Abstract
Electrophoretic analysis of 18 allozyme loci was used to estimate the levels and structuring of genetic variation within and among natural populations of the protected endemic palm species from the Canary Islands (Phoenix canariensis) to evaluate its genetic relationship with the widespread congener P. dactylifera, and to assess comparatively the genetic variation in the populations where the two species coexist with morphologically intermediate plants (mixed populations). Our survey revealed that the within-population component explains roughly 75% of the genetic variation levels detected in P. canariensis (A=1.59; P=41.8; He=0.158), which rank higher than those reported for other species of the Arecaceae. A Principal Component analysis (PCA) based on allele frequencies consistently separates populations of P. canariensis and P. dactylifera, and reveals a close genetic relationship between P. canariensis and the mixed populations. Reduced levels of genetic variation in P. canariensis with respect to P. dactylifera, the fact that the genetic makeup of the Canarian endemic (with no unique alleles) is a subset of that found in P. dactylifera, and the high genetic identity between both species strongly suggest that P. canariensis is recently derived from a common ancestor closely related to P. dactylifera.
Similar content being viewed by others
Introduction
Phoenix canariensis Hort. ex Chabaud and P. dactylifera L. are diploid (n=18), long-lived, dioecious, arborescent monocots that are closely related (Barrow, 1998; Sosa et al, 1998) and exhibit strikingly different distribution ranges. While P. canariensis is endemic to the Canary Islands (Kunkel and Kunkel, 1974), P. dactylifera is distributed from western Asia to north-eastern Africa, including the Canarian archipelago. The need to conserve P. canariensis has been formalised by the Canarian Government through approving a decree that recognises its threatened status (BOC, 1991), and its communities are included in the European Habitat Directive (92/43/EEC) as priority. However, the implementation of sensible management strategies to warrant long-term survival of this Canarian endemic has been stalled by the lack of knowledge regarding the levels and distribution of its genetic variability. Although Sosa et al (1998) reported data on the levels of isozyme genetic variation within P. canariensis, only two populations from Gran Canaria were analysed in that study.
The population structure of the Canarian endemic P. canariensis presents several interlinked problems that interfere with the design of strategies to conserve its threatened variability. One of them stems from the difficulty of establishing the identity of individuals or populations for anthropogenic and biological reasons. For example, Canarian farmers have often introduced date palm (P. dactylifera) specimens in natural populations of the Canary palm (P. canariensis) for enhanced exploitation (Santana and Toledo, 1997). However, the lack of historical records and the ad hoc nature of these introductions make it difficult to determine where planting was performed and where populations grew naturally.
Closely related to the above, the distinction of P. canariensis from P. dactylifera is at present based solely on morphological traits, and it would be easily accomplished if the differences among individuals of both species were as obvious as described by different authors (Kunkel and Kunkel, 1974; Chabaud, 1882; Barrow, 1998), who agree in that P. canariensis has a bushier crown, darker green leaves and a thicker trunk with no offshoots, and smaller fruits than P. dactylifera. However, in nature, juvenile individuals of both species are impossible to distinguish using these indicators, and we frequently find adult specimens that share features of both species.
In addition, hybridisation between P. dactylifera and P. canariensis in nature has been hypothesised by different authors (Kunkel and Kunkel, 1974; Hodel, 1995; Barrow, 1998; Morici, 1998; Sosa et al, 1998). The ability of P. canariensis to hybridise with P. dactylifera might pose the biggest problem for conservation, because it could foster the coexistence of both species and their supposed hybrids in stands where planting was carried out and because it would be difficult to detect and remove the hybrid individuals (Barrow, 1998).
Unwitting planting in the past, the inherent plasticity of morphological traits and the apparent hybridisation of P. dactylifera with P. canariensis hinder the characterisation of many palm populations in the Canaries.
Allozyme polymorphisms have provided valuable data to assess the genetic variability of endemic species (Allphin et al, 1998; Godt and Hamrick, 1998; Batista et al, 2001), and their population genetic structuring (Schnabel et al, 1991; Shapcott, 1995; Caujapé-Castells et al, 1999). They have also been used successfully to characterise and differentiate close by related species, ecological varieties or natural and artificial hybrids (Rieseberg et al, 1989; Booij et al, 1995; Bendiab et al, 1998; Elisiário et al, 1999). Therefore, they could provide valuable insights into the genetic problems posed by the Canarian endemic P. canariensis.
Our aims in this paper are to (1) explore the levels of genetic isozymic variation in Phoenix populations from the Canary Islands; (2) analyse the genetic structure of the natural populations of P. canariensis; (3) compare P. dactylifera, P. canariensis and their putative hybrids using allozyme variation and (4) use the resulting information to provide practical guidelines for the conservation genetics of P. canariensis.
Materials and methods
Plant material
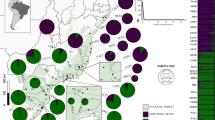
We sampled 20 populations (Figure 1) that were classified into one of three classes according to morphological traits: (i) nine natural populations of phenotipically pure P. canariensis localised in La Palma and La Gomera (where P. dactylifera has not been recorded; Izquierdo et al, 2001), and in very isolated areas of Tenerife and Gran Canaria located several kilometres from the nearest P. dactylifera population; (ii) four populations of phenotipically pure P. dactylifera: three of them imported recently from the Mediterranean region and planted in landscape areas, and one from Elche (a locality in the mid east of Spain); and (iii) seven populations from Gran Canaria, Lanzarote and Fuerteventura that contained individuals of P. canariensis, P. dactylifera and a continuous range of morphologically intermediate plants, designated mixed populations. Hybrid zones rarely represent a single generation and hybrids from these zones are a mosaic of parental and intermediate characters rather than solely intermediate ones (Rieseberg and Ellstrand, 1993).
Populations sampled of Phoenix canariensis (•), Phoenix dactylifera (▪) and mixed populations (▴) in the Canary Islands. Population codes are as in Table 1.
Finally, we also included one population of P. theophrasti (a Mediterranean species from the Island of Crete, Barrow, 1998) that was used as an external reference.
The leaves of the sampled individuals were kept in a portable cooler until subsequent manipulation in the laboratory, where they were rinsed with distilled water and dried. After this procedure, the samples were kept at −80°C until electrophoresed.
Electrophoretic analyses
Enzymes were extracted by crushing leaves with liquid nitrogen using a mortar and pestle until a fine-grained powder was obtained. Subsequently, an extraction buffer (Torres and Tisserat, 1980) was added to the cold, powdered leaf material to dissolve and stabilise the enzymes. Enzyme extracts were absorbed onto Whatman n°3 paper wicks and kept at −80°C until analysed electrophoretically.
Horizontal starch gel electrophoresis was carried out for 10 isozyme systems, namely aconitase (ACO, EC. 4.2.1.3), alcohol dehydrogenase (ADH, EC. 1.1.1), glucose- 6-phosphate dehydrogenase (G6PDH, EC. 1.1.1.49), isocitrate dehydrogenase (IDH, EC. 1.1.1.42), malate dehydrogenase (MDH, EC. 1.1.1.37), malic enzyme (ME, EC. 1.1.1.40), phosphoglucose isomerase (PGI, EC. 5.3.1.9), phosphoglucose mutase (PGM, EC. 5.4.2.2), shikimate dehydrogenase (SDH, EC. 1.1.1.25) and 6-phosphogluconate dehydrogenase (6pgdh, EC. 1.1.1.44).
Electrophoresis was conducted in 12% starch gels in different electrode/gel buffer systems. For ADH, G6PDH, IDH, MDH, ME, SDH and 6PGDH, we used Morpholine-Citrate 6.1/Morpholine-Citrate 6.1 (Clayton and Tretiak, 1972). Systems ACO and PGI were resolved on Boric acid pH 8.7/Tris Citrate pH 7.9 (Torres and Tisserat, 1980). Finally, PGM was resolved on Tris-Citrate pH 7/Histidine-Citrate 5.7 (Stuber et al, 1977). All the staining recipes were based on Wendel and Weeden (1989), although, for some isozymes, some modifications related to substrate concentration and final pHs of the staining solutions were introduced to improve band resolution.
Data interpretation
For each locus, the alleles were labelled and ordered following the alphabetical sequence in order of their mobility towards the anode. In most cases, the number of bands in heterozygous individuals was consistent with the expected quaternary structures of the corresponding enzyme (Wendel and Weeden, 1989), although null alleles were inferred in Mdh-1 and Pgm-1 (González-Pérez, 2001).
Data analysis
Elementary genetic allozymic variability statistics, genetic diversity indices (Nei, 1973) and genetic distance values (Nei, 1972) were calculated from genotype data per population using BIOSYS-1 (Swofford and Selander, 1981) and GENSTAT-PC 3.31 (Lewis, 1993).
Exact Hardy–Weinberg tests to measure the significance of deviations from the null hypothesis of random union of gametes (Weir, 1990; Guo and Thompson, 1992) were carried out on natural populations of P. canariensis and on the mixed populations using GENEPOP 3.1 (Raymond and Rousset, 1997). Nei's (1973) statistics of intra- and interpopulation variation (GST, HT, DST) were calculated with GENSTAT-PC 3.31 (Lewis, 1993).
A multivariate representation of the palm populations sampled was carried out by subjecting allele frequencies to a Principal Component Analysis (PCA) in SPSS version 10.0 (SPSS Inc., Chicago, IL, USA). The genetic differentiation among individual populations and among the three classes of populations identified was studied using the genetic differentiation coefficient (FST) calculated through the genetic analysis software GENEPOP 3.1 (Raymond and Rousset, 1997).
Results
Allozyme diversity within and among populations
The 10 isozyme systems analysed allowed us to interpret 18 putative loci, four of which (G6pdh, Mdh-2, Mdh-3 and Pgi-1) were monomorphic throughout, while all the remaining loci were polymorphic. Four of the total 43 alleles were exclusive to P. dactylifera populations (Mdh1-c, Mdh1-e, Pgm2-a and Pgm2-e). One exclusive locus (Mdh-4) was detected in the eastern Mediterranean species P. theophrasti. Furthermore, populations of P. canariensis and P. dactylifera had diverged conspicuously in terms of allele frequencies at several loci (eg Idh2-a, Mdh1-b and Pgm2-c). The frequencies of these alleles ranged between 0.013 and 0.319 in the mixed populations, while they reached values of 1.000 in P. dactylifera. There were no alleles shared exclusively by P. canariensis and the mixed populations.
On an average, P. canariensis showed lower values of genetic variability than P. dactylifera, and mixed populations (Table 1). Overall, the level of genetic variability varied widely among populations. Thus, the number of alleles per locus ranged from 1.25 in La Culata (ANC, P. canariensis) to 2.17 in Maspalomas (MAS, mixed populations). The lowest percentage of polymorphic loci was detected in P. canariensis (P=18.8 in La Culata, ANC) and the highest in the mixed populations of Maspalomas (MAS) and Río Palma (RP), both with P=72.2 (Table 1).
Genetic diversity statistics (Table 2) indicated that most of the genetic variability in P. canariensis, P. dactylifera and the mixed populations was contained within the populations, (Hs=75.12%, Hs=79.48% and Hs=85.59%, respectively).
Population genetic structure
Fixation indices (FIS) varied widely, and none of the populations surveyed was found to fit Hardy–Weinberg equilibrium proportions (Table 1). All P. canariensis populations (except for Acusa and La Sorrueda) showed heterozygosity excess, while all mixed populations (except for Barranco Angostura) exhibited a slight heterozygosity deficit (Table 1).
Genetic relationships within and among species
PCA, the first two eigenvectors of which accounted for 81.64% of the total variance (Figure 2), separated the Canary palm populations (AC, LS, RC, ANC, TAM, HY, VP, MIR and ZU) from the date palm populations (HM, GT, PB and EL). In the multivariate space defined by PCA, the mixed populations are much closer to P. canariensis, whereas the population of P. theophrasti is next to those of P. dactylifera. The values of genetic differentiation coefficient (FST) between pairs of P. canariensis populations (Table 3) ranged from FST=0.003, between Vegaipala (VP) and Las Hayas (HY), to FST=0.652, between VP and ANC. VP and HY, both from La Gomera, showed substantial genetic differences with respect to the remaining P. canariensis populations. In fact, the genetic differentiation among Canarian date palm populations decreased considerably (FST=0.170) when HY and (VP) populations were excluded from analysis (Figure 3). On the whole, genetic differentiation within P. canariensis was relatively high (FST=0.287) when compared to the values recorded for species sharing the same ecological and life history traits (Hamrick and Godt, 1990).
Mixed populations showed genetic differentiation values ranging from 0.012 to 0.379, exhibiting a moderate FST value (FST=0.152) among populations (Wright, 1978). As regards P. dactylifera, FST ranged from 0.129 to 0.391, showing a considerably higher genetic differentiation coefficient among populations (FST=0.252).
The FST values revealed a close genetic relationship between P. canariensis populations and the mixed populations (Table 3). The values of FST obtained by comparing P. dactylifera with P. canariensis (FST=0.442) or P. dactylifera with the mixed populations (FST=0.307) were higher than those corresponding to the comparison of the mixed populations with P. canariensis (FST=0.088; Figure 3).
Nei's (1972) genetic identities between P. canariensis population pairs (Table 3) ranged from I=0.847 to I=0.999 (average I=0.935). The identities between the Canarian date palm populations and the mixed populations were also high (average I=0.916). Genetic identities between pairs of P. dactylifera populations were slightly lower, spanning from I=0.806 to I=0.942 (average I=0.864) and indicating a moderate level of genetic differentiation among populations.
Discussion
Allozyme diversity within and among populations
A high degree of genetic variation was found in P. canariensis populations (A=1.59, P=41.8), similar to the values expected for monocotyledons (A=1.66, P=40.3) and for species with sexual reproduction (A=1.53, P=34.9), but higher than that for endemic species (A=1.39, P=26.3) (Hamrick and Godt, 1990). Average levels of expected heterozygosity, the more integrative measure, in P. canariensis (He=0.158), were higher than those found in most other species within the Arecaceae. Thus, although Eguiarte et al. (1992) found a similar value to P. canariensis' in the rain forest palm Astrocaryum mexicanum (He=0.153), lower values were described in Carpentaria acuminata, (He=0.143), Ptychosperma bleeseri (He=0.006) and Pinanga tenella (He=0.133) (Shapcott, 1998a, 1998b, 1999) and in Washingtonia filifera (He=0.008) (McClenaghand and Beauchamp, 1986). This result agrees with other studies that show higher genetic diversity levels in Canarian endemic plants than in those distributed in other oceanic islands (Francisco-Ortega et al, 2000; Batista et al, 2001; Batista and Sosa, 2002; Bouza et al, 2002). On the whole, the three groups of populations exhibited higher genetic variability values than species with similar life history traits (monocotyledons, endemics and species with sexual reproduction). However, genetic variation levels detected in the P. dactylifera populations analysed was lower than described by other authors (eg, Bennaceur et al, 1991) in date palm cultivars.
