- NEWS
Omicron-targeted vaccines do no better than original jabs in early tests
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
doi: https://doi.org/10.1038/d41586-022-00003-y
References
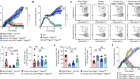
Gagne, M. et al. Preprint at bioRxiv https://www.biorxiv.org/content/10.1101/2022.02.03.479037v1?s=08 (2022).
Ying, B. et al. Preprint at bioRxiv https://www.biorxiv.org/content/10.1101/2022.02.07.479419v1 (2022).
Lee, I.-J. et al. Preprint at bioRixv https://www.biorxiv.org/content/10.1101/2022.01.31.478406v1 (2022).
Hawman, D. W. et al. Preprint at bioRxiv https://www.biorxiv.org/content/10.1101/2022.01.31.478520v1 (2022).

 Omicron thwarts some of the world’s most-used COVID vaccines
Omicron thwarts some of the world’s most-used COVID vaccines
 Will Omicron end the pandemic? Here’s what experts say
Will Omicron end the pandemic? Here’s what experts say
 How well can Omicron evade immunity from COVID vaccines?
How well can Omicron evade immunity from COVID vaccines?
 Does the world need an Omicron vaccine? What researchers say
Does the world need an Omicron vaccine? What researchers say