Abstract
PAX3 (paired box 3) is known to have an important role in melanocyte development through modulation of microphthalmia-associated transcription factor transcription. Here we found that PAX3 transcriptional activity could be regulated through FGF2 (basic fibroblast growth factor)-STAT3 (signal transducer and activator of transcription 3) signaling in the pigment cells. To study its function in vivo, we have generated a transgenic mouse model expressing PAX3 driven by tyrosinase promoter in a tissue-specific fashion. These animals exhibit hyperpigmentation in the epidermis, evident in the skin color of their ears and tails. We showed that the darker skin color results from both increased melanocyte numbers and melanin synthesis. Together, our study delineated a novel pathway in the melanocyte lineage, linking FGF2-STAT3 signaling to increased PAX3 transcription. Moreover, our results suggest that this pathway might contribute to the regulation of melanocyte numbers and melanin levels, and thereby provide an alternative strategy to induce pigmentation.
Similar content being viewed by others
Main
Basic fibroblast growth factor (FGF2) is an important keratinocyte-derived factor in mlenaocytes. It is known to regulate a wide range of biological processes, such as the development of limb and nervous system. Specifically in melanocytes, FGF2 is essential for their proliferation in vitro.1 However, the molecular mechanism of FGF2-induced melanogenesis response remains unclear. Signal transducer and activator of transcription 3 (STAT3) encode a transcription factor of STAT family.2, 3 Upon stimulation by several growth factors and cytokines, it becomes tyrosine phosphorylated and activated by receptor-associated kinases. It regulates diverse cellular processes through the modulation of gene transcription. Although these two signaling proteins have been well studied in many tissue types and biological processes, it is unclear whether STAT3 could mediate signaling from FGF2, especially in regulating pigmentation in the melanocytes.
PAX3 (paired box 3) transcription factor is required for the development of neural crest as well as several other tissue types.4 It modulates key regulators of melanocytes through transcriptional activation or repression. In humans, heterozygous PAX3 loss-of-function mutations are associated with Waardenburg syndrome type I, manifesting hypopigmentation of the hair and skin, pigmentation abnormality of the iris and premature hair graying.4 In mice, heterozygous PAX3 mutations result in defects in multiple tissues derived from the neural crest lineage including the patchy hypopigmentation in skin and fur, whereas homozygosity leads to prenatal or perinatal lethality.5 Here we have focused our attention on PAX3, a key transcriptional factor in the neural crest lineage from which the mature melanocytes arise. We sought to better understand the upstream signaling events regulating PAX3 activity and its key downstream effectors in the melanocytes, in order to characterize its role in regulating ultraviolet (UV)B-induced pigmentation.
We delineated a novel pathway that might contribute the regulation of UV-induced melanogenesis. We show that this pathway transduces signals through STAT3 from FGF2 to PAX3. Specifically, we found that UV-induced FGF2 secretion is required in low-dose UVB-induced pigment production, and FGF2-STAT3 signaling modulates PAX3 transcription in the melanocytes. Upon activation by FGF2, STAT3 binds to PAX3 upstream regulatory sequences to transactivate its promoter. Finally, we observed that constitutive PAX3 expression in melanocytes in vivo leads to upregulation of the melanocyte number and melanin synthesis, which produce a hyperpigmentation phenotype in the epidermis of these animals.
Results
UVB treatment induces FGF2 secretion from keratinocytes.
Previous data had suggested that the FGF2 is a paracrine factor from keratinocytes in the UV-induced melanogenesis response.1 Specifically, this function of FGF2 is shown to be mediated through UVB-induced FGF2 transcription in keratinocytes.6 To demonstrate that FGF2 secretion is also induced by UVB exposure, we irradiated human primary keratinocytes with UVB at 400 J/m2. We observed an induction of FGF2 section by UV that peaked at 24 h and was sustained until at least 48 h (Figure 1a), consistent with FGF2’s potential role in UVB-induced melanogenesis. In melanocytes, multiple studies have showed that FGF2 treatment can induce proliferation in vitro7 and pigmented lesions in grafts.8 Here we reproduced FGF2 proliferative effects in our in vitro cultured melanocytes (Supplementary Figure 1A), suggesting FGF2 might enhance pigmentation through promoting melanocyte proliferation and maybe other mechanisms.