Earlier workers predicted that hybrid taxa would be more variable genetically and have greater evolutionary potential than their parental species because they would combine the alleles of both parents (Rieseberg, 1997). In our case, mixed populations exhibited higher levels of genetic diversity than P. canariensis and slightly higher than P. dactylifera populations (Table 1). The number of individuals of each Phoenix species notwithstanding, there are other factors that might be influencing diversity levels in the mixed populations. These might include the degree of genetic differentiation between both species, the breeding system, the possible hybridisation between P. canariensis and P. dactylifera, and historical contingency.
Most genetic variation in P. canariensis is maintained within populations (%Hs=75.12), as expected for predominantly outcrossing species (Hamrick and Godt, 1990). A similar percentage of genetic variation was maintained within populations in P. dactylifera (%Hs=79.48). The probable reason for the high intrapopulational gene diversity exhibited by the mixed populations (%Hs = 85.59) is the heterogeneous genetic makeups that coexist in these disturbed populations.
Population genetic structure
On the whole, none of the P. canariensis populations analysed was found to conform to Hardy–Weinberg proportions. Except for Acusa and La Sorrueda, all the Canarian date palm populations showed a heterozygote excess (Table 1). A heterozygosity excess can be the possible result of random stochastic events or the consequence of balancing selection promoting high heterozygosity (Linhart et al, 1981; Waser, 1987; Eguiarte et al, 1992). Although we do not have conclusive data to discard either possibility, it is highly improbable that most P. canariensis populations have an excess of heterozygotes by drift alone.
Populations from Acusa and La Sorrueda showed a heterozygosity deficit. This was not significant in Acusa, where deviations from Hardy–Weinberg equilibrium might be due mainly to stochastic factors associated with its small size (N=25). In La Sorrueda, the distribution of specimens in several clumps considerably separated in space (100–300 m) argues for the incidence of the Wahlund effect as the more likely explanation of the heterozygosity deficit observed, whereby heterozygosity reduces if the clumps sampled have diverged sufficiently (Elseth and Baumgardner, 1981).
Contrary to the general pattern for P. canariensis, all mixed populations showed a heterozygosity deficit save for Barranco Angostura, which exhibited a slight heterozygosity excess. Small heterozygosity deficit in outbreeding species are often a consequence of biparental inbreeding, especially in small populations or in those exhibiting spatial genetic structure (Sampson et al, 1988).
Owing to the cultivated condition of the P. dactylifera populations, it is not surprising that they do not conform to Hardy–Weinberg equilibrium proportions.
Genetic relationships within among species
The high level of allozyme similarity found among P. canariensis and P. dactylifera populations fits the values expected for closely related species (Gottlieb, 1981). Considering the extensive distribution of P. dactylifera in the Mediterranean area, it is feasible that P. canariensis is recently derived from a common ancestor closely related to P. dactylifera. The hypothesis of a recent divergence from P. dactylifera gains additional support from the fact that P. canariensis has less allozyme variation than P. dactylifera, its allelic makeup is a subset of that found in P. dactylifera, with no unique alleles, and there is a high genetic identity between both taxa; all of these features are characteristic of recently derived species (Purdy and Bayer, 1996). Consistent with the values of isozyme variation in P. canariensis, recent speciation from a widespread progenitor has been suggested as one of the causes of high genetic variation in endemic plant species (Loveless and Hamrick, 1988; Pleasants and Wendel, 1989). Thus, the isozyme data bolster Morici's (1998) morphological argument of a close phylogenetic relationship of P. canariensis with P. dactylifera. However, the high levels of genetic variation detected in P. canariensis populations indicate that more than one colonisation event could have occurred, as suggested by Francisco-Ortega et al (2000) for different Canarian endemics.
In close agreement with previous evidence based on morphology (Barrow, 1998), P. theophrasti is closer to P. dactylifera than to P. canariensis in the multivariate representation (Figure 2).
Unfortunately, we could not obtain specific and unambiguous isozyme molecular markers to differentiate P. canariensis and P. dactylifera individuals. Only four exclusive alleles were detected in P. dactylifera (Mdh1-c, Mdh1-e, Pgm2-c and Pgm2-e). However, given that none of these was taxon specific or even monomorphic, our expectations of having a marker of taxonomic circumscription at this molecular level of analysis are not met. The lack of an isozyme marker further supports a recent speciation of P. canariensis from an ancestor similar to P. dactylifera. Of the 43 alleles, 39 were common to both Phoenix species analysed, and this furnishes evidence of their shared evolutionary history. In a recent survey, the sharing of 14 of the 25 total alleles detected in five Canarian taxa of Cistus (Batista et al, 2001) was used to support the close phylogenetic relationship among them. Similarly, a close relatedness between two Brighamia species from Hawaii was inferred from their sharing of only six alleles out of 22 (Gemmill et al, 1998).
