Abstract
IT has been shown spectroscopically (the evidence will be published shortly1) that the nitrosonium ion in solution forms a molecular compound with nitrogen dioxide:  . As the compound may also be produced from nitric oxide and a solution of nitronium ion, one might also consider representing it thus:
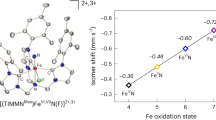
. As the compound may also be produced from nitric oxide and a solution of nitronium ion, one might also consider representing it thus:  . Actually, its spectral properties indicate that it has an intermediate constitution, but one which much more closely resembles structure I than II. Thus the high vibration frequency (2,240 cm.−1), depending on the multiple link in the NO-component of the complex, is displaced only one-seventh of the way from the frequency of the nitrosonium ion (2,300 cm.−1) towards the frequency of nitric oxide (1,880 cm.−1).
. Actually, its spectral properties indicate that it has an intermediate constitution, but one which much more closely resembles structure I than II. Thus the high vibration frequency (2,240 cm.−1), depending on the multiple link in the NO-component of the complex, is displaced only one-seventh of the way from the frequency of the nitrosonium ion (2,300 cm.−1) towards the frequency of nitric oxide (1,880 cm.−1).
Similar content being viewed by others
Article PDF
References
By Goulden and Millen, probably in J. Chem. Soc.
Nature, 147, 512 (1941); Trans. Farad. Soc., 37, 780 (1941); J. Chem. Soc., 245 (1942); 462 (1943).
Cf. Bateman, Hughes and Ingold, J. Chem. Soc., 243 (1944).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
GOULDEN, J., INGOLD, C. & MILLEN, D. The Ion N2O3+. Binding by One-Electron Transfer. Nature 165, 565 (1950). https://doi.org/10.1038/165565a0
Issue Date:
DOI: https://doi.org/10.1038/165565a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.