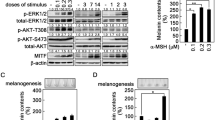
UVB induces FGF2 secretion from keratinocytes yet MITF protein is upregulated in melanocytes. (a) FGF2 is induced by UVB in vivo. FGF2 levels were measured by enzyme immunoassay in culture media at indicated time points. (b) The production of melanin granules, which were induced in skin constructs with HPK and human primary melanocyte (HPM) by a very low-dose UVB irradiation (25 J/m2 per day for 3 days), and inhibited by PD173074. Fontana–Masson staining in skin constructs, × 400. (c) HPMs were stimulated with FGF2 (10 nmol/l). RNA and protein were collected at time 0 and at different time points after stimulation. The mRNA expression of MITF was measured by quantitative RT-PCR and normalized to GAPDH. Protein level of MITF, STAT3, phosphorylated STAT3 at Tyr 705 and Ser 727, phosphorylated ERK1/2 was analyzed by western blot along with tubulin, which served a as loading control
To test the role of FGF2 in UV-induced melanogenesis, we used skin constructs to investigate the role of FGF2-induced melanogenesis response. Skin reconstructs exhibits in vivo-like morphological, ultrastructural characteristics and the production of melanin granules, which were induced in skin constructs with HPK and HPM by a very lose-dose UVB irradiation (25 J/m2 per day for 3 days) and progressively populated the layers of the tissue. Pigmentation production was undetectable in skin reconstructs if we add PD173074, a potent, cell-permeable and ATP-competitive inhibitor of FGF receptor, in the culture medium (Figure 1b). However, if skin constructs treated with PD173074 are irradiated by 100 J/m2 UVB for 4 days, we still can detect UVB-induced pigment production (data not shown). To ensure that PD173074 completely inhibits FGFR signaling even at high UVB dose, we examined MAPK (ERK1/2) activation in the whole protein extracts of skin constructs.9 This phosphorylation increase was completely abolished in skin constructs treated with PD173074 (Supplementary Figure 1). Together, this study indicates that FGF2 is required in a low-dose UVB-induced melanogenesis response, but not a high-dose UVB-induced melanogenesis response.
FGF2 upregulates PAX3 expression in melanocytes
Microphthalmia-associated transcription factor (MITF) is the key transcription regulator downstream of MSH/MC1R signaling to promote pigment synthesis upon UV irradiation.10, 11 We wondered whether MITF is also the primary effector of FGF2 in melanocytes. To test this hypothesis, we exposed primary human melanocytes to 10 nmol/l FGF2 (chosen based on previous report12) and measured MITF mRNA and protein levels at indicated time points after treatment. We saw a 4.5-fold induction of MITF mRNA expression by 24 h (Figure 1c). MITF protein level, especially the phospho-MITF level, was minimally affected by FGF2 (Figure 1c), suggesting that it is unlikely that FGF2 exerts its effects in melanocytes through promoting MITF activity.
We then looked at the expression levels of another key melanocyte transcriptional regulator, PAX3, upon FGF2 stimulation as previous reports have demonstrated that MITF is directly regulated by PAX3 in the transcriptional level.13, 14, 15, 16 We observed a nearly 15-fold induction of PAX3 mRNA by 24 h, accompanied by a marked induction of PAX3 protein expression (Figure 2a). In addition, we asked whether PAX3 expression is induced by FGF2 in a dose-dependent manner (Figure 2b). Indeed, PAX3 mRNA and protein levels are increasingly upregulated with escalating doses of FGF2 (Figure 2b). Together, these results suggest that PAX3 could be an important transcription factor that mediates FGF2-induced melanogenesis.
FGF2 upregulates PAX3 mRNA and protein levels in melanocytes. (a) Human primary melanocytes (HPMs) were stimulated with FGF2 (10 nmol/l). RNA and protein were collected at time 0 and at different time points after stimulation. (b) FGF2-induced PAX3 upregulation is in a FGF2 dose-dependent manner. HPMs were stimulated with different doses of FGF2 as indicated. RNA and protein were collected at 6 h after FGF2 stimulation. The mRNA expression of PAX3 was measured by quantitative RT-PCR and normalized to GAPDH. Protein level of PAX3 was analyzed by western blot along with tubulin, which served as a loading control
STAT3 tyrosine phosphorylation is stimulated by FGF2 in melanocytes and important for melanocyte proliferation
As the upregulation of PAX3 expression is at the mRNA level upon FGF2 stimulation (Figure 2a), we wondered which factor is transducing the signal from FGF2 to upregulate PAX3 transcripts. Previous reports have demonstrated that FGF2 induces melanocyte proliferation, which is mediated by MEK1/2-ERK1/2 signaling. Our studies have confirmed this conclusion (Figure 1). ERK1/2 usually regulates the activities of several transcription factors, including C-myc, CREB and C-Fos.17 To identify which one of them mediates FGF2-induced PAX3 upregulation in melanocytes, we reduced the expression of each of them by siRNA approach and then detected FGF2-induced PAX3 upregulation in melanocytes. Unfortunately, knockdown of anyone of them cannot block FGF2-induced PAX3 upregulation (data not shown). Previous reports also demonstrated that ERK family of mitogen-activated protein kinases, but not JNK or p38, phosphorylates and activates STAT3 in response to growth factors.18 We therefore detected the phosphorylation of STAT3 at tyrosine 705 (Y705) and serine 727 (S727) in melanocytes after the stimulation of FGF2 in human primary melanocytes. Upon FGF2 stimulation, tyrosine phosphorylation of STAT3 at Y705 and S727 was markedly upregulated by 1 h and sustained until at least 24 h (Figure 1c).
In parallel, we investigated whether the effects of FGF2 in melanocytes were inhibited by the specific STAT3 inhibitor, stattic19 in human primary melanocytes. We found that FGF2-induced melanocyte proliferation was abolished if STAT3 was inhibited in human primary melanocytes (Supplementary Figure 2). This result supports STAT3 being an important downstream effector of FGF2 in regulating melanocyte proliferation.
STAT3 activation is required for FGF2-induced PAX3 transcription
To determine whether FGF2-induced PAX3 expression is STAT3 activation dependent, we assayed PAX3 mRNA and protein levels upon FGF2 simulation in the presence of either STAT3 small molecular inhibitors or siRNAs. First, we examined the effects of Stattic, a STAT3 inhibitor,19 and FLLL31, a STAT3 phosphorylation inhibitor,20 on the intervention of FGF2-induced STAT3 phosphorylation and PAX3 upregulation. As shown in Figures 3a and b, inhibition of STAT3 or STAT3 phosphorylation diminished FGF2-induced PAX3 upregulation. These results support the notion that the activation of STAT3 is required for the FGF2-induced PAX3 upregulation.
Phospho STAT3 is required for FGF2-induced PAX3 upregulation. Human primary melanocytes cells were treated with specific STAT3 inhibitor, stattic (2 μmol/l) or STAT3 phosphorylation inhibitor FLLL31 (5 μmol/l) together with FGF2 (10 nmol/l) for 6 h. Cells were processed for RNA and protein isolation. PAX3 expression was detected by real-time PCR (a) and western blot (b). FGF2 induction of PAX3 RNA and protein are inhibited after STAT3 is inhibited or STAT3 phosphorylation is inhibited. (b) B16 melanoma cells with siSTAT3 were stimulated with FGF2 (10 nmol/l). RNA and protein were collected at 6 h after stimulation. PAX3 RNA levels as measured by quantitative RT-PCR and normalized to GAPDH (c). Protein levels of phosphorylated STAT3 at Tyr 705 were analyzed by western blot, are shown on the right along with tubulin, which served a as loading control (d)
Next, we explored the need for STAT3 protein in PAX3 upregulation after FGF2 stimulation using the RNAi knockdown approach. In particular, we reduced the expression of STAT3 by specific siRNA in B16 mouse melanoma cells (Figure 3c) and the PAX3 expression was measured at 6 h after FGF2 stimulation. As shown in Figures 3c and d, FGF2 stimulation upregulated the expression of PAX3 in both mRNA and protein levels in control siRNA-expressing cells. In contrast, similar levels of PAX3 expression were detected in B16 cells treated with both FGF2 and siSTAT3 compared with untreated control cells. In other words, the stimulation effect of FGF2 was abolished in the presence of siSTAT3. Together, these results indicate that STAT3 is required for the FGF2-induced PAX3 upregulation.
PAX3 is a direct transcriptional target of STAT3
As STAT3 is a transcription factor,21, 22 we wondered whether STAT3 could mediate PAX3 mRNA upregulation through direct transactivation of its promoter. To test this hypothesis, we first examined the proximal 1-kb promoter region of PAX3 to search for the consensus STAT3-binding element (SBE).23, 24 Indeed, we found three potential general STAT3-binding sites at −846 to −837, −769 to −760 and −60 to −51 and one selective STAT3-binding site at −139 to −131 in the PAX3 5′-flanking region ∼1000 bp upstream of the transcription initiation site in humans (Figure 4a). The −60 to −51 SBE is conserved among human, monkey and rat (Figure 4a). To further identify which SBE(s) were bound by STAT3 in PAX3 promoter, we performed chromatin immunoprecipitation (ChIP) analysis. ChIPs analysis revealed that SBEs near the transcription start site of PAX3 promoter were amplified from crosslinked chromatin isolated from FGF2-treated human primary melanocytes and immunoprecipitated with specific anti-STAT3 antibody, but not from multiple controls. Furthermore, to identify which SBE near the transcription start site of PAX3 promoter is essential for FGF2-induced activation of PAX3 transcription, a series of previously reported16 PAX3-promoter-luciferase reporters were tested for FGF2 responsiveness in B16 mouse melanoma cells. Site-specific mutation at the evolutionarily conserved STAT3-binding site (−60 to −51) abolished the FGF2-induced activation of PAX3 promoter whereas mutation at −139 to −131 site had minimal effects on the reporter activity (Figure 4c). This data indicates that the SBE of PAX3 promoter at −60 to −51 from the transcriptional start site mediates its transactivation by STAT3. Together, these data suggest that STAT3 directly modulates activation of the PAX3 promoter.
STAT3 is a direct transactivator of PAX3 promoter in melanocytes. (a) Schematic representation of the human PAX3 locus, which indicates the locations of a STAT3-SBE that is highly conserved among human (−60 to −51), rat (−24 to −16) and monkey (−60 to −51) promoters. (b) In vivo association of STAT3 with the PAX3 promoter by chromatin immunoprecipitation. (c) Analysis of PAX3 promoter transactivation capacity by FGF2 stimulation of B16 cells transiently transfected with PAX3 reporter, employed mutations at STAT3-binding sites (Mut). Promoter activity was studied using reporter assays with different promoter portions (including mutated SBEs) to drive expression of firefly luciferase. Relative luciferase activity was normalized to empty pGL4-basic plasmid using the dual-luciferase assay system (Promega, Madison, WI, USA)
Constitutive PAX3 expression in melanocytes leads to hyperpigmentation in vivo
To further address the role of PAX3 in melanocytes in vivo, we generated a transgenic mouse with melanocyte-specific PAX3 expression under the control of tyrosinase (Tyr) enhancer/promoter (Figure 5a). The PAX3 expression construct was designed such that the PAX3 gene (NM_181457), cloned from human primary melanocytes, was inserted into the vector (pNEB 193) downstream of the murine tyrosinase locus control region (enhancer/promoter) (Figure 5a). This construct was injected into single-cell embryos of F1 C57BL/J6XCBA mice and the offspring were genotyped by Southern blot analysis (Figure 5b) and PCR (Figure 5c). One founder line was studied in detail, in which the relative protein expression level of the PAX3 transgene is nearly equal in comparison to the endogenous protein level in the epidermis of neonatal mice (Figure 5d). RT-PCR analysis shows that the transgene was expressed in the skin and brain, but not in the heart, kidney, lung or liver (Figure 5e), with the expression in the skin resulting most likely from expression in melanocytes. In comparison with wild-type C57BL/J6XCBA mice, the animals that overexpressed PAX3 have hyperpigmented skin, most evident in the ear pinnae and tail, but normal coat color (Figure 5f). Epidermal melanin assay demonstrated that hyperpigmentation was consequent to an increased melanin production (Figure 5g). Furthermore, no melanoma was diagnosed in transgenic PAX3 mice to date (24 week old).
