Abstract
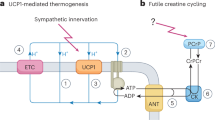
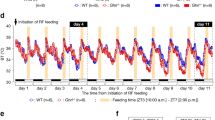
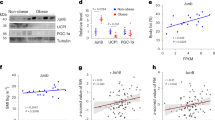
Brown adipose tissue is considered to be the main site of thermoregulatory non-shivering thermogenesis in the newborn of many mammalian species, in arousing hibernators, and in adult cold-adapted rats and mice1–4. Several recent studies have now suggested that the tissue may also be important in the regulation of energy balance. Evidence has been obtained indicating that the ability of genetically obese (ob/ob and db/db) mice to become obese on a normal energy intake is mainly due to reduced brown adipose tissue thermogenesis5–7, and a reduction in the activity of the tissue has also been observed in rats made obese by lesions of the ventromedial hypothalamus8. In contrast, increases in thermogenesis in brown adipose tissue have been implicated in the immediate thermic effect of food9 and in cancer cachexia10, and a substantial augmentation of the activity of the tissue has been demonstrated in rats and mice exhibiting regulatory diet-induced thermogenesis as a response to chronic voluntary over-feeding11–14. Whether or not brown adipose tissue thermogenesis changes in other, more physiological, states where energy flux and metabolic efficiency are altered (independently of environmental temperature) has not been established. We now report that the thermogenic activity of brown adipose tissue is suppressed in mice during the ‘physiological hyperphagia’ of lactation, and show that this probably results in a substantial reduction in the energy requirement for maintenance of the lactating animal.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Smith, R. E. & Horwitz, B. A. Physiol. Rev. 49, 330–425 (1969).
Jansky, L. Biol. Rev. 48, 85–132 (1973).
Foster, D. O. & Frydman, M. L. Can. J. Physiol. Pharmac. 56, 110–122 (1978).
Foster, D. O. & Frydman, M. L. Can. J. Physiol. Pharmac. 57, 257–270 (1979).
Himms-Hagen, J. & Desautels, M. Biochem. biophys. Res. Commun. 83, 628–634 (1978).
Thurlby, P. L. & Trayhurn, P. Pflügers Arch. ges. Physiol. 385, 193–201 (1980).
Goodbody, A. E. & Trayhurn, P. Biochem. J. 194, 1019–1022 (1981).
Seydoux, J., Rohner-Jeanrenaud, F., Assimacopoulos-Jeannet, F., Jeanrenaud, B. & Girardier, L. Pflügers Arch. ges. Physiol. 390, 1–4 (1981).
Glick, Z., Teague, R. J. & Bray, G. A. Science 213, 1125–1127 (1981).
Brooks, S. L., Neville, A. M., Rothwell, N. J., Stock, M. J. & Wilson, S. Biosci. Rep. 1, 509–517 (1981).
Rothwell, N. J. & Stock, M. J. Nature 281, 31–35 (1979).
Brooks, S. L., Rothwell, N. J., Stock, M. J., Goodbody, A. E. & Trayhurn, P. Nature 286, 274–276 (1980).
Rothwell, N. J. & Stock, M. J. Pflügers Arch. ges. Physiol. 389, 237–241 (1981).
Trayhurn, P., Jones, P. M., McGuckin, M. M. & Goodbody, A. E. Nature 295, 323–325 (1982).
Thurlby, P. L. & Trayhurn, P. Br. J. Nutr. 39, 397–402 (1978).
Nicholls, D. G. Biochim. biophys. Acta 549, 1–29 (1979).
Nicholls, D. G. Eur. J. Biochem. 62, 223–228 (1976).
Heaton, G. M., Wagenvoord, R. J., Kemp, A. & Nicholls, D. G. Eur. J. Biochem. 82, 515–521 (1978).
Rafael, J. & Heldt, H. W. FEBS Lett. 63, 304–308 (1976).
Desautels, M., Zaror-Behrens, G. & Himms-Hagen, J. Can. J. Biochem. 56, 378–383 (1978).
Desautels, M. & Himms-Hagen, J. Can. J. Biochem. 57, 968–976 (1979).
Sundin, U. & Cannon, B. Comp. Biochem. Physiol. 65B, 463–471 (1980).
Trayhurn, P. & James, W. P. T. Pflügers Arch. ges. Physiol. 373, 189–193 (1978).
Nicholls, D. G. Eur. J. Biochem. 49, 573–583 (1974).
Bukowiecki, L. J. & Lindberg, O. Biochim. biophys. Acta 348, 115–125 (1974).
Agius, L. & Williamson, D. H. Biochem. J. 190, 477–480 (1980).
Agius, L. & Williamson, D. H. Biochem. Soc. Trans. 8, 590–591 (1980).
Yonetani, T. & Ray, G. S. J. biol. Chem. 240, 3392–3398 (1965).
Schacterle, G. R. & Pollack, R. L. Analyt. Biochem. 51, 654–655 (1973).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Trayhurn, P., Douglas, J. & McGuckin, M. Brown adipose tissue thermogenesis is ‘suppressed’ during lactation in mice. Nature 298, 59–60 (1982). https://doi.org/10.1038/298059a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/298059a0
This article is cited by
-
Food supply and individual quality influence seabird energy expenditure and reproductive success
Oecologia (2022)
-
Mammary gland adipocytes in lactation cycle, obesity and breast cancer
Reviews in Endocrine and Metabolic Disorders (2021)
-
The role of autophagy in brown and beige adipose tissue plasticity
Journal of Physiology and Biochemistry (2020)
-
High-fat feeding reprograms maternal energy metabolism and induces long-term postpartum obesity in mice
International Journal of Obesity (2019)
-
A basic scientist’s odyssey in nutrition
European Journal of Clinical Nutrition (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.