Abstract
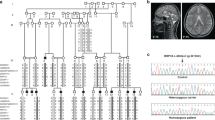
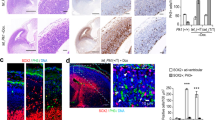
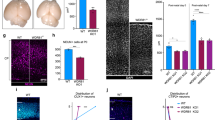
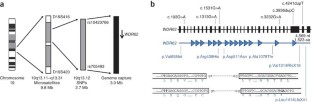
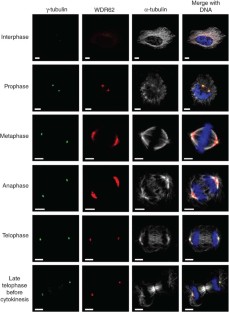
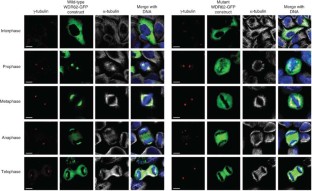
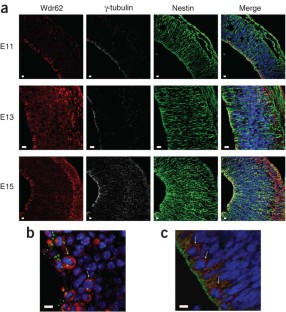
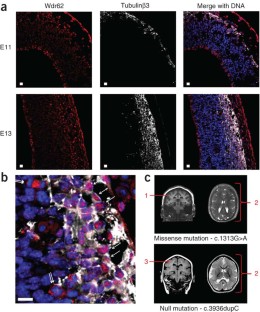
Autosomal recessive primary microcephaly (MCPH) is a disorder of neurodevelopment resulting in a small brain1,2. We identified WDR62 as the second most common cause of MCPH after finding homozygous missense and frame-shifting mutations in seven MCPH families. In human cell lines, we found that WDR62 is a spindle pole protein, as are ASPM and STIL, the MCPH7 and MCHP7 proteins3,4,5. Mutant WDR62 proteins failed to localize to the mitotic spindle pole. In human and mouse embryonic brain, we found that WDR62 expression was restricted to neural precursors undergoing mitosis. These data lend support to the hypothesis that the exquisite control of the cleavage furrow orientation in mammalian neural precursor cell mitosis, controlled in great part by the centrosomes and spindle poles, is critical both in causing MCPH when perturbed and, when modulated, generating the evolutionarily enlarged human brain6,7,8,9.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Mochida, G.H. & Walsh, C.A. Genetic basis of developmental malformations of the cerebral cortex. Arch. Neurol. 61, 637–640 (2004).
Woods, C.G., Bond, J. & Enard, W. Autosomal recessive primary microcephaly (MCPH): a review of clinical, molecular, and evolutionary findings. Am. J. Hum. Genet. 76, 717–728 (2005).
Bond, J. et al. ASPM is a major determinant of cerebral cortical size. Nat. Genet. 32, 316–320 (2002).
Pfaff, K.L. et al. The zebra fish cassiopeia mutant reveals that SIL is required for mitotic spindle organization. Mol. Cell. Biol. 27, 5887–5897 (2007).
Bond, J. & Woods, C.G. Cytoskeletal genes regulating brain size. Curr. Opin. Cell Biol. 18, 95–101 (2006).
Fish, J.L., Kosodo, Y., Enard, W., Pääbo, S. & Huttner, W.B. Aspm specifically maintains symmetric proliferative divisions of neuroepithelial cells. Proc. Natl. Acad. Sci. USA 103, 10438–10443 (2006).
Fish, J.L., Dehay, C., Kennedy, H. & Huttner, W.B. Making bigger brains–the evolution of neural-progenitor-cell division. J. Cell Sci. 121, 2783–2793 (2008).
Marthiens, V. & ffrench-Constant, C. Adherens junction domains are split by asymmetric division of embryonic neural stem cells. EMBO Rep. 10, 515–520 (2009).
Thornton, G.K. & Woods, C.G. Primary microcephaly: do all roads lead to Rome? Trends Genet. 25, 501–510 (2009).
Roberts, E. et al. The second locus for autosomal recessive primary microcephaly (MCPH2) maps to chromosome 19q13.1–13.2. Eur. J. Hum. Genet. 7, 815–820 (1999).
Jackson, A.P. et al. Identification of microcephalin, a protein implicated in determining the size of the human brain. Am. J. Hum. Genet. 71, 136–142 (2002).
Bond, J. et al. A centrosomal mechanism involving CDK5RAP2 and CENPJ controls brain size. Nat. Genet. 37, 353–355 (2005).
Kumar, A., Girimaji, S.C., Duvvari, M.R. & Blanton, S.H. Mutations in STIL, encoding a pericentriolar and centrosomal protein, cause primary microcephaly. Am. J. Hum. Genet. 84, 286–290 (2009).
Passemard, S. et al. Primary autosomal recessive microcephaly. GeneReviews. (University of Washington, Seattle, WA, 2009) <http://www.ncbi.nlm.nih.gov/pubmed/20301772>.
Roberts, E. et al. Autosomal recessive primary microcephaly: an analysis of locus heterogeneity and phenotypic variation. J. Med. Genet. 39, 718–721 (2002).
Mardis, E.R. New strategies and emerging technologies for massively parallel sequencing: applications in medical research. Genome Med. 1, 40 (2009).
Field, M. et al. Mutations in the BRWD3 gene cause X–linked mental retardation associated with macrocephaly. Am. J. Hum. Genet. 81, 367–374 (2007).
Nousiainen, M., Silljé, H.H., Sauer, G., Nigg, E.A. & Körner, R. Phosphoproteome analysis of the human mitotic spindle. Proc. Natl. Acad. Sci. USA 103, 5391–5396 (2006).
Malik, R. et al. Quantitative analysis of the human spindle phosphoproteome at distinct mitotic stages. J. Proteome Res. 8, 4553–4563 (2009).
do Carmo Avides, M., Tavares, A. & Glover, D.M. Polo kinase and Asp are needed to promote the mitotic organizing activity of centrosomes. Nat. Cell Biol. 3, 421–424 (2001).
Caspi, M. et al. LIS1 missense mutations: variable phenotypes result from unpredictable alterations in biochