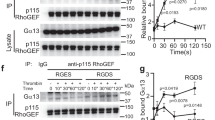
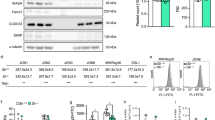
Abstract
Integrins not only bind adhesive ligands1, they also act as signalling receptors2. Both functions allow the integrin αIIbβ3 to mediate platelet aggregation3. Platelet agonists activate αIIbβ3 (inside-out signalling) to allow the binding of soluble fibrinogen. Subsequent platelet aggregation leads to outside-in αIIbβ3 signalling, which results in calcium mobilization4, tyrosine phosphorylation of numerous proteins5,6 including β3 itself7, increased cytoskeletal reorganisation8 and further activation of αIIbβ3 (ref. 2). Thus, outside-in signals enhance aggregation, although the mechanisms and functional consequences of specific signalling events remain unclear. Here we describe a mouse that expresses an αIIbβ3 in which the tyrosines in the integrin cytoplasmic tyrosine motif have been mutated to phenylalanines. These mice are selectively impaired in outside-in αIIbβ3 signalling, with defective aggregation and clot-retraction responses in vitro, and an in vivo bleeding defect which is characterized by a pronounced tendency to rebleed. These data provide evidence for an important role of outside-in signalling in platelet physiology. Furthermore, they identify the integrin cytoplasmic tyrosine motif as a key mediator of β-integrin signals and a potential target for new therapeutic agents.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Hynes,R. O. Integrins: versatility, modulation, and signaling in cell adhesion. Cell 69, 11–25 (1992).
Shattil,S., Kashiwagi, H. & Pampori,N. Integrin signaling: the platelet paradigm. Blood 91, 1–14 (1998).
Phillips,D. R., Charo,I. F. & Scarborough,R. M. GPIIb-IIIa: the responsive integrin. Cell 65, 359–362 (1991).
Yamaguchi,K. et al. Flow cytometric analysis of changes in cytoskeletal proteins during platelet destruction and activation using a monoclonal antibody against platelet myosin. Am. J. Hematol. 44, 106–111 (1993).
Clark,E. A., Shattil,S. J., Ginsberg,M. H., Bolen,J. & Brugge,J. S. Regulation of the protein tyrosine kinase pp72syk by platelet agonists and the integrin αIIbβ3. J. Biol. Chem. 269, 28859–28864 (1994).
Lipfert,L. et al. Integrin-dependent phosphorylation and activation of the protein tyrosine kinase pp125FAK in platelets. J. Cell. Biol. 119, 905–912 (1992).
Law,D. A., Nannizzi-Alaimo,L. & Phillips,D. R. Outside-in integrin signal transduction. αIIbβ3-(GP IIb IIIa) tyrosine phosphorylation induced by platelet aggregation. J. Biol. Chem. 271, 10811–10815 (1996).
Phillips,D. R., Jennings,L. K. & Edwards,H. H. Identification of membrane proteins mediating the interaction of human platelets. J. Cell. Biol. 86, 77–86 (1980).
van der Geer,P. et al. A conserved amino-terminal Shc domain binds to phosphotyrosine motifs in activated receptors and phosphopeptides. Curr. Biol. 5, 404–412 (1995).
Jenkins,A. et al. Tyrosine phosphorylation of the β3 cytoplasmic domain mediates integrin-cytoskeletal interactions. J. Biol. Chem. 273, 13878–13885 (1998).
Guinto,E. R. et al. Unexpected crucial role of residue 225 in serine proteases. Proc. Natl Acad. Sci. USA 96, 1852–1857 (1999).
Filardo,E. J., Brooks,P. C., Deming,S. L., Damsky,C. & Cheresh,D. A. Requirement of the NPXY motif in the integrin β3 subunit cytoplasmic tail for melanoma cell migration in vitro and in vivo. J. Cell. Biol. 30, 441–450 (1995).
Romzek,N. C. et al. Use of a β1 integrin-deficient human T cell to identify β1 integrin cytoplasmic domain sequences critical for integrin function. Mol. Biol. Cell 9, 2715–2777 (1998).
Ylanne,J. et al. Distinct functions of integrin α and β subunit cytoplasmic domains in cell spreading and formation of focal adhesions. J. Cell. Biol. 122, 223–233 (1993).
Blystone,S. D., Williams,M. P., Slater,S. E. & Brown,E. J. Requirement of integrin β3 tyrosine 747 for β3 tyrosine phosphorylation and regulation of αvβ3 avidity. J. Biol. Chem. 272, 28757–28761 (1997).
Fox,J. E. et al. On the role of the platelet membrane skeleton in mediating signal transduction. Association of GP IIb-IIIa, pp60c-src, pp62c-yes, and the p21ras GTPase-activating protein with the membrane skeleton. J. Biol. Chem. 268, 25973–25984 (1993).
Hodivala-Dilke,K. M. et al. β3-integrin-deficient mice are a model for Glanzmann thrombasthenia showing placental defects and reduced survival. J. Clin. Invest. 103, 229–238 (1999).
Coller,B. S. Platelets and thrombolytic therapy. N. Engl. J. Med. 322, 33–42 (1990).
Chen,Y. P. et al. Ser-752→Pro mutation in the cytoplasmic domain of integrin β3 subunit and defective activation of platelet integrin αIIb β3 (glycoprotein IIb-IIIa) in a variant of Glanzmann thrombasthenia. Proc.f Natl Acad. Sci. USA 89, 10169–10173 (1992).
Chen,Y. P., O'Toole,T. E., Ylanne,J., Rosa,J. P. & Ginsberg,M. H. A point mutation in the integrin β3 cytoplasmic domain (S752→P) impairs bidirectional signaling through αIIbβ3 (platelet glycoprotein IIb–IIIa). Blood 84, 1857–1865 (1994).
Williams,M. J., Hughes,P. E., O'Toole,T. E. & Ginsberg,M. H. The inner world of cell adhesion:integrin cytoplasmic domains. Trends Cell Biol. 4, 109–112 (1994).
Fitzgerald,L. A., Steiner,B., Rall,S. C. Jr, Lo,S. S. & Phillips,D. R. Protein sequence of endothelial glycoprotein IIIa derived from a cDNA clone. Identity with platelet glycoprotein IIIa and similarity to “integrin”. J. Biol. Chem. 262, 3936–3939 (1987).
Dymecki,S. M. Flp recombinase promotes site-specific DNA recombination in embryonic stem cells and transgenic mice. Proc. Natl Acad. Sci. USA 93, 6191–6196 (1996).
Law,D. A. et al. Genetic and pharmacologic analyses of Syk function in αIIbβ3 signaling in platelets. Blood 93, 2645–2652 (1999).
Acknowledgements
We thank R. Wong for his help with the figures.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Law, D., DeGuzman, F., Heiser, P. et al. Integrin cytoplasmic tyrosine motif is required for outside-in αIIbβ3 signalling and platelet function. Nature 401, 808–811 (1999). https://doi.org/10.1038/44599
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/44599
This article is cited by
-
Mechanisms of glabridin inhibition of integrin αIIbβ3 inside-out signals and NF-κB activation in human platelets
Chinese Medicine (2023)
-
Structure, signal transduction, activation, and inhibition of integrin αIIbβ3
Thrombosis Journal (2023)
-
Integrin αIIbβ3 outside-in signaling activates human platelets through serine 24 phosphorylation of Disabled-2
Cell & Bioscience (2021)
-
Modulation of human platelet activation and in vivo vascular thrombosis by columbianadin: regulation by integrin αIIbβ3 inside-out but not outside-in signals
Journal of Biomedical Science (2020)
-
Platelet integrin αIIbβ3: signal transduction, regulation, and its therapeutic targeting
Journal of Hematology & Oncology (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.