Hyperpigmentation was detected in transgenic PAX3 mouse. (a) Schematic diagrams of Tyr-PAX3 construct. (b) Southern blot analysis of transgene. (c) PCR analysis of transgene. (d) PAX3 protein expression in primary melanocytes isolated from wild-type and transgenic PAX3 mouse. (e) RT-PCR analysis of transgene expression in the skin, liver, lung, heart, brain and kidney. (f) The ears and tails of transgenic PAX3 mice is uniformly hyperpigmented. (g) The epidermal melanocytes were quantified by flow cytometry assays with anti-Trp2 antibody (*P<0.001 by t-test). (h) Melanin concentration was detected in 5-weeks-old mouse ear epidermis as described
To identify whether melanin production is a result from melanocyte proliferation, differentiation or both, we used FACS analysis to isolate and quantify melanocytes from mouse epidermis. The Anti-Trp2 antibody was used as a marker of melanocyte and confirmed by anti-CD31 antibody.25, 26 To detail the melanocyte populations expressing the transgene, PAX3 expression was tested by immunohistochemical staining in hair follicles and epidermis of wild-type and PAX3-tg mice. In hair follicles, the PAX3-specific antibody showed stronger nuclear staining in PAX3-tg mice than in wild-type mice (Supplementary Figure 3). As shown in Figure 5h, the number of epidermal melanocytes is significantly higher in PAX3-tg mice than in wild-type mice (≥1.5-fold). Together, these results support the notion that hyperpigmentation in PAX3-tg mice is caused by PAX3-induced melanocyte hyper-proliferation and increased melanin synthesis. More importantly, this experiment demonstrated that PAX3 gain-of-function enhances pigmentation in vivo.
Discussion
Here we delineate a new signaling pathway in UVB-induced melanogenesis response. In this signaling pathway, UVB radiation stimulates FGF2 protein secretion from human keratinocytes, which in turn induces PAX3 transcription through activation of STAT3 in melanocytes. Specifically, we showed that PAX3 is regulated by STAT3 protein at the transcriptional level directly (Figure 6).
Here we found UVB radiation stimulated FGF2 protein secretion from human keratinocytes, and FGF2 is required in a low-dose UVB-induced pigment production, but not in a high-dose UVB-induced pigment production. We showed that FGF2 augmented PAX3 transcription through activation of STAT3 in melanocytes. These results also indicate that alternative signaling pathways are induced in an UVB-dose-dependent manner. It remains to be determined whether other signaling pathways could also modulate pigment induction upon UV exposure and to what extend each pathway contributes to the phenotype. Importantly, identification and comprehensive characterization of these pathways could help to build the entire signaling network linking the external stimulus (i.e. UV irradiation) to the eventual biological outcomes (i.e. pigmentation and prevention/repair of UV damage).
ChIP revealed that the STAT3 protein specifically occupies the endogenous PAX3 promoter after FGF2 stimulation in human primary melanocytes. Our previous report has demonstrated that the −200 bp relative to the transcription start site of PAX3 is crucial for regulating PAX3 transcription.16 In this region of PAX3 promoter, a Smad-binding element (−85 to −107)16 and a myc-binding element (−110 to −105)27 were identified. Here, we further identified a SBE (−60 to −51) in this region. Other critical transcriptional regulatory regions in PAX3 promoter include binding sites for POU transcription factors at −1359 to −1334 and −856 to −827,28, 29 and Teads transcription factors at NCE1 and NCE2 (both around −160030 relative to the transcription start site of PAX3). The contribution of each transcription factor and their interactions in the regulation of PAX3 transcription, especially after UV radiation, should be further characterized.
STAT3 is a known downstream effector of several growth factor receptors.2, 3 It has been shown that FGF2 could activate STAT3 in some cellular contexts.31, 32, 33 Yet it remains unclear whether STAT3 directly associates with FGFRs, or if its activation is mediated through cross-talk between signaling pathways. STAT3 regulates the transcription of genes with diverse functions, including immunity, apoptosis, angiogenesis and proliferation.2 Here we found that upon FGF2 simulation, STAT3 becomes activated and in turn directly transactivates PAX3 promoter in melanocytes. In other words, our results identified PAX3 as a novel direct target of STAT3, suggested a novel function of STAT3 in the pigment cell lineage, and broadened our understanding of the transcriptional network involved in regulation of melanogenesis.
To better understand the role of PAX3 in differentiated melanocytes, we have generated a transgenic mouse model in which PAX3 transgene expression is under the control of tyrosine promoter. There was a twofold increase in the PAX3 protein level in the transgenic mice. Importantly, we observed hyperpigmentation in the skin of these mice resulting from both increased melanocyte cell number and increased pigment production. These results suggest that PAX3 indeed promotes proliferation and stimulates melanin synthesis in mature melanocytes, independent of its known role during melanocyte development. These observations also support the hypothesis that PAX3 could contribute to pigment induction upon UVB irradiation in these cells.16 Together, this mouse model provides an excellent tool to understand PAX3 function in differentiated melanocytes in vivo.