The frequency of the alleles shared exclusively by P. dactylifera and the mixed populations can help us estimate the degree with which date palm individuals were introduced in the latter (Gallagher et al, 1997). In all cases, these shared alleles are poorly represented in the mixed populations (with frequencies from 0.000 to 0.319), thereby suggesting that a relatively low number of P. dactylifera specimens were introduced. This argument is cogent with FST values, which imply a much closer genetic relationship between P. canariensis and the mixed populations than between either of these and P. dactylifera (Figure 3).
Given that we did not detect different monomorphic alleles in P. canariensis and P. dactylifera that were present in heterozygosity in individuals with intermediate morphological traits, this allozyme survey cannot substantiate the existence of putative hybrid individuals in nature. Although molecular markers represent a powerful tool for identifying hybrid taxa, even this approach can generate ambiguous results (Rieseberg, 1997). A taxon can share molecular markers with related taxa due to the joint retention of alleles following speciation from a polymorphic ancestor (symplesiomorphy). This phenomenon has also been referred to as lineage sorting when discussed in the context of gene lineage data (Avise, 1994; Rieseberg, 1997). Besides, the likelihood of finding exclusive molecular markers decreases as the time since divergence reduces. Consequently, it is usually easier to reject the hypothesis of hybrid origin than to confirm it with molecular data sets.
The PCA (Figure 2) separated P. dactylifera and P. canariensis clearly, and showed a closer relationship between the latter species and the mixed populations. In general, the percentage of P. canariensiś individuals in the mixed populations outnumbers that of P. dactyliferás, which probably contributes further the closer genetic relationship between P. canariensis and the mixed populations.
Since hybrids are a mosaic of parental and intermediate characters (Rieseberg and Ellstrand, 1993), it is possible that some of the individuals that we characterised as morphologically intermediate are in fact different ecotypes of P. canariensis. Also, morphologically intermediate individuals may be hybrid progeny corresponding to F2 or later generations that have lost P. dactylifera alleles by backcrossing to pure P. canariensis. As most gene flow occurs between the hybrid and a single parent (P. canariensis), the segregating generations will be mostly advanced generation backcrosses and have multilocus associations typical of the most compatible parent (P. canariensis), as suggested earlier (Rieseberg et al, 1989; Arnold et al, 1991; Nason et al, 1992; Rieseberg and Ellstrand, 1993). In addition, selection against recombinants should be intense in hybrid zones; therefore, the surviving individuals would be those that retained the ecological traits of one parent, such as backcrosses (Anderson, 1998). As our data do not offer an insight on whether hybridisation between P. canariensis and P. dactylifera occurs, we cannot discard any of the two explanations.
Populations of P. dactylifera have not been described in La Gomera (Izquierdo et al, 2001) and, therefore, a hybrid origin of the highly differentiated VP and HY seems improbable. Hence, until additional studies of these populations are carried out, it seems safer to construe their high genetic differentiation mainly as the result of genetic drift in combination with a long history of isolation.
The higher degree of genetic differentiation detected within the Canarian range of P. dactylifera (FST=0.252) probably reflects low levels of gene flow in combination with different geographical origins of the introduced date palms.
Conservation implications
One of the purposes of this work was to use the information regarding the degree and distribution of genetic variability in P. canariensis for the implementation of conservation strategies. Fortunately, the high levels of genetic diversity present in P. canariensis are encouraging for conservation efforts because they should help buffer the effects of selection and potential inbreeding in the populations (Travis et al, 1996). As most of the high genetic variability in P. canariensis is maintained within populations, avoiding fragmentation to prevent genetic variability loss through a cessation of interpopulation genetic interchange must be a crucial commitment for ‘in situ’ conservation strategies. As it is not possible to design a reserve that includes all the populations of P. canariensis in any given island of occurrence, establishing multiple small ecological reserves for this species should be most effective at buffering inbreeding and genetic drift (Hawkes et al, 1997), especially if these reserves are managed in a coordinated way to facilitate gene flow. Ideally, these reserves should include the more polymorphic populations of P. canariensis, as this strategy will enhance the potential of surviving environmental change (Lande and Schemske, 1985; Charlesworth and Charlesworth, 1987). According to this criterion, populations Rambla de Castro (RC; Ho=0.249, He=0.244) and La Sorrueda (LS; Ho=0.187, He=0.216) would be the best-suited management targets in Tenerife and Gran Canaria, respectively.
Genetic conservation strategies based on seed and germplasm collection and preservation in gene banks are necessary for the ex situ conservation of any endangered plant. The population at RC is the best candidate for sampling for this purpose, as it shows the higher values of the basic indicators of allozyme variation.
At this stage, transplanting between two different populations must be strongly discouraged for two important reasons. First, we can neither ascribe the morphologically intermediate individuals unambiguously to P. canariensis or P. dactylifera, nor differentiate between juvenile individuals of either species. And second, the mixing of genetically distinct populations of P. canariensis may pose the risk of outbreeding depression, whereby a reduction in fitness arises due to a loss of local adaptations or the break-up of coadapted gene complexes (Storfer, 1999).
