Abstract
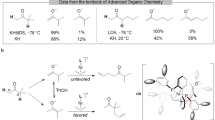
THE thermal reactions of o-phenylenediamine with carbonyl compounds have been investigated for a number of years1–3. Recently, Elderfield4–6 has extensively investigated the cleavage of ketones with this reagent, and has found that the reaction affords 2-alkylbenzimidazoles and alkanes in high yields. Investigations of the scope of the degradation have indicated that the more substituted group is eliminated as the alkane7–9. The mechanism of this elimination is not, however, known at the present time.
Similar content being viewed by others
Article PDF
References
Landenburg, A., and Rügheimer, L., Ber., 12, 951 (1879).
Thiele, J., and Steimmig, G., Ber., 40, 955 (1907).
Wright, J. B., Chem. Revs., 48, 397 (1907).
Elderfield, R. C., and Kreysa, F. J., J. Amer. Chem. Soc., 70, 44 (1948).
Elderfield, R. C., Kreyson, F. J., Dunn, J. H., and Humphreys, D. D., J. Amer. Chem. Soc., 70, 40 (1948).
Elderfield, R. C., and McCarthy, J. R., J. Amer. Chem. Soc., 73, 975 (1951).
Elderfield, R. C., and Meyer, V. B., J. Amer. Chem. Soc., 76, 1887 (1954).
Elderfield, R. C., and Burgess, K. L., J. Amer. Chem. Soc., 82, 1975 (1960).
Elderfield, R. C., and McClenachan, J. Amer. Chem. Soc., 82, 1982 (1960).
Risinger, G. E., and Mach, E. E., Nature, 199, 484 (1963).
Risinger, G. E., Nature, 198, 382 (1963); 200, 1203 (1963); 201, 700 (1964).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
RISINGER, G. Mechanism of the o-Phenylenediamine Cleavage of Ketones. Nature 209, 1022 (1966). https://doi.org/10.1038/2091022a0
Issue Date:
DOI: https://doi.org/10.1038/2091022a0
This article is cited by
-
Synthesis of some N-polyhydroxyalkyl-substituted benzimidazoles
Pharmaceutical Chemistry Journal (1967)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.