Materials and Methods
Cell lines, reagents and UV exposure
Primary keratinocytes and melanocytes were isolated from normal discarded foreskins as described.34, 35 FGFb (FGF2), Stattic and FLLL31 were purchased from Sigma (St. Louis, MO, USA). Short hairpin RNA duplexes that target PAX3 were purchased from Origene (Rockville, MD, USA) and described elsewhere.16
Reconstructed skin
Skin reconstructs were generated using MelanoDerm (MatTek Corp., Ashland, MA, USA) with modifications.
Generation of PAX3 transgenic mice
The mouse tyrosinase enhancer, promoter and SV40 poly-A tail was purchased from Addgene (Cambridge, MA, USA) and originally generated by Dr. Marcus at Dr. Lynda Chin's lab (Dana Farber Cancer Institute, Boston, MA, USA). Human PAX3 cDNA was inserted downstream of the tyrosinase enhancer promoter. The DNA construct was gel purified (Qiagen, Valencia, CA, USA), sequenced and microinjected into single-cell embryos of F1 C57BL/6 mice at Transgenic Core Facility of Boston University.
Melanin concentration detection
Mouse ear epidermal separation was described elsewhere.36 The epidermis was treated with 0.5% sodium dodecyl sulfate in 0.05 M Tris buffer (pH 7.0) containing 2 mg/ml Pronase-CB. Mixtures were incubated at 30 °C for 24 h. The digests were collected by centrifuge (10 000 × g for 30 min). Melanin was obtained by centrifugation and washing three times with 4 ml KCI solution (0.01%) and then solutes it in 0.1 N NaOH. The melanin content was detected by spectrophotometer at absorbance 400 nm.37, 38
Western blotting
Western blotting was performed using the following antibodies: anti-PAX3 (The Developmental Studies Hybridoma Bank at the University of Iowa, Iowa, IA, USA); anti-MITF (C5, generously provided by Dr. David E Fisher at Massachusetts General Hospital, Boston, MA, USA); anti-p-STAT3 (Tyr705) (Cell Signaling, Beverly, MA, USA); anti-p-STAT3 (Ser727) (Cell Signaling); anti-STAT3 (9D8, Thermo Scientific, Beverly, MA, USA); anti-p-ERK1/2 (Cell Signaling); and anti-tubulin (Sigma).
Abbreviations
- FGF2:
-
basic fibroblast growth factor
- ChIP:
-
chromatin immunoprecipitation
- MITF:
-
microphthalmia-associated transcription factor
- PAX3:
-
paired box 3
- STAT3:
-
signal transducer and activator of transcription 3
- UV:
-
ultraviolet
References
Halaban R . The regulation of normal melanocyte proliferation. Pigment Cell Res 2000; 13: 4–14.
Bromberg J, Darnell Jr JE . The role of STATs in transcriptional control and their impact on cellular function. Oncogene 2000; 19: 2468–2473.
Kano A, Wolfgang MJ, Gao Q, Jacoby J, Chai GX, Hansen W et al. Endothelial cells require STAT3 for protection against endotoxin-induced inflammation. J Exp Med 2003; 198: 1517–1525.
Pingault V, Ente D, Dastot-Le Moal F, Goossens M, Marlin S, Bondurand N . Review and update of mutations causing Waardenburg syndrome. Hum Mutat 2010; 31: 391–406.
Epstein DJ, Vogan KJ, Trasler DG, Gros P . A mutation within intron 3 of the Pax-3 gene produces aberrantly spliced mRNA transcripts in the splotch (Sp) mouse mutant. Proc Natl Acad Sci USA 1993; 90: 532–536.
Brenner M, Degitz K, Besch R, Berking C . Differential expression of melanoma-associated growth factors in keratinocytes and fibroblasts by ultraviolet A and ultraviolet B radiation. Br J Dermatol 2005; 153: 733–739.
Imokawa G, Yada Y, Miyagishi M . Endothelins secreted from human keratinocytes are intrinsic mitogens for human melanocytes. J Biol Chem 1992; 267: 24675–24680.
