate/nitrite colorimetric assay for NO production was performed. Bars represent means±s.e.m. from three independent experiments. There are no statistical difference between some groups (same characters have no statistical difference). (b) Old WKY and SHR VSMCs were untreated or treated with 12(S)-HETE (500 nM) for 1 h. After cell supernatants were isolated, nitrate/nitrite colorimetric assay for NO production was performed. Bars represent means±s.e.m. from three independent experiments. **P<0.01 vs. untreated VSMCs. (c) WKY and SHR VSMCs were plated on 24-well plates, grown to a confluency of 90% and then transfected with 12-LO siRNA oligomers (50 nmol l−1). Total RNAs from transfected or non-transfected VSMCs were analyzed by RT–PCR to confirm successful transfection, NO levels were then measured. The data are representative of three independent experiments. Bars represent means±s.e.m. from three independent experiments.
It is widely accepted that growth of VSMCs derived from SHR is faster than that of age-matched normotensive VSMCs, and 12-LO has been shown to play a role in VSMCs proliferation associated with hypertension.13 Thus, we compared the basal proliferation of old WKY VSMCs with SHR VSMCs. There was no significant difference in basal VSMCs proliferation between old WKY and SHR VSMCs (Figure 6a). Additionally, no significant difference was observed in VSMCs proliferation between old WKY and SHR VSMCs stimulated with Ang II or 12(S)-HETE (Figure 6b). We next examined the effect of a 12-LO inhibitor, baicalein, on VSMCs proliferation in old WKY VSMCs and SHR VSMCs. Baicalein inhibited VSMCs proliferation slightly, only in SHR VSMCs (Figure 6c). To confirm this result, [3H]-thymidine incorporation was performed on old WKY and SHR VSMCs treated with 12-LO-directed siRNA. VSMCs proliferation was significantly decreased in SHR VSMCs transfected with 12-LO siRNA compared with non-transfected SHR VSMCs. However, there was no significant difference in VSMCs proliferation between non-transfected and 12-LO siRNA-transfected old WKY VSMCs (Figure 6d).
Proliferation of old WKY VSMCs is not dependent on the 12-LO pathway. (a) WKY and SHR VSMCs were cultured for 24 h in medium containing [3H]-thymidine (1 μCi ml−1). [3H]-thymidine incorporation is shown on the y axis. Bars represent means±s.e.m. from four independent experiments. There are no statistical difference between some groups (same characters have no statistical difference). (b,c) WKY and SHR VSMCs were untreated or treated with Ang II (0.1 μmol l−1, b), 12(S)-HETE (500 nM, b), or baicalein (10 μmol l−1, c) for 24 h in medium containing [3H]-thymidine (1 μCi ml−1). [3H]-thymidine incorporation is shown on the y axis. Bars represent means±s.e.m. from four independent experiments. There are statistical differences between the three or two different groups (there are statistical differences among three different characters or between the two different characters). (b) *P<0.05 vs. untreated S13 VSMCs (c). (d) Old WKY and SHR VSMCs were plated on 24-well plates, grown to 90% confluence and then transfected with 12-LO siRNA oligomers (50 nmol l−1). VSMCs were then cultured for 24 h in medium containing [3H]-thymidine (1 μCi ml−1). [3H]-thymidine incorporation is shown on the y axis. Bars represent means±s.e.m. from three independent experiments. *P<0.05 vs. non-transfected S13 VSMCs.
Discussion
Molecular modifications of vasoactive mediators in the vessel walls from normotensive old vessels have been shown to be similar to the changes observed in hypertension vessels. A vasoactive mediator, 12-LO, has been implicated in the pathogenesis of hypertension and atherosclerosis, and its activity is elevated in SHR compared with that in age matched WKY.12, 27, 28 However, as far as we know, a comparison of 12-LO expression and activity between old WKY and SHR VSMCs has not yet been reported.