Although outbreeding depression commonly manifests through the decrease in the value of fitness-related traits such as fruit production, survivorship or seed germination, it often results in a heterozygosity loss in the outbred populations (Fenster and Galloway, 2000). At odds with this prediction, the mixed populations (where outbreeding could have proceeded for several generations) display much higher average heterozygosity values (He=0.254, Ho=0.239) than P. canariensis (He=0.158, Ho=0.179). Hence, our isozyme data do not provide evidence that historical transplanting of P. dactylifera specimens into P. canariensis' populations compromised population survival. However, we must bear in mind that the effects of outbreeding depression might take a long time to manifest and may even be preceded by several generations of heterosis. Remarkably, Fenster and Galloway (2000) detect a significant heterozygosity loss and a decrease in five fitness-related traits in Chamaecrista fasciculata (Fabaceae) after only three generations of outbred crosses, where the first generation far outperformed either parental line. Thus, having adequate demographic data for the mixed Phoenix populations would be crucial to detecting outbreeding depression.
Hybridisation has recently been shown to have both beneficial and harmful consequences for the conservation of plant diversity, leading to increased diversity in some instances and to possible extinction of populations or species by genetic swamping in other. This occurs when a locally rare species loses its genetic integrity and becomes assimilated into a locally common species as a result of repeated events of hybridisation and introgression (Rieseberg, 1991; Ellstrand, 1992; Ellstrand et al, 1999).
One of the aims of P. canariensis conservation should be the search for a molecular marker able to discriminate unambiguously P. canariensis and P. dactylifera individuals from their putative hybrids. Since we have not been able to find these molecular markers through isozyme electrophoresis, and bearing in mind that the mutation rate of isozyme markers is usually lower than that of DNA markers (Li, 1997; Yan et al, 1999), DNA markers will be the best candidates for differentiating among individuals belonging to both species.
References
Allphin L, Windham MT, Harper KT (1998). Genetic diversity and gene flow in the endangered dwarf bear poppy, Arctomecon humilis (Papaveraceae). Am J Bot 85: 1251–1261.
Anderson E (1998). Hybridization of the habitat. Evolution 2: 1–9.
Arnold Ml, Bucker CM, Robinson JJ (1991). Pollen mediated introgression and hybrid speciation in Louisiana irises. Proc Natl Acad USA 88: 1398–1402.
Avise JC (1994). Molecular Markers, Natural History and Evolution. Chapman & Hall: New York.
Barrow SC (1998). A monograph of Phoenix L. (Palmae: Coryphoideae). Kew Bull 53: 513–575.
Batista F, Bañares A, Caujapé-Castells J, Carque E, Marrero-Gómez M, Sosa PA (2001). Allozyme diversity in three endemic species of Cistus (Cistaceae) from the Canary Islands: intraspecific comparisons and implications for genetic conservation. Am J Bot 88: 1582–1592.
Batista F, Sosa PA (2002). Allozyme diversity in natural populations of Viola palmensis Webb & Berth. (Violaceae) from La Palma (Canary Islands): implications for conservation genetics. Ann Bot 90: 725–733.
Bendiab K, Baaziz M, Majourhat K (1998). Preliminary date palm cultivar composition of Moroccan palm groves as revealed by leaf isoezymes phenotypes. Bioch Syst Ecol 26: 71–82.
Bennaceur M, Lanaud C, Chevallier MH, Bounaga N (1991). Genetic diversity of the date palm (Phoenix dactylifera L.) from Algeria revealed by enzyme markers. Plant Breed 107: 56–69.
BOC (1991). Orden de 20 Febrero: protección de especies de la flora vascular silvestre de las Islas Canarias. Bol Canarias 35: 1324–1334.
Booij I, Monfort S, Ferry M (1995). Characterization of thirteen date palm (Phoenix dactylifera) cultivars by enzymes electrophoresis using the PhastSystem. J Plant Physiol 145: 62–66.
Bouza N, Caujapé-Castells J, González-Pérez MA, Batista F, Sosa PA (2002). Population structure and genetic diversity of two endangered endemic species of the Canarian Laurel Forest: Dorycnium spectabile (Fabaceae) and Isoplexis chalcantha (Scrophulariaceae). Int J Plant Sci 163: 619–630.
Caujapé-Castells J, Pedrola-Monfort J, Membrives N (1999). Contrasting patterns of genetic structure in the South African species Androcymbium bellum, A. guttatum, and A. pulchrum (Colchicaceae). Bioch Syst Ecol 27: 591–605.
Chabaud B (1882). Le Phoenix canariensis. Provence Agric Hortic Ilustr 19: 293–297.
Charlesworth D, Charlesworth B (1987). Inbreeding depression and its evolutionary consequences. Ann Rev Ecol Syst 18: 237–268.
Clayton JW, Tretiak DN (1972). Amine citrate buffer for pH control in starch gel electrophoresis. J Fish Res Board Can 29: 1169–1172.
Eguiarte LE, Perez-Nasser N, Piñero D (1992). Genetic structure, outcrossing rate and heterosis in Astrocaryum mexicanum (tropical palm): implications for evolution and conservation. Heredity 69: 217–228.
Elisiário PJ, Justo EM, Leitao JM (1999). Identification of mandarin hybrids by isozyme and RAPD analysis. Sci Hortic 81: 287–299.
Ellstrand NC (1992). Gene flow by pollen: implications for plant conservation. Oikos 63: 77–86.
Ellstrand NC, Prentice HC, Hancock JF (1999). Gene flow and introgression from domesticated plants into their wild relatives. Ann Rev Ecol Syst 30: 539–563.
Elseth GD, Baumgardner KD (1981). Population Biology. Van Nostrand: New York.
Fenster CB, Galloway LF (2000). Population differentiation in an annual legume: genetic architecture. Evolution 54: 1157–1172.
Francisco-Ortega F, Santos-Guerra A, Kim SC, Crawford DJ (2000). Plant genetic diversity in the Canary Islands: a conservation perspective. Am J Bot 87: 909–919.
Gallagher KG, Schierenbeck KA, D'Antonio CM (1997). Hybridization and introgression in Carpobrotus spp. (Aizoaceae) in California. II. Allozyme evidence. Am J Bot 84: 905–911.
Gemmill CEC, Ranker TA, Ragone D, Perlman SP, Wood KR (1998). Conservation genetics of the endangered endemic Hawaiian genus Brighamia (Campanulaceae). Am J Bot 85: 528–539.
Godt MJW, Hamrick JL (1998). Allozyme diversity in the endangered pitcher plant Sarracenia rubra ssp. alabamensis (Sarraceniaceae) and its close relative S. rubra ssp. rubra. Am J Bot 85: 802–810.
González-Pérez, MA (2001). Caracterización molecular de la palmera canaria (Phoenix canariensis) como base para su conservación (PhD. Thesis). Las Palmas de Gran Canaria. Canary Islands. University of Las Palmas de Gran Canaria: Spain.
Gottlieb LD (1981). Electrophoretic evidence and plant populations. Progr Phytochem 7: 1–46.
Guo SW, Thompson EA (1992). Performing the exact test of Hardy–Weinberg proportions for multiple alleles. Biometrics 48: 361–372.
Hamrick JL, Godt MJW (1990). Allozyme diversity in plant species. Brown HD, Clegg MT, Kahler AL, Weir BS (eds) Plant Populations Genetics, Breeding and Genetic Resources. Sinauer: Sunderland, MA.
Hawkes JG, Maxted N, Zohary D (1997). Reserve design. lang2057In: Maxted N, Ford-Lloyd BV, Hawkes JG (eds) Plant Genetic Conservation. Chapman and Hall: London, pp 132–143.
Hodel DR (1995). Phoenix: the date palms. Palm J 122: 14–36.
Izquierdo I, Martín JL, Zurita N, Arechavaleta M (2001). Lista de especies silvestres de Canarias (hongos, plantas y animales terrestres). Consejería de Política Territorial y Medio Ambiente del Gobierno de Canarias.
Kunkel G, Kunkel MA (1974). Flora de Gran Canaria. Arboles y arbustos arboreos. Las Palmas de Gran Canaria: Excmo. Cabildo Insular de Gran Canaria, Vol, 1: 4–5.
Lande R, Schemske DW (1985). The evolution of self-fertilization and inbreeding depression in plants. I. Genetic models. Evolution 29: 24–40.
Lewis PO (1993). GeneStat-PC 3.31. Raleigh. North Carolina State University: North Carolina.
Li WH (1997). Molecular Evolution. Sinauer Publishers: Sunderland, MA.
Linhart YB, Mitton JB, Sturgeon KB, Davis ML (1981). Genetic variation in space and time in a population of ponderosa pine. Heredity 46: 407–426.
Loveless MD, Hamrick JL (1988). Genetic organization and evolutionary history in two North American species of Cirsium. Evolution 42: 254–265.
McClenaghand LR, Beauchamp AC (1986). Low genetic differentiation among isolated populations of the California fan palm (Washingtonia filifera). Evolution 40: 315–322.
Morici C (1998). Phoenix in the wild. Principes 42: 85–89.
Nason JD, Ellstrand NC, Arnold Ml (1992). Patterns of hybridization and introgression in populations of oaks, manzanitas, and irises. Am J Bot 79: 101–111.
Nei M (1972). Genetic distance between populations. Am Nat 106: 283–292.
Nei M (1973). Analysis of gene diversity in subdivided populations. Proc Natl Acad Sci USA 70: 3321–3323.
Pleasants JM, Wendel JF (1989). Genetic diversity in a clonal narrow endemic, Erythronium propullens, and its progenitor, Erythronium albidum. Am J Bot 76: 1136–1151.
Purdy BG, Bayer RJ (1996). Genetic variation in populations of the endemic Achillea millefolium ssp. megacephala from the Athabasca sand dunes and the widespread ssp. lanulosa in western North America. Can J Bot 74: 1138–1146.