Berking C, Takemoto R, Satyamoorthy K, Elenitsas R, Herlyn M . Basic fibroblast growth factor and ultraviolet B transform melanocytes in human skin. Am J Pathol 2001; 158: 943–953.
Bansal R, Magge S, Winkler S . Specific inhibitor of FGF receptor signaling: FGF-2-mediated effects on proliferation, differentiation, and MAPK activation are inhibited by PD173074 in oligodendrocyte-lineage cells. J Neurosci Res 2003; 74: 486–493.
Lin JY, Fisher DE . Melanocyte biology and skin pigmentation. Nature 2007; 445: 843–850.
Goding CR . Mitf from neural crest to melanoma: signal transduction and transcription in the melanocyte lineage. Genes Dev 2000; 14: 1712–1728.
Yang X, Qiao D, Meyer K, Friedl A . Signal transducers and activators of transcription mediate fibroblast growth factor-induced vascular endothelial morphogenesis. Cancer Res 2009; 69: 1668–1677.
Bondurand N, Pingault V, Goerich DE, Lemort N, Sock E, Le Caignec C et al. Interaction among SOX10, PAX3 and MITF, three genes altered in Waardenburg syndrome. Hum Mol Genet 2000; 9: 1907–1917.
Lang D, Epstein JA . Sox10 and Pax3 physically interact to mediate activation of a conserved c-RET enhancer. Hum Mol Genet 2003; 12: 937–945.
Potterf SB, Furumura M, Dunn KJ, Arnheiter H, Pavan WJ . Transcription factor hierarchy in Waardenburg syndrome: regulation of MITF expression by SOX10 and PAX3. Hum Genet 2000; 107: 1–6.
Yang G, Li Y, Nishimura EK, Xin H, Zhou A, Guo Y et al. Inhibition of PAX3 by TGF-beta modulates melanocyte viability. Mol Cell 2008; 32: 554–563.
Balmanno K, Cook SJ . Tumour cell survival signalling by the ERK1/2 pathway. Cell Death Differ 2009; 16: 368–377.
Chung J, Uchida E, Grammer TC, Blenis J . STAT3 serine phosphorylation by ERK-dependent and -independent pathways negatively modulates its tyrosine phosphorylation. Mol Cell Biol 1997; 17: 6508–6516.
Schust J SB, Hollis A, Mayer TU, Berg T . Stattic: a small-molecule inhibitor of STAT3 activation and dimerization. Chem Biol 2006; 13: 1235–1242.
Lin L, Hutzen B, Zuo M, Ball S, Deangelis S, Foust E et al. Novel STAT3 phosphorylation inhibitors exhibit potent growth-suppressive activity in pancreatic and breast cancer cells. Cancer Res 2010; 70: 2445–2454.
Yu H, Pardoll D, Jove R . STATs in cancer inflammation and immunity: a leading role for STAT3. Nat Rev Cancer 2009; 9: 798–809.
Sano S, Chan KS, DiGiovanni J . Impact of Stat3 activation upon skin biology: a dichotomy of its role between homeostasis and diseases. J Dermatol Sci 2008; 50: 1–14.
Darnell Jr JE, Kerr IM, Stark GR . Jak-STAT pathways and transcriptional activation in response to IFNs and other extracellular signaling proteins. Science 1994; 264: 1415–1421.
Seidel HM, Milocco LH, Lamb P, Darnell Jr JE, Stein RB, Rosen J . Spacing of palindromic half sites as a determinant of selective STAT (signal transducers and activators of transcription) DNA binding and transcriptional activity. Proc Natl Acad Sci USA 1995; 92: 3041–3045.
Orchard GE, Calonje E . The effect of melanin bleaching on immunohistochemical staining in heavily pigmented melanocytic neoplasms. Am J Dermatopathol 1998; 20: 357–361.
Burns T, Breathnach S, Cox N, Griffiths C (eds). The Melanocyte. Rook’s Textbook Dermatol, vol. 8. Blackwell Publishing: Oxford, UK, 2010, pp 58–60.
Harris RG, White E, Phillips ES, Lillycrop KA . The expression of the developmentally regulated proto-oncogene Pax-3 is modulated by N-Myc. J Biol Chem 2002; 277: 34815–34825.
