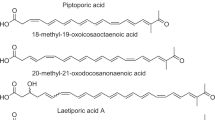
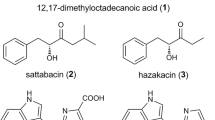
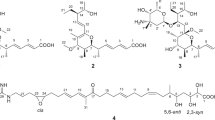
Abstract
CORYNOMYCOLIC acid, C32H64O3, m.p. 70°, [α]D = + 7.5° has been isolated from the lipids of Corynebacterium diphtheriæ 1 and C. ovis 2. Its structure (III, R = H) has been established by degradation3 and confirmed by synthesis: the condensation of two molecules of methyl palmitate (I, R = CH3) in presence of NaH gives the β-ketoester (II, R = CH3) which is reduced by NaBH4 to a mixture of two diastereo-isomers (III, R = CH3)4; one of these has been shown to be racemic methyl corynomycolate5.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Lederer, E., and Pudles, J., Bull. Soc. Chim. Biol., Paris, 33, 1003 (1951).
Diara, A., and Pudles, J., Bull. Soc. Chim. Biol., Paris, 41, 481 (1959).
Lederer, E., Pudles, J., Barbezat, S., and Trillat, J. J., Bull. Soc. Chim., France, 93 (1952).
Lederer, E., Portelance, V., and Serck-Hanssen, K., Bull. Soc. Chim., France, 413 (1952).
Polonsky, J., and Lederer, E., Bull. Soc. Chim., France, 504 (1954).
Pudles, J., Fourth Intern. Congr. Microbiol. comm., 99 (1953).
Asselineau, J., and Lederer, E., Progr. Chem. natur. Organ. compounds, 10, 170 (1953).
Pudles, J., and Lederer, E., Bull. Soc. Chim. Biol. Paris, 36, 759 (1954).
Asselineau, J., and Lederer, E., Ciba Foundation Symposium on Experimental Tuberculosis, 14 (J. and A. Churchill Ltd., London, 1955).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
GASTAMBIDE-ODIER, M., LEDERER, E. Biosynthesis of Corynomycolic Acid from Two Molecules of Palmitic Acid. Nature 184, 1563–1564 (1959). https://doi.org/10.1038/1841563b0
Issue Date:
DOI: https://doi.org/10.1038/1841563b0
This article is cited by
-
Cell walls of mycobacteria and related organisms; Chemistry and immunostimulant properties
Molecular and Cellular Biochemistry (1975)
-
Plant waxes
Lipids (1970)
-
Chemical Genetics of Nonacosan-15-one and Related Compounds in Brassica oleracea
Nature (1969)
-
Biogenesis of Nonacosan-15-one in Brassica oleracea : Dual Mechanisms in the Synthesis of Long-chain Compounds
Nature (1968)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.