Raymond M, Rousset F (1997). GENEPOP. Version 3.1. Laboratorie de Génétique et Environnement. Montpellier. Université de Montpellier II.: France.
Rieseberg LH (1991). Homoploid reticulate evolution in Helianthus (Asteraceae): evidence from ribosomal genes. Am J Bot 78: 1218–1237.
Rieseberg LH (1997). Hybrid origins of plant species. Ann Rev Ecol Syst 28: 359–389.
Rieseberg LH, Ellstrand NC (1993). What can molecular and morphological markers tell us about plant hybridization? Crit Rev Plant Sci 12: 213–241.
Rieseberg LH, Zona S, Aberbom L, Martin TD (1989). Hybridization in the island endemic, Catalina Mahogany. Conserv Biol 3: 52–58.
Sampson JF, Hopper SD, Jones SH (1988). Genetic diversity and the conservation of Eucalyptus crucis Maiden. Aust J Bot 36: 447–460.
Santana SA, Toledo RJM (1997). Introducción y dispersión de Phoenix dactylifera en el Archipiélago Canario: elementos de discusión. Second International Symposium on Ornamental Palms and other Monocts from the Tropics. Canary Islands, Spain.
Schnabel A, Laushman RH, Hamrick JL (1991). Comparative genetic structure of two co-occurring tree species: Maclura pomifera (Moraceae) and Gleditsia triacanthos (Leguminoseae). Heredity 67: 357–364.
Shapcott A (1995). The spatial genetic structure in natural populations of the Australian temperate rainforest tree Atherosperma moscatum (Labill.) (Monimiaceae). Heredity 74: 28–38.
Shapcott A (1998a). The patterns of genetic diversity in Carpentaria acuminata (Arecaceae), and rainforest history in northern Australia. Mol Ecol 7: 833–847.
Shapcott A (1998b). The genetics of Ptychosperma bleeseri a rare palm from the Northern Territory, Australia. Biol Conserv 85: 203–209.
Shapcott A (1999). Comparison of the population genetics and densities of five Pinanga palm species at Kuala Belalong, Brunei. Mol Ecol 8: 1641–1654.
Sosa PA, Bouza N, Cabrera-Pérez MA, Luque A (1998). Genetic variation in Phoenix canariensis and P. dactylifera (Arecaceae) populations of Gran Canaria using isoezyme electroforesis. Bol Mus Mun Funchal 5: 443.
Storfer A (1999). Geneflow and endangered species translocations: a topic revisited. Biol Conserv 87: 173–180.
Stuber CW, Goodman MM, Johnson FM (1977). Genetic control and racial variation of β-glucosidase isozymes in maize (Zea mays L.). Biochem Genet 15: 383–394.
Swofford DL, Selander RB (1981). BIOSYS-1: a Computer Program for the Analysis of Allelic Variation in Genetics. Realese 1. Useŕs manual. University of Illinois: Urbana, Ill.
Torres AM, Tisserat B (1980). Leaf isozymes as genetic markers in date palms. Am J Bot 67: 162–167.
Travis SE, Maschinski J, Keim P (1996). An analysis of genetic variation in Astragalus cremnophylax var. cremnophylax, a critically endangered plant, using AFLP markers. Mol Ecol 5: 735–745.
Waser NM (1987). Spatial genetic heterogeneity in a population of the montane perennial plant Delphinium nelsonii. Heredity 58: 249–256.
Weir BS (1990). Genetic Data Analysis. Sinauer Publishers: Sunderland, MA.
Wendel F, Weeden F (1989). Visualization and interpretation of plant isozymes. In: Soltis D, Soltis P (eds) Isozyme in Plant Biology. Dioscorides Press: Portland.
Wright S (1978). Evolution and the Genetics of Populations, Vol 4: Variability within and among Natural Populations. University of Chicago Press: Chicago.
Yan G, Romero-Severson J, Walton M, Chadee DD, Severson DW (1999). Population genetics of the yellow fever mosquito in Trinidad: comparisons of AFLP and RFLP markers. Mol Ecol 8: 951–964.
Acknowledgements
We thank the Gobierno de Canarias for financial support through its ‘Programa de Becas de postgrado para la realización de tesis doctorales’. We thank Dr Michel Ferry for providing samples of Phoenix dactylifera from the Hort del Gat, Research Station on Date Palm and Arid Land Farming Systems in Elche (Spain). We also thank Joan Pedrola-Monfort for kindly sending leaves of Phoenix theophrasti. We are grateful to all the people who provided helpful field assistance. This paper is dedicated to Jaime O'Shanahan for his great interest in this work and his defense of the Canarian date palm. This work was funded by Viceconsejeria de Educación, Gobierno de Canarias Ref. 93/163 and 94/2614. We thank A Stephens for English corrections.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
González-Pérez, M., Caujapé-Castells, J. & Sosa, P. Allozyme variation and structure of the Canarian endemic palm tree Phoenix canariensis (Arecaceae): implications for conservation. Heredity 93, 307–315 (2004). https://doi.org/10.1038/sj.hdy.6800507
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.hdy.6800507