Pruitt SC, Bussman A, Maslov AY, Natoli TA, Heinaman R . Hox/Pbx and Brn binding sites mediate Pax3 expression in vitro and in vivo. Gene Expr Patterns 2004; 4: 671–685.
Smit DJ, Smith AG, Parsons PG, Muscat GE, Sturm RA . Domains of Brn-2 that mediate homodimerization and interaction with general and melanocytic transcription factors. Eur J Biochem 2000; 267: 6413–6422.
Milewski RC, Chi NC, Li J, Brown C, Lu MM, Epstein JA . Identification of minimal enhancer elements sufficient for Pax3 expression in neural crest and implication of Tead2 as a regulator of Pax3. Development 2004; 131: 829–837.
Xie TX, Huang FJ, Aldape KD, Kang SH, Liu M, Gershenwald JE et al. Activation of stat3 in human melanoma promotes brain metastasis. Cancer Res 2006; 66: 3188–3196.
Deo DD, Axelrad TW, Robert EG, Marcheselli V, Bazan NG, Hunt JD . Phosphorylation of STAT-3 in response to basic fibroblast growth factor occurs through a mechanism involving platelet-activating factor, JAK-2, and Src in human umbilical vein endothelial cells. Evidence for a dual kinase mechanism. J Biol Chem 2002; 277: 21237–21245.
Sahni M, Ambrosetti DC, Mansukhani A, Gertner R, Levy D, Basilico C . FGF signaling inhibits chondrocyte proliferation and regulates bone development through the STAT-1 pathway. Genes Dev 1999; 13: 1361–1366.
Horikawa T, Norris DA, Zekman T, Morelli JG . Effective elimination of fibroblasts in cultures of melanocytes by lowering calcium concentration in TPA depleted medium following geneticin treatment. Pigment Cell Res 1996; 9: 58–62.
Dunham WR, Klein SB, Rhodes LM, Marcelo CL . Oleic acid and linoleic acid are the major determinants of changes in keratinocyte plasma membrane viscosity. J Invest Dermatol 1996; 107: 332–335.
Dong L WJ, Zhang X, Zhang B, Dong F, Ziegler N, Mysz M et al. Melanocytes-stimulating Hormone, a potential activator of XPA DNA repair. Cancer Res 2010; 70: 3547–3556.
Watts KP, Fairchild RG, Slatkin DN, Greenberg D, Packer S, Atkins HL et al. Melanin content of hamster tissues, human tissues, and various melanomas. Cancer Res 1981; 41: 467–472.
Das KC, Abramson MB, Katzman R . Neuronal pigments: spectroscopic characterization of human brain melanin. J Neurochem 1978; 30: 601–605.
Acknowledgements
The PAX3 monoclonal antibody developed by Dr. Charles P Ordahl was obtained from the Developmental Studies Hybridoma Bank developed under the auspices of the NICHD and maintained by the University of Iowa, Department of Biology, Iowa City, IA, USA. This work was supported by National Institutes of Health (7RO1CA137098 for RC), American Cancer Society (RSG-09-022-01-CNE) and The Harry J. Lloyd Charitable Trust (RC). RC is an American Cancer Society Research Scholar.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Edited by RA Knight
Supplementary Information accompanies the paper on Cell Death and Differentiation website
Supplementary information
Rights and permissions
This work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Dong, L., Li, Y., Cao, J. et al. FGF2 regulates melanocytes viability through the STAT3-transactivated PAX3 transcription. Cell Death Differ 19, 616–622 (2012). https://doi.org/10.1038/cdd.2011.132
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cdd.2011.132
Keywords
This article is cited by
-
Single-cell sequencing reveals increased LAMB3-positive basal keratinocytes and ZNF90-positive fibroblasts in autologous cultured epithelium
Communications Biology (2024)
-
Efficient prioritization of CRISPR screen hits by accounting for targeting efficiency of guide RNA
BMC Biology (2023)
-
Differentiation of adipose-derived stem cells to functional CD105neg CD73low melanocyte precursors guided by defined culture condition
Stem Cell Research & Therapy (2019)
-
A STATement on Vemurafenib-Resistant Melanoma
Journal of Investigative Dermatology (2013)
-
Functional Melanocytes Are Readily Reprogrammable from Multilineage-Differentiating Stress-Enduring (Muse) Cells, Distinct Stem Cells in Human Fibroblasts
Journal of Investigative Dermatology (2013)