Abstract
Intestinal metaplasia has been proposed to be a precursor lesion of adenocarcinoma in the urinary bladder. CDX2 is a transcription factor that is encoded by a homeotype gene that plays an essential role in the differentiation and proliferation of intestinal epithelial cells. Hepatocyte-specific antigen (Hep) has also been shown to be a useful marker of intestinal metaplasia. Tissues from 46 patients, including 22 cases of intestinal metaplasia of the urinary bladder, 11 cases of typical cystitis glandularis, and 13 cases containing both lesions, were selected and immunohistochemical stains for CDX2, Hep, cytokeratin 20 (CK20), and cytokeratin 7 (CK7) were performed. Nuclear staining for CDX2 was observed in 29 of 35 (83%) cases of intestinal metaplasia of the urinary bladder. In contrast, nuclear staining for CDX2 was not observed in any case of typical cystitis glandularis; however, seven of 24 (29%) cases showed aberrant cytoplasmic expression in a mean of 37% of cells. CK20 was expressed in 28 of 35 (80%) cases of intestinal metaplasia, but was observed in only one of 24 (4%) cases of cystitis glandularis in 15% of cells. CK7 was expressed in only six of 35 (17%) cases of intestinal metaplasia, whereas expression of CK7 was observed in all cases (100%) of typical cystitis glandularis with a mean percentage of positively staining cells of 63%. The mean percentages of positively staining cells in intestinal metaplasia with CDX2, CK20, and CK7 were 55, 49, and 53%, respectively. All examples of both intestinal metaplasia and typical cystitis glandularis were uniformly negative for Hep. In the urinary bladder, intestinal metaplasia and typical cystitis glandularis have sharply contrasting immunoprofiles. Additionally, the absence of Hep staining in intestinal metaplasia of the urinary bladder, despite its morphologic resemblance to normal colonic mucosa and intestinal metaplasia in other organs, may signify the presence of unique metaplastic pathways in the urinary bladder.
Similar content being viewed by others
Main
Cystitis glandularis is a metaplastic alteration of the urothelium in the urinary bladder that is thought to be induced by chronic inflammation or irritation.1, 2 Two subtypes with distinct morphology and behavior exist. The typical type is characterized by luminal structures within the lamina propria having an innermost lining of columnar or cuboidal cells and bounded peripherally by transitional cells. The intestinal type, also referred to as intestinal metaplasia, has a similar glandular architecture in the lamina propria but contains abundant mucin-secreting goblet cells in the lining epithelium.3 These two subtypes may coexist but one or the other is usually the predominant or exclusive type in a given case.4 The exact incidences of the two subtypes are uncertain; however, intestinal metaplasia is much less common compared with the typical type of cystitis glandularis.5 Florid cystitis glandularis of intestinal type may at times resemble adenocarcinoma,4 and intestinal metaplasia is a risk factor and a putative precursor of adenocarcinoma.6, 7, 8, 9
The human CDX2 gene is a member of the caudal-type homeobox gene family, an analog of the caudal gene in Drosophila melanogaster. The gene product, CDX2 protein, was initially identified in epithelial cells throughout the small and large intestines, and is believed to play a critical role in the differentiation and maintenance of intestinal epithelium via the regulation of intestine-specific gene transcription.10, 11, 12, 13, 14 Subsequent studies have demonstrated its expression in adenocarcinomas in the esophagus, stomach, ovary, uterus, and lung,15, 16, 17, 18, 19, 20 as well as in intestinal metaplasia in the upper gastrointestinal tract and the gall bladder.18, 21, 22, 23, 24 Hepatocyte-specific antigen (Hep) is a cytoplasmic antigen initially identified in normal hepatocytes and in hepatic neoplasia.25, 26, 27 It has also been shown to be a useful marker of intestinal metaplasia in the esophagus and stomach.28, 29 The aim of this study is to evaluate and compare the expression of CDX2, Hep, cytokeratin 7 (CK7), and cytokeratin 20 (CK20) in intestinal metaplasia of the urinary bladder and in typical cystitis glandularis in order to determine whether these two entities demonstrate distinct immunophenotypes, and hence whether these two morphologically similar entities are derived via different pathogenetic mechanisms.
Materials and methods
Tissues from 46 patients, including 22 cases of intestinal metaplasia of the urinary bladder, 11 cases of typical cystitis glandularis, and 13 cases containing both lesions accessioned between 1989 and 2005, were obtained from the Surgical Pathology archives of the participating institutions. All cases were retrospectively reviewed and diagnosed according to well-accepted criteria.3, 4, 5, 30 The lesions characterized by luminal structures within the lamina propria having an innermost lining of columnar or cuboidal cells and peripheral transitional cells were classified as typical cystitis glandularis. Similar luminal structures present within the lamina propria but containing mucin-secreting goblet cells in their lining epithelium were considered diagnostic of intestinal metaplasia.
Thick sections (4 μm) were cut from formalin-fixed and paraffin-embedded tissue blocks and stained with hematoxylin and eosin. Additional sections from appropriately selected blocks were cut for immunohistochemistry, which was performed on an automated immunostainer. All slides were analyzed with the following monoclonal antibodies: CDX2 (clone CDX2-88, Biogenex, San Ramon, CA, USA, prediluted), Hep (clone OCH1E5, Cell Marque, Hot Springs, AR, USA, prediluted), CK7 (clone OV-TL 12/30, Dako, Carpinteria, CA, USA, prediluted), and CK20 (clone Ks20.8, Dako, Carpinteria, CA, USA, prediluted). The slides were deparaffinized twice in xylene for 5 min and rehydrated through graded ethanol solutions to distilled water. Antigen retrieval was performed by heating sections in citrate buffer (CDX2, Hep, and CK20) or enzymatically with proteinase K (CK7). Inactivation of endogenous peroxidase activity was obtained by incubating sections in 3% H2O2 for 10 min. Localization of bound antibodies was performed with a peroxidase-labeled streptavidin–biotin system (DAKO, LSAB2 Kit) with 3,3′-diaminobenzidine as a chromogen for CK7 and CK20. For CDX2 and Hep immunostains, a highly sensitive polymer-based detection system (DAKO, EnVision™+System) was utilized. Appropriate positive controls for each antibody were run concurrently and showed adequate immunostaining.
Cytoplasmic staining for CK7, CK20, and Hep, and nuclear and aberrant cytoplasmic staining for CDX2 were evaluated in a semiquantitative manner in intestinal metaplasia and typical-type cystitis glandularis. The percentages of positively staining cells were estimated and the staining intensity was classified as negative (0), weak (1), moderate (2), and strong (3), as previously described.31, 32, 33
Results
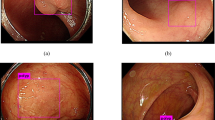
The results of the immunohistochemical staining are summarized in Tables 1 and 2. Positive nuclear staining for CDX2 was observed in 29 of 35 (83%) cases of intestinal metaplasia of the urinary bladder. One specimen showed only aberrant cytoplasmic staining. In sharp contrast, nuclear staining for CDX2 was not identified in any case of typical cystitis glandularis; however, seven of 24 (29%) cases showed aberrant cytoplasmic expression in a mean of 37% of cells. Three cases of intestinal metaplasia revealed nuclear CDX2 expression in the adjacent non-metaplastic urothelium and aberrant cytoplasmic staining of non-metaplastic urothelium was observed in four other cases. Three cases of typical cystitis glandularis displayed aberrant cytoplasmic CDX2 staining in the nonmetaplastic urothelium. CK20 was expressed in 28 of 35 (80%) cases of intestinal metaplasia, but was observed in only one of 24 (4%) cases of typical cystitis glandularis in 15% of cells. CK7 was expressed in only six of 35 (17%) cases of intestinal metaplasia, whereas expression of CK7 was identified in all 24 cases (100%) of typical cystitis glandularis with a mean percentage of positively staining cells of 63%. The mean percentage of positively staining cells in intestinal metaplasia with CDX2, CK20, and CK7 was 55, 49, and 53%, respectively. All examples of both intestinal metaplasia and typical cystitis glandularis were uniformly negative for Hep (Figure 1).
