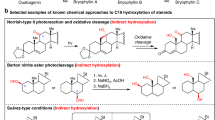
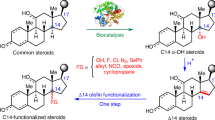
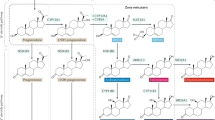
Abstract
Ruff and Reichstein1 have described a pregnan-7-ol and a pregnan-12-ol prepared by Wolff–Kishner reduction of acetoxypregnane ketones. They state that these two substances are probably the 7α- and 12α-hydroxy compounds; but since epimerization during Wolff–Kishner reduction is not excluded, they name them provisionally ‘7ξ’ and ‘12ξ’2. The molecular rotations of pregnan-7ξ-ol and of pregnan-12ξ-ol and their derivatives show that the two new compounds are indeed 7α and 12α.
Similar content being viewed by others
Article PDF
References
Ruff, A., and Reichstein, T., Helv. chim. Acta, 34, 70 (1951).
The letter ‘ξ’ is used to indicate that the configuration of the substituent is unknown. Cf. “Proposals for Steroid Nomenclature” made by a conference at the CIBA Foundation, London. Chem. and Indust., p. SN1 (June 23, 1951).
Barton, D. H. R., and Klyne, W., Chem. and Indust., 755 (1948).
Barton, D. H. R., J. Chem. Soc., 813 (1945).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
KLYNE, W. Pregnan-7-ol and Pregnan-12-ol. Nature 168, 835 (1951). https://doi.org/10.1038/168835a0
Issue Date:
DOI: https://doi.org/10.1038/168835a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.