Abstract
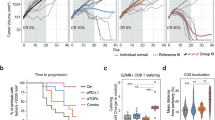
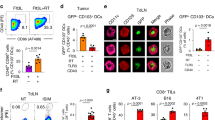
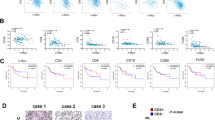
CD40 ligand (CD40L) is a potent stimulator of tumor immunity via its activation of dendritic cells, which in turn initiate T-cell activation. However, T cells are inhibited by suppressive myeloid cells, which constitute an important part of immune evasion. We hypothesized that CD40L may revert the function of suppressive myeloid cells to generate a T-cell stimulatory environment, and this was investigated in the murine bladder cancer model MB49/C57BL/6. Upon intratumoral adenoviral CD40L (AdCD40L) gene therapy, the infiltration of CD11b+Gr-1+ cells was significantly reduced, whereas activated T cells were increased. In vitro, CD40L-expressing MB49 cells tilted the myeloid subpopulations in favor of granulocytic CD11b+Gr-1high myeloid cells instead of monocytic CD11b+Gr-1int/low myeloid cells. Further, the level of macrophages in splenocyte co-cultures with MB49 cells was evaluated. In cultures with MB49 cells expressing CD40L, the overall level of macrophages was reduced and the remaining cells were differentiated into M1-like cells. Hence, these data support that CD40L tilts myeloid immune cell populations in favor of anti-tumor immunity (M1) instead of immunosuppression (CD11b+Gr-1int/low and M2), and this was accompanied by an increased level of activated T cells in the tumor tissue.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Gabrilovich DI, Ostrand-Rosenberg S, Bronte V . Coordinated regulation of myeloid cells by tumours. Nat Rev Immunol 2012; 12: 253–268.
Ribechini E, Greifenberg V, Sandwick S, Lutz MB . Subsets, expansion and activation of myeloid-derived suppressor cells. Med Microbiol Immunol 2010; 199: 273–281.
Gabrilovich DI, Nagaraj S . Myeloid-derived suppressor cells as regulators of the immune system. Nat Rev Immunol 2009; 9: 162–174.
Loskog AS, Eliopoulos AG . The Janus faces of CD40 in cancer. Semin Immunol 2009; 21: 301–307.
Essand M, Loskog AS . Genetically engineered T cells for the treatment of cancer. J Intern Med 2013; 273: 166–181.
Rosenberg SA, Dudley ME . Adoptive cell therapy for the treatment of patients with metastatic melanoma. Curr Opin Immunol 2009; 21: 233–240.
Cella M, Scheidegger D, Palmer-Lehmann K, Lane P, Lanzavecchia A, Alber G . Ligation of CD40 on dendritic cells triggers production of high levels of interleukin-12 and enhances T cell stimulatory capacity: T-T help via APC activation. J Exp Med 1996; 184: 747–752.
Lindqvist C, Sandin LC, Fransson M, Loskog A . Local AdCD40L gene therapy is effective for disseminated murine experimental cancer by breaking T-cell tolerance and inducing tumor cell growth inhibition. J Immunother 2009; 32: 785–792.
Loskog AS, Fransson ME, Totterman TT . AdCD40L gene therapy counteracts T regulatory cells and cures aggressive tumors in an orthotopic bladder cancer model. Clin Cancer Res 2005; 11 (24 Pt 1): 8816–8821.
Malmstrom PU, Loskog AS, Lindqvist CA, Mangsbo SM, Fransson M, Wanders A et al. AdCD40L immunogene therapy for bladder carcinoma—the first phase I/IIa trial. Clin Cancer Res 2010; 16: 3279–3287.
Westberg S, Sadeghi A, Svensson E, Segall T, Dimopoulou M, Korsgren O et al. Treatment efficacy and immune stimulation by AdCD40L gene therapy of spontaneous canine malignant melanoma. J Immunother 2013; 36: 350–358.
von Euler H, Sadeghi A, Carlsson B, Rivera P, Loskog A, Segall T et al. Efficient adenovector CD40 ligand immunotherapy of canine malignant melanoma. J Immunother 2008; 31: 377–384.
Loskog A, Dzojic H, Vikman S, Ninalga C, Essand M, Korsgren O et al. Adenovirus CD40 ligand gene therapy counteracts immune escape mechanisms in the tumor microenvironment. J Immunol 2004; 172: 7200–7205.
Ostrand-Rosenberg S, Sinha P . Myeloid-derived suppressor cells: linking inflammation and cancer. J Immunol 2009; 182: 4499–4506.
Matzinger P, Kamala T . Tissue-based class control: the other side of tolerance. Nat Rev Immunol 2011; 11: 221–230.
Pan PY, Ma G, Weber KJ, Ozao-Choy J, Wang G, Yin B et al. Immune stimulatory receptor CD40 is required for T-cell suppression and T regulatory cell activation mediated by myeloid-derived suppressor cells in cancer. Cancer Res 2010; 70: 99–108.
Dolcetti L, Peranzoni E, Ugel S, Marigo I, Fernandez Gomez A, Mesa C et al. Hierarchy of immunosuppressive strength among myeloid-derived suppressor cell subsets is determined by GM-CSF. Eur J Immunol 2010; 40: 22–35.
Movahedi K, Guilliams M, Van den Bossche J, Van den Bergh R, Gysemans C, Beschin A et al. Identification of discrete tumor-induced myeloid-derived suppressor cell subpopulations with distinct T cell-suppressive activity. Blood 2008; 111: 4233–4244.
Mangsbo SM, Sandin LC, Anger K, Korman AJ, Loskog A, Totterman TH . Enhanced tumor eradication by combining CTLA-4 or PD-1 blockade with CpG therapy. J Immunother 2010; 33: 225–235.
Hao NB, Lu MH, Fan YH, Cao YL, Zhang ZR, Yang SM . Macrophages in tumor microenvironments and the progression of tumors. Clin Dev Immunol 2012; 2012: 948098.
Bierie B, Moses HL . Transforming growth factor beta (TGF-beta) and inflammation in cancer. Cytokine Growth Factor Rev 2010; 21: 49–59.
Mocellin S, Marincola FM, Young HA . Interleukin-10 and the immune response against cancer: a counterpoint. J Leukoc Biol 2005; 78: 1043–1051.
Umemura N, Saio M, Suwa T, Kitoh Y, Bai J, Nonaka K et al. Tumor-infiltrating myeloid-derived suppressor cells are pleiotropic-inflamed monocytes/macrophages that bear M1- and M2-type characteristics. J Leukoc Biol 2008; 83: 1136–1144.
Youn JI, Nagaraj S, Collazo M, Gabrilovich DI . Subsets of myeloid-derived suppressor cells in tumor-bearing mice. J Immunol 2008; 181: 5791–5802.
Acknowledgements
We thank Liye Chen at the Uppsala University for helpful assistance. The project was financed by grants to AL from the Swedish Cancer Society, the Swedish Research Council, the EU FP6/Apotherapy, AFA Insurance AB, ALF Uppsala University Hospital funding and the Medical Faculty at the Uppsala University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
AL and SMM have a royalty agreement with Alligator Bioscience AB. Further, AL is the CEO of Lokon Pharma AB and a scientific advisor at NEXTTOBE AB. The other authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on Cancer Gene Therapy website
Supplementary information
Rights and permissions
About this article
Cite this article
Liljenfeldt, L., Dieterich, L., Dimberg, A. et al. CD40L gene therapy tilts the myeloid cell profile and promotes infiltration of activated T lymphocytes. Cancer Gene Ther 21, 95–102 (2014). https://doi.org/10.1038/cgt.2014.2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cgt.2014.2
Keywords
This article is cited by
-
Intratumoral immunostimulatory AdCD40L gene therapy in patients with advanced solid tumors
Cancer Gene Therapy (2021)
-
Adenovirus-mediated CD40L gene transfer increases Teffector/Tregulatory cell ratio and upregulates death receptors in metastatic melanoma patients
Journal of Translational Medicine (2017)
-
Activation of myeloid and endothelial cells by CD40L gene therapy supports T-cell expansion and migration into the tumor microenvironment
Gene Therapy (2017)