This study focuses on the comparison of old normotensive VSMCs with hypertensive VSMCs. It was expected that the function of the old hypertensive cells would be markedly different from that of the young hypertensive cells, so it was decided that the young hypertensive cells were a suitable choice for the comparison of old normotensive VSMCs. Furthermore, the reason for choosing 13-week old SHRs was that hypertension is only apparent when a certain age has been reached; at 6–8 weeks, these rats experience ‘prehypertensive SHR’; and, from 10–12 weeks, they have ‘established SHR’.29 Thus, 13-week old SHRs were chosen to ensure that hypertension occurred. In addition, in this animal experiment, as the SHR’s lifespan was shorter than that of the WKY, 90-week old SHR VSMCs could not be obtained for comparison with 90-week old WKY cells; this is in agreement with other reports stating that the SHR has a lower life expectancy than the WKY.30, 31
ET-1 is a well known vasoconstrictor and has been shown to play an important role in experimental hypertensive animal models, as has 12-LO.21, 22 In this study, ET-1 expression in SHR VSMCs was higher than in old WKY VSMCs. However, despite the important roles of 12-LO in hypertensive vasculature, the expression of basal or Ang II-induced 12-LO was higher in old WKY VSMCs than in SHR VSMCs. But the expression level of 12-LO mRNA in old WKY VSMCs might not always coincide with the bioactivity of 12-LO. The 12-LO protein level induced by Ang II in old WKY VSMCs did not reflect the mRNA expression level to the mRNA expression level of Ang II-induced 12-LO. In addition, the stability of 12-LO mRNA in old WKY VSMCs was higher than that in SHR VSMCs. However, the degradation rate of basal or Ang II-induced 12-LO mRNA was more rapid in both old WKY and SHR VSMCs than in young WKY VSMCs. These results suggest that although a higher expression of 12-LO in old WKY VSMCs than in SHR VSMCs may be related to the slower degradation rate of 12-LO mRNA compared with SHR VSMCs, the expression of basal or Ang II-induced 12-LO mRNA is not likely regulated by posttranscriptional mRNA stability in both old WKY and SHR VSMCs, and aging or a hypertensive state leads to a decrease in the stability of 12-LO mRNA in VSMCs.
Ang II has two subtype receptors, the AT1R and the AT2R, and the density of AT2R is lower than that of AT1R expression in VSMCs.32 Most of the Ang II activity is mediated via AT1R, such as vasoconstriction, VSMCs proliferation, renal tubular sodium reabsorption.33, 34 Moreover, Ang II-induced 12-LO expression is mediated by AT1R activation, and high expression of AT1R is observed in the SHR.18, 23, 35 Xu et al.23 reported that the 12/15-LO pathway can regulate AT1R expression and AT1R expression is lower in mouse mesangial cells derived from 12/15-LO knockout (12/15-LOKO) mice compared with control mice. In addition, they also observed that overexpression of AT1R enhances Ang II-induced 12-LO expression in mouse mesangial cells. Therefore, we hypothesized that the high expression of 12-LO in old WKY VSMCs might be associated with AT1R expression and activation. Thus, we evaluated the basal expression levels of AT1R and AT2R in the old WKY and SHR VSMCs. The expression of AT1R in old WKY VSMCs was significant higher than that in SHR VSMCs. However, there was no significant difference in 12-LO mRNA expression between old WKY and SHR VSMCs transfected with AT1R siRNA. This result suggests that high basal expression of 12-LO mRNA in old WKY VSMCs may be due, in part, to upregulation of AT1R. It has been known that AT2R expression increases with aging, especially in VSMCs.33 Although we also detected an increase in the expression of AT2R in old WKY VSMCs, the increase level of AT1R expression was more remarkable.
NO bioavailability is reduced with aging and reduced NO activity leads to decreased vasodilatory, prothrombotic, and increased oxidative stress environment.1 The interaction between LOs and NO pathways potentially impacts the development of vascular disease.26, 36, 37 Anning et al.26 reported that 12/15-LOKO mice increase NO biosynthesis and 12/15-LO can decrease NO by catalyzing ROS-dependent decay and/or directly affecting 12/15-LO products, 12(S)-HETE. Thus, we hypothesized that the NO level in old WKY VSMCs would be lower than that in SHR VSMCs because the 12-LO level was significantly higher in old WKY VSMCs. However, no significant difference in basal NO levels was observed. In addition, 12(S)-HETE had not differential effect on NO production between old WKY and SHR VSMCs. Moreover, the increased levels of NO by 12-LO siRNA transfection were very small in the both old WKY and SHR VSMCs. Thus, in aging and hypertensive rat VSMCs, 12-LO is not likely to significantly impact NO bioavailability.
Rapid proliferation and migration of VSMCs play important roles in the pathogenesis of hypertension.38 Rapid growth of SHR VSMCs compared with WKY VSMCs is observed under basal conditions. Some studies have suggested that endogenous Ang II in SHR VSMCs promotes their basal growth and leads to the exaggerated growth of SHR VSMCs.39, 40 Satoh et al.39 reported that increases in growth factors, including TGF-β1, platelet derived growth factor (PDGF) by endogenous Ang II may contribute to the exaggerated growth of SHR VSMCs. In addition, it is well known that proliferation of VSMCs isolated from old animals is more rapid than that of VSMCs isolated from young animals.41 The increased p44/p42 MAPK and PDGF activity of VSMCs are at least in part responsible for the increase of VSMCs proliferation with the aging process.42, 43 Additionally, The change in the activation of AP-1 and increases of cyclin-dependent kinase 2 (CDK2) and cyclin A promoter activity with aging process have been shown to contribute to the increase in VSMCs proliferation.44, 45 In our study, there were no significant differences in basal or Ang II-induced VSMCs proliferation between old WKY and SHR VSMCs. It has been reported that the proliferation of mouse VSMCs isolated from 12/15-LOKO mice is reduced compared with VSMCs proliferation from control mice, and 12(S)-HETE directly induces VSMC hypertrophy.13, 46 In addition, Preston et al.47 suggested that 12-LO and 12(S)-HETE are important intermediates in hypoxia-induced pulmonary artery SMCs proliferation. Thus, we also compared the direct effect of 12(S)-HETE on VSMCs proliferation in old WKY VSMCs and SHR VSMCs. Although the 12(S)-HETE-induced VSMCs proliferation was lower than Ang II-induced VSMCs proliferation, 12(S)-HETE increased VSMCs proliferation in all three groups of VSMCs, and there was no significant difference in VSMCs proliferation induced by 12(S)-HETE between old WKY and SHR VSMCs. But, while a significant decrease in VSMCs proliferation was observed in 12-LO siRNA-transfected SHR VSMCs, VSMCs proliferation was not decreased in 12-LO siRNA-transfected old WKY VSMCs. In other words, although the 12-LO product 12(S)-HETE itself induced a significant increase in the proliferation of old WKY VSMCs, the proliferation of old WKY VSMCs was not mediated through the 12-LO pathway. This result suggests that the main mechanism of proliferation of old WKY VSMCs is likely to be mediated not through the 12-LO pathway but through the dysregulation of the cell cycle via changes in signaling pathways, including augmented cyclin A activity.
Aging is one of the strongest risk factors for developing neurogeneration, and oxygen- mediated events are considered possible mechanisms responsible for the increasing neuronal vulnerability in aging.48, 49 12/15-LO is considered one of the major sources of brain oxidative stress. According to one report, 12/15-LO proteins and enzyme activity are increased and correlates with an oxidative imbalance in the Alzheimer’s disease.50 But, a biological role of 12/15-LO in vascular aging process has not been established. Therefore, in this study, although the effect of the increased expression of 12-LO in old WKY VSMCs compared with SHR VSMCs was not correlated with NO inhibition or VSMCs proliferation, the increase of 12-LO activity in old WKY VSMCs is likely to modulate oxidative stress responses in vascular cells leading to facilitate vascular aging process.
Taken together, in old normotensive WKY VSMCs, the expression and stability of 12-LO mRNA are higher than those in SHR VSMCs, and the high expression of 12-LO is associated with AT1R upregulation but does not inhibit NO production. In addition, the proliferation of old WKY VSMCs is not dependent on the 12-LO pathway. Thus, these results suggest that 12-LO activity may play a different pathophysiological role in the normotensive aging vasculature than in the hypertensive vasculature.
References
Camici GG, Sudano I, Noll G, Tanner FC, Lüscher TF . Molecular pathways of aging and hypertension. Curr Opin Nephrol Hypertens 2009; 18: 134–137.
Wang M, Monticone RE, Lakatta EG . Arterial aging: a journey into subclinical arterial disease. Curr Opin Nephrol Hypertens 2010; 19: 201–207.
Taddei S, Virdis A, Mattei P, Ghiadoni L, Fasolo CB, Sudano I, Salvetti A . Hypertension causes premature aging of endothelial function in humans. Hypertension 1997; 29: 736–743.
Spinetti G, Wang M, Monticone R, Zhang J, Zhao D, Lakatta EG . Rat aortic MCP-1 and its receptor CCR2 increase with age and alter vascular smooth muscle cell function. Arterioscler Thromb Vasc Biol 2004; 24: 1397–1402.
Jiang L, Wang M, Zhang J, Monticone RE, Telljohann R, Spinetti G, Pintus G, Lakatta EG . Increased aortic calpain-1 activity mediates age-associated angiotensin II signaling of vascular smooth muscle cells. PLoS One 2008; 3: e2231.
Wang M, Takagi G, Asai K, Resuello RG, Natividad FF, Vatner DE, Vatner SF, Lakatta EG . Aging increases aortic MMP-2 activity and angiotensin II in nonhuman primates. Hypertension 2003; 41: 1308–1316.
Csiszar A, Wang M, Lakatta EG, Ungvari Z . Inflammation and endothelial dysfunction during aging: role of NF-kappaB. J Appl Physiol 2008; 105: 1333–1341.
Zhou X, Bohlen HG, Unthank JL, Miller SJ . Abnormal nitric oxide production in aged rat mesenteric arteries is mediated by NAD(P)H oxidase-derived peroxide. Am J Physiol Heart Circ Physiol 2009; 297: H2227–H2233.
Van der Loo B, Labugger R, Skepper JN, Bachschmid M, Kilo J, Powell JM, Palacios-Callender M, Erusalimsky JD, Quaschning T, Malinski T, Gygi D, Ullrich V, Lüscher TF . Enhanced peroxynitrite formation is associated with vascular aging. J Exp Med 2000; 92: 1731–1744.
Uddin M, Yang H, Shi M, Polley-Mandal M, Guo Z . Elevation of oxidative stress in the aorta of genetically hypertensive mice. Mech Ageing Dev 2003; 124: 811–817.
Wu R, Millette E, Wu L, de Champlain J . Enhanced superoxide anion formation in vascular tissues from spontaneously hypertensive and desoxycorticosterone acetate-salt hypertensive rats. J Hypertens 2001; 19: 741–748.
Sasaki M, Hori MT, Hino T, Golub MS, Tuck ML . Elevated 12-lipoxygenase activity in the spontaneously hypertensive rat. Am J Hypertens 1997; 10: 371–378.
Reddy MA, Kim YS, Lanting L, Natarajan R . Reduced growth factor responses in vascular smooth muscle cells derived from 12/15-lipoxygenase-deficient mice. Hypertension 2003; 41: 1294–1300.
Funk CD, Cyrus T . 12/15-lipoxygenase, oxidative modification of LDL and atherogenesis. Trends Cardiovasc Med 2001; 11: 116–124.
Natarajan R, Stern N, Nadler JL . The role of arachidonic acid metabolites on rennin and vascular smooth muscle cell growth. In Sowers JR, (ed.). Contemporary Endocrinology: Endocrinology of the Vasculature. Humana Press Inc: Totowa, NJ. 1996 pp 373–387.
Yamamoto S . Mammalian lipoxygenases: molecular structures and functions. Biochim Biophys Acta 1992; 1128: 117–131.
Funk CD . The molecular biology of mammalian lipoxygenases and the quest for eicosanoid functions using lipoxygenase-deficient mice. Biochim Biophys Acta 1996; 1304: 65–84.
Natarajan R, Gu JL, Rossi J, Gonzales N, Lanting L, Xu L, Nadler J . Elevated glucose and angiotensin II increase 12-lipoxygenase activity and expression in porcine aortic smooth muscle cells. Proc Natl Acad Sci USA 1993; 90: 4947–4951.
Kim JA, Gu JL, Natarajan R, Berliner JA, Nadler JL . A leukocyte type of 12-lipoxygenase is expressed in human vascular and mononuclear cells. Evidence for upregulation by angiotensin II. Arterioscler Thromb Vasc Biol 1995; 15: 942–948.
Griendling KK, Taubman MB, Akers M, Mendlowitz M, Alexander RW . Characterization of phosphatidylinositol-specific phospholipase C from cultured vascular smooth muscle cells. J Biol Chem 1991; 266: 15498–15504.
Van Guilder GP, Westby CM, Greiner JJ, Stauffer BL, DeSouza CA . Endothelin-1 vasoconstrictor tone increases with age in healthy men but can be reduced by regular aerobic exercise. Hypertension 2007; 50: 403–409.
Touyz RM, Schiffrin EL . Role of endothelin in human hypertension. Can J Physiol Pharmacol 2003; 81: 533–541.
Xu ZG, Yuan H, Lanting L, Li SL, Wang M, Shanmugam N, Kato M, Adler SG, Reddy MA, Natarajan R . Products of 12/15-lipoxygenase upregulate the angiotensin II receptor. J Am Soc Nephrol 2008; 19: 559–569.
Kim JH, Kim HS . Downregulation of angiotensin ll-induced 12-Lipoxygenase expression and cell proliferation in vascular smooth muscle cells from spontaneously hypertensive rats by CCL5. Korean J Physiol Pharmacol 2009; 13: 385–392.
Cruzado M, Castro C, Risler N, Miatello R . Changes of inducible nitric oxide synthase in aortic cells during the development of hypertension: effect of angiotensin II. Biocell 2002; 26: 61–67.
Anning PB, Coles B, Bermudez-Fajardo A, Martin PE, Levison BS, Hazen SL, Funk CD, Kühn H, O'Donnell VB . Elevated endothelial nitric oxide bioactivity and resistance to angiotensin-dependent hypertension in 12/15-lipoxygenase knockout mice. Am J Pathol 2005; 166: 653–662.
Natarajan R, Rosdahl J, Gonzales N, Bai W . Regulation of 12-lipoxygenase by cytokines in vascular smooth muscle cells. Hypertension 1997; 30: 873–879.
Nozawa K, Tuck ML, Golub M, Eggena P, Nadler JL, Stern N . Inhibition of lipoxygenase pathway reduces blood pressure in renovascular hypertensive rats. Am J Physiol 1990; 259: 1774–1780.
Cruzado MC, Risler NR, Miatello RM, Yao G, Schiffrin EL, Touyz RM . Vascular smooth muscle cell NAD(P)H oxidase activity during the development of hypertension: effect of angiotensin II and role of insulinlike growth factor-1 receptor transactivation. Am J Hypertens 2005; 18: 81–87.
Freis ED, Ragan D . Effect of treatment on longevity in spontaneously hypertensive rats. Proc Soc Exp Biol Med 1975; 150: 422–424.
Baiardi G, Bregonzio C, Jezova M, Armando I, Saavedra JM . Angiotensin II AT1 receptor blockade prolongs the lifespan of spontaneously hypertensive rats and reduces stress-induced release of catecholamines, glucocorticoids, and vasopressin. Ann NY Acad Sci 2004; 1018: 131–136.
El Mabrouk M, Touyz RM, Schiffrin EL . Differential ANG II-induced growth activation pathways in mesenteric artery smooth muscle cells from SHR. Am J Physiol Heart Circ Physiol 2001; 281: 30–39.
Carey RM . Angiotensin receptors and aging. Hypertension 2007; 50: 33–34.
Carey RM, Siragy HM . Newly recognized components of the renin-angiotensin system: potential roles in cardiovascular and renal regulation. Endocr Rev 2003; 24: 261–271.
Natarajan R, Gonzales N, Lanting L, Nadler J . Role of the lipoxygenase pathway in angiotensin II-induced vascular smooth muscle cell hypertrophy. Hypertension 1994; 23: 142–147.
O'Donnell VB, Taylor KB, Parthasarathy S, Kühn H, Koesling D, Friebe A, Bloodsworth A, Darley-Usmar VM, Freeman BA . 15-Lipoxygenase catalytically consumes nitric oxide and impairs activation of guanylate cyclase. J Biol Chem 1999; 274: 20083–20091.
Coffey MJ, Natarajan R, Chumley PH, Coles B, Thimmalapura PR, Nowell M, Kühn H, Lewis MJ, Freeman BA, O'Donnell VB . Catalytic consumption of nitric oxide by 12/15- lipoxygenase: inhibition of monocyte soluble guanylate cyclase activation. Proc Natl Acad Sci USA 2001; 98: 8006–8011.
Fukuda N, Satoh C, Hu WY, Soma M, Kubo A, Kishioka H, Watanabe Y, Izumi Y, Kanmatsuse K . Production of angiotensin II by homogeneous cultures of vascular smooth muscle cells from spontaneously hypertensive rats. Arterioscler Thromb Vasc Biol 1999; 19: 1210–1217.
Satoh C, Fukuda N, Hu WY, Nakayama M, Kishioka H, Kanmatsuse K . Role of endogenous angiotensin II in the increased expression of growth factors in vascular smooth muscle cells from spontaneously hypertensive rats. J Cardiovasc Pharmacol 2001; 37: 108–118.
Kubo A, Fukuda N, Soma M, Izumi Y, Kanmatsuse K . Inhibitory effect of an angiotensin II type 1 receptor antagonist on growth of vascular smooth muscle cells from spontaneously hypertensive rats. J Cardiovasc Pharmacol 1996; 27: 58–63.
Hariri RJ, Hajjar DP, Coletti D, Alonso DR, Weksler ME, Rabellino E . Aging and arteriosclerosis: cell cycle kinetics of young and old arterial smooth muscle cells. Am J Pathol 1988; 131: 132–136.
Gennaro G, Ménard C, Giasson E, Michaud SE, Palasis M, Meloche S, Rivard A . Role of p44/p42 MAP kinase in the age-dependent increase in vascular smooth muscle cell proliferation and neointimal formation. Arterioscler Thromb Vasc Biol 2003; 23: 204–210.
McCaffrey TA, Nicholson AC, Szabo PE, Weksler ME, Weksler BB . Aging and arteriosclerosis: the increased proliferation of arterial smooth muscle cells isolated from old rats is associated with increased platelet-derived growth factor-like activity. J Exp Med 1988; 167: 163–174.
Li M, Fukagawa NK . Age-related changes in redox signaling and VSMC function. Antioxid Redox Signal 2010; 12: 641–655.
Rivard A, Principe N, Andrés V . Age-dependent increase in c-fos activity and cyclin A expression in vascular smooth muscle cells A potential link between aging, smooth muscle cell proliferation and atherosclerosis. Cardiovasc Res 2000; 45: 1026–1034.
Reddy MA, Thimmalapura PR, Lanting L, Nadler JL, Fatima S, Natarajan R . The oxidized lipid and lipoxygenase product 12(S)-hydroxyeicosatetraenoic acid induces hypertrophy and fibronectin transcription in vascular smooth muscle cells via p38 MAPK and cAMP response element-binding protein activation. Mediation of angiotensin II effects. J Biol Chem 2002; 277: 9920–9928.
Preston IR, Hill NS, Warburton RR, Fanburg BL . Role of 12-lipoxygenase in hypoxia induced rat pulmonary artery smooth muscle cell proliferation. Am J Physiol Lung Cell Mol Physiol 2006; 290: 367–374.
Praticò D, Delanty N . Oxidative injury in diseases of the central nervous system: focus on Alzheimer's disease. Am J Med 2000; 109: 577–585.
Praticò D . Alzheimer's disease and oxygen radicals: new insights. Biochem Pharmacol 2002; 63: 563–567.
Praticò D, Zhukareva V, Yao Y, Uryu K, Funk CD, Lawson JA, Trojanowski JQ, Lee VM . 12/15-lipoxygenase is increased in Alzheimer's disease: possible involvement in brain oxidative stress. Am J Pathol 2004; 164: 1655–1662.
Acknowledgements
This work was supported by the 2010 Yeungnam University Research Center.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, H., Jeong, D., Park, H. et al. Comparison of 12-lipoxygenase expression in vascular smooth muscle cells from old normotensive Wistar-Kyoto rats with spontaneously hypertensive rats. Hypertens Res 36, 65–73 (2013). https://doi.org/10.1038/hr.2012.119
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/hr.2012.119
Keywords
This article is cited by
-
Activation of the D4 dopamine receptor attenuates proliferation and migration of vascular smooth muscle cells through downregulation of AT1a receptor expression
Hypertension Research (2015)
-
CCL5 upregulates IL-10 expression and partially mediates the antihypertensive effects of IL-10 in the vascular smooth muscle cells of spontaneously hypertensive rats
Hypertension Research (2015)