Immunohistochemical analysis of intestinal metaplasia of the urinary bladder (a–e) and typical cystitis glandularis (f–j). Intestinal metaplasia (a) demonstrates positive nuclear staining for CDX2 (b), positive cytoplasmic staining for CK20 (c), and negative staining for CK7 (d) and Hep (e). Typical cystitis glandularis (f) demonstrates negative staining for CDX2 (g), CK20 (h), and Hep (j), and positive cytoplasmic staining for CK7 (i).
Discussion
In this study, intestinal metaplasia of the urinary bladder and typical cystitis glandularis were found to have different and contrasting immunoprofiles. Nuclear staining for CDX2 is often present in intestinal metaplasia, but is not characteristic of typical cystitis glandularis. Expression of CK20 and absence of CK7 expression typifies the great majority of cases of intestinal metaplasia. However, typical cystitis glandularis is characterized by the opposite cytokeratin profile. Additionally, the absence of Hep staining in intestinal metaplasia of the urinary bladder, despite its morphologic resemblance to normal colonic mucosa and intestinal metaplasia in other organs, may signify the presence of a unique metaplastic pathway in the urinary bladder.
CDX2 is a transcription factor that is encoded by a homeotype gene and plays an essential role in the differentiation and proliferation of intestinal epithelial cells.9, 10, 12, 13, 14 Expression of CDX2 has been reported in adenocarcinomas of many sites, including the esophagus, stomach, intestine, ovary, uterus, and lung.15, 16, 17, 18, 19, 20 In the present study, nuclear expression of CDX2 was observed in the great majority of cases of intestinal metaplasia of the urinary bladder, which is not an unexpected finding considering the regulatory role of CDX2 in intestinal differentiation. In sharp contrast, no case of typical cystitis glandularis revealed similar nuclear staining.
Weak and aberrant cytoplasmic staining was noted in seven cases of typical cystitis glandularis and in seven specimens of nonmetaplastic urothelium. This aberrant immunostaining was often observed in a setting of stromal inflammation. Similar aberrant cytoplasmic CDX2 expression has also been reported in the esophagus and the gall bladder. Eda et al21 described fine, granular cytoplasmic staining of CDX2 in Barrett's epithelium and in chronically inflamed esophageal mucosa. Aberrant cytoplasmic expression of CDX2 in a background of inflammation was also observed in the gallbladder by Osawa et al.23 Aberrant cytoplasmic CDX2 expression may imply that CDX2 plays an important role in the early stages of intestinal metaplasia in the urinary bladder, and its expression may not be the end result of, but rather the trigger for, the development of intestinal metaplasia. Thus, aberrant CDX2 expression in metaplastic mucosal cells in a setting of background inflammmation may be an early indicator of subsequent intestinal differentiation. This hypothesis was supported by the study of Eda et al,22 in which they found that the expression of CDX2 precedes that of CDX1 and other intestine-specific genes during the development of intestinal metaplasia in the stomach.
CK20 is one of the cytoskeletal-associated intermediate filaments and its immunohistochemical expression has been reported in gastrointestinal epithelium, as well as in superficial urothelium, and to a lesser extent, intermediate urothelium.34, 35 It is regarded as a useful marker in the diagnosis of metastatic urothelial carcinomas in conjunction with other cytokeratin markers.36, 37, 38, 39 CK7 is another intermediate weight cytokeratin that is often expressed in normal urothelium, urothelial neoplasia, and in a proportion of glandular epithelial proliferations in bladder mucosa, whereas normal colonic epithelium and the majority of colonic adenocarcinomas lack CK7 immunoreactivity.34, 38 In the current study, we observed that coexpression of CK20 and CDX2 is very common in intestinal metaplasia in the urinary bladder, a reflection of the regulatory role of CDX2 in intestinal differentiation and the frequent expression of CK20 by intestinal-type tissue. The majority of cases of intestinal metaplasia of the urinary bladder lacked immunoreactivity with antibodies against CK7. In sharp contrast, moderate to strong CK7 expression was observed in all cases of typical cystitis glandularis, an expression pattern that is similar to that of normal urothelium. The divergent immunoprofiles suggest that these two subtypes of cystitis glandularis probably have fundamentally distinct molecular developmental pathways. Typical cystitis glandularis is probably more closely related to normal urothelium, as suggested by their virtually identical immunoprofiles. Intestinal metaplasia of the urinary bladder appears to represent a different part of the metaplastic spectrum, with morphologic and immunohistochemical characteristics that are markedly different from those of typical cystitis glandularis. However, this hypothesis would be more tenable if supported by appropriate molecular genetic analysis.
Hep is a cytoplasmic antigen present in normal hepatocytes and hepatic neoplasia. A monoclonal antibody against this antigen was developed by Wennerberg et al26 by using tissue from a formalin-fixed failed allograft liver. Initial reports showed it to be a sensitive and specific marker for hepatocellular differentiation.25, 26 However, subsequent studies have demonstrated its expression in adenocarcinomas in a variety of sites, including the esophagus, stomach, lung, colon, and female genital organs.40, 41 In addition, Chu et al28 proposed using Hep as a diagnostic tool for detection of intestinal metaplasia in the esophagus and stomach due to its superior sensitivity and specificity over other markers. However, no cases of intestinal metaplasia of the urinary bladder showed expression of Hep in our study, in contrast to its expression in the upper gastrointestinal tract as reported by Chu et al28 and Fan et al.29 These differing immunohistochemical profiles imply that divergent metaplastic pathways exist in the upper gastrointestinal tract and the urinary bladder, despite their identical morphological appearance. Additional studies are needed to further characterize the various pathogenetic mechanisms leading to intestinal-type metaplasia in different organ systems.
Primary adenocarcinoma of the urinary bladder is uncommon, accounting for less than 2% of all primary bladder tumors.42 The exact molecular genetic mechanisms underlying its development have not been elucidated. Although it is widely accepted that some cases of adenocarcinoma of the urinary bladder arise as a consequence of longstanding diffuse intestinal metaplasia, this notion has been challenged. There have been numerous reports in which adenocarcinoma of the urinary bladder has been seen in association with intestinal metaplasia of the urothelium;6, 7, 8, 9 however, a report by Corica et al43 showed that none of 53 patients with urinary bladder intestinal metaplasia developed bladder cancer in more than 10 years of follow up.43 The available data do not allow a conclusive appraisal of the natural history of intestinal metaplasia, and hence its exact clinical significance remains indeterminate. It is likely that investigations of intestinal metaplasia of the urothelium may contribute to an understanding of the pathogenesis of adenocarcinomas in the urinary tract.
The vital role of CDX2 in the development of intestinal metaplasia in the upper gastrointestinal tract has been well established.24 CDX2 can bind to the promoter of intestinal-specific genes, such as alkaline phosphatase, villin, or MUC2 and can activate subsequent transcription.44 Overexpression of CDX2 will induce differentiation to selected lineages of intestinal epithelial cells.44, 45 Mutoh et al46 found that longstanding intestinal metaplasia induced by CDX2 results in the development of intestinal-type adenocarcinomas in the stomachs of all CDX2-transgenic mice. These findings suggest that intestinal metaplasia itself may promote carcinogenesis through the sequential steps of the intestinal metaplasia–dysplasia–carcinoma sequence. Thus, detailed investigation of molecular events involving CDX2 in the upper gastrointestinal tract might provide us new insights into the ill-defined relationship between urothelial intestinal metaplasia and adenocarcinoma in the urinary bladder.
In his original article published in 1954, Mostofi47 used the term ‘cystitis glandularis’ to include those lesions with inward epithelial proliferation and resembling luminal or glandular structures, regardless of the presence or absence of mucus-secreting cells (goblet cells). The majority of authors in recent years, however, have separated cystitis glandularis into two subtypes with different morphologic findings.3, 4, 5, 30 The typical type is characterized by luminal structures within the lamina propria having an innermost lining of columnar or cuboidal cells, bounded peripherally by transitional cells. The intestinal type, also referred to as intestinal metaplasia, has a similar glandular architecture in the lamina propria but contains mucin-secreting goblet cells in the lining epithelium. According to these updated criteria, which were applied in our current study, those lesions containing goblet cells are classified as intestinal metaplasia, or cystitis glandularis of the intestinal type, rather than typical cystitis glandularis. Based on this simple and readily applicable definition, we believe that the distinct immunoprofiles observed in our investigation offer new insights into the nature of these two entities. We recommend that the term ‘intestinal metaplasia’ be used uniformly among the investigators. The term ‘cystitis glandularis of the intestinal type’ should be abandoned.
In conclusion, a panel of antibodies against CDX2, Hep, CK20, and CK7 revealed that intestinal metaplasia of the urinary bladder and typical cystitis glandularis have distinctly different immunoprofiles, a finding that emphasizes the importance of making the distinction between these two entities, and suggests that further investigation of the possible role of intestinal metaplasia in the pathogenesis of urinary tract adenocarcinoma is warranted. In addition, the absence of Hep staining in intestinal metaplasia of the urinary bladder, despite its morphologic resemblance to intestinal metaplasia in other organs, may signify the presence of unique metaplastic pathways in the urinary bladder.
References
Bell TE, Wendel RG . Cystitis glandularis: benign or malignant? J Urol 1968;100:462–465.
Davies G, Castro JE . Cystitis glandularis. Urology 1977;10:128–129.
Murphy WM, Grignon DJ, Perlman EJ . Tumors of the Kidney, Bladder, and Related Urinary Structures. American Register of Pathology: Washington, DC, 2004.
Young RH, Bostwick DG . Florid cystitis glandularis of intestinal type with mucin extravasation: a mimic of adenocarcinoma. Am J Surg Pathol 1996;20:1462–1468.
Jacobs LB, Brooks JD, Epstein JI . Differentiation of colonic metaplasia from adenocarcinoma of urinary bladder. Hum Pathol 1997;28:1152–1157.
Bullock PS, Thoni DE, Murphy WM . The significance of colonic mucosa (intestinal metaplasia) involving the urinary tract. Cancer 1987;59:2086–2090.
Edwards PD, Hurm RA, Jaeschke WH . Conversion of cystitis glandularis to adenocarcinoma. J Urol 1972;108:568–570.
Lin JI, Yong HS, Tseng CH, et al. Diffuse cystitis glandularis. Associated with adenocarcinomatous change. Urology 1980;15:411–415.
Susmano D, Rubenstein AB, Dakin AR, et al. Cystitis glandularis and adenocarcinoma of the bladder. J Urol 1971;105:671–674.
Freund JN, Domon-Dell C, Kedinger M, et al. The Cdx-1 and Cdx-2 homeobox genes in the intestine. Biochem Cell Biol 1998;76:957–969.
James R, Erler T, Kazenwadel J . Structure of the murine homeobox gene cdx-2. Expression in embryonic and adult intestinal epithelium. J Biol Chem 1994;269:15229–15237.
Silberg DG, Swain GP, Suh ER, et al. Cdx1 and cdx2 expression during intestinal development. Gastroenterology 2000;119:961–971.
Suh E, Chen L, Taylor J, et al. A homeodomain protein related to caudal regulates intestine-specific gene transcription. Mol Cell Biol 1994;14:7340–7351.
Suh E, Traber PG . An intestine-specific homeobox gene regulates proliferation and differentiation. Mol Cell Biol 1996;16:619–625.
Almeida R, Silva E, Santos-Silva F, et al. Expression of intestine-specific transcription factors, CDX1 and CDX2, in intestinal metaplasia and gastric carcinomas. J Pathol 2003;199:36–40.
Chu PG, Schwarz RE, Lau SK, et al. Immunohistochemical staining in the diagnosis of pancreatobiliary and ampulla of Vater adenocarcinoma: application of CDX2, CK17, MUC1, and MUC2. Am J Surg Pathol 2005;29:359–367.
De Lott LB, Morrison C, Suster S, et al. CDX2 is a useful marker of intestinal-type differentiation: a tissue microarray-based study of 629 tumors from various sites. Arch Pathol Lab Med 2005;129:1100–1105.
Ko S, Chu K, Luk JM, et al. CDX2 co-localizes with liver-intestine cadherin in intestinal metaplasia and adenocarcinoma of the stomach. J Pathol 2005;205:615–622.
Rossi G, Murer B, Cavazza A, et al. Primary mucinous (so-called colloid) carcinomas of the lung: a clinicopathologic and immunohistochemical study with special reference to CDX-2 homeobox gene and MUC2 expression. Am J Surg Pathol 2004;28:442–452.
Werling RW, Yaziji H, Bacchi CE, et al. CDX2, a highly sensitive and specific marker of adenocarcinomas of intestinal origin: an immunohistochemical survey of 476 primary and metastatic carcinomas. Am J Surg Pathol 2003;27:303–310.
Eda A, Osawa H, Satoh K, et al. Aberrant expression of CDX2 in Barrett's epithelium and inflammatory esophageal mucosa. J Gastroenterol 2003;38:14–22.
Eda A, Osawa H, Yanaka I, et al. Expression of homeobox gene CDX2 precedes that of CDX1 during the progression of intestinal metaplasia. J Gastroenterol 2002;37:94–100.
Osawa H, Kita H, Satoh K, et al. Aberrant expression of CDX2 in the metaplastic epithelium and inflammatory mucosa of the gallbladder. Am J Surg Pathol 2004;28:1253–1254.
Phillips RW, Frierson Jr HF, Moskaluk CA . Cdx2 as a marker of epithelial intestinal differentiation in the esophagus. Am J Surg Pathol 2003;27:1442–1447.
Minervini MI, Demetris AJ, Lee RG, et al. Utilization of hepatocyte-specific antibody in the immunocytochemical evaluation of liver tumors. Mod Pathol 1997;10:686–692.
Wennerberg AE, Nalesnik MA, Coleman WB . Hepatocyte paraffin 1: a monoclonal antibody that reacts with hepatocytes and can be used for differential diagnosis of hepatic tumors. Am J Pathol 1993;143:1050–1054.
Zimmerman RL, Burke MA, Young NA, et al. Diagnostic value of hepatocyte paraffin 1 antibody to discriminate hepatocellular carcinoma from metastatic carcinoma in fine-needle aspiration biopsies of the liver. Cancer 2001;93:288–291.
Chu PG, Jiang Z, Weiss LM . Hepatocyte antigen as a marker of intestinal metaplasia. Am J Surg Pathol 2003;27:952–959.
Fan Z, Li J, Dong B, et al. Expression of Cdx2 and hepatocyte antigen in gastric carcinoma: correlation with histologic type and implications for prognosis. Clin Cancer Res 2005;11:6162–6170.
Epstein JI, Amin MB, Reuter VE . Bladder Biopsy Interpretation. Lippincott Williams & Wilkins: Philadelphia, 2004.
Cheng L, Nagabhushan M, Pretlow TP, et al. Expression of E-cadherin in primary and metastatic prostate cancer. Am J Pathol 1996;148:1375–1380.
Cheng L, Pan CX, Zhang JT, et al. Loss of 14-3-3sigma in prostate cancer and its precursors. Clin Cancer Res 2004;10:3064–3068.
Zeng G, Hu Z, Kinch MS, et al. High-level expression of EphA2 receptor tyrosine kinase in prostatic intraepithelial neoplasia. Am J Pathol 2003;163:2271–2276.
Kende AI, Carr NJ, Sobin LH . Expression of cytokeratin 7 and 20 in carcinomas of the gastrointestinal tract. Histopathology 2003;42:137–140.
Mallofre C, Castillo M, Morente V, et al. Immunohistochemical expression of CK20, p53, and Ki-67 as objective markers of urothelial dysplasia. Mod Pathol 2003;16:187–191.
Emerson RE, Cheng L . Immunohistochemical markers in the evaluation of tumors of the urinary bladder: a review. Anal Quant Cytol Histol 2005;27:301–316.
Jiang J, Ulbright TM, Younger C, et al. Cytokeratin 7 and cytokeratin 20 in primary urinary bladder carcinoma and matched lymph node metastasis. Arch Pathol Lab Med 2001;125:921–923.
Chu PG, Wu E, Weiss LM . Cytokeratin 7 and cytokeratin 20 expression in epithelial neoplasms: a survey of 435 cases. Mod Pathol 2000;13:962–972.
Moll R, Lowe A, Laufer J, et al. Cytokeratin 20 in human carcinomas. A new histodiagnostic marker detected by monoclonal antibodies. Am J Pathol 1992;140:427–447.
Fan Z, van de Rijn MKM, Rouse RV . Hep par 1 antibody stain for the differential diagnosis of hepatocellular carcinoma: 676 tumors tested using tissue microarrays and conventional tissue sections. Mod Pathol 2003;16:137–144.
Kakar S, Muir T, Murphy L, et al. Immunoreactivity of Hep Par 1 in hepatic and extrahepatic tumors and its correlation with albumin in situ hybridization in hepatocellular carcinoma. Am J Clin Pathol 2003; 119:361–366.
Grignon DJ, Ro JY, Ayala AG, et al. Primary adenocarcinoma of the urinary bladder. A clinicopathologic analysis of 72 cases. Cancer 1991;67:2165–2172.
Corica FA, Husmann DA, Churchill BM, et al. Intestinal metaplasia is not a strong risk factor for bladder cancer: study of 53 cases with long-term follow-up. Urology 1997;50:427–431.
Silberg DG, Sullivan J, Kang E, et al. Cdx2 ectopic expression induces gastric intestinal metaplasia in transgenic mice. Gastroenterology 2002;122:689–696.
Mutoh H, Sakurai S, Satoh Y, et al. Cdx1 induced intestinal metaplasia in the transgenic mouse stomach: comparative study with Cdx2 transgenic mice. Gut 2004;53:1416–1423.
Mutoh H, Sakurai S, Satoh K, et al. Development of gastric carcinoma from intestinal metaplasia in Cdx2-transgenic mice. Cancer Res 2004;64:7740–7747.
Mostofi FK . Potentialities of bladder epithelium. J Urol 1954;71:705–714.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sung, MT., Lopez-Beltran, A., Eble, J. et al. Divergent pathway of intestinal metaplasia and cystitis glandularis of the urinary bladder. Mod Pathol 19, 1395–1401 (2006). https://doi.org/10.1038/modpathol.3800670
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/modpathol.3800670
Keywords
This article is cited by
-
Intestinal metaplasia of the urinary tract harbors potentially oncogenic genetic variants
Modern Pathology (2021)
-
SIU–ICUD on bladder cancer: pathology
World Journal of Urology (2019)
-
Next-generation sequencing-based molecular characterization of primary urinary bladder adenocarcinoma
Modern Pathology (2017)
-
Präneoplastische Läsionen und Vorstufen des Urothelkarzinoms
Der Pathologe (2016)
-
Unexplained association between cystitis glandularis and interstitial cystitis in females: a retrospective study
International Urogynecology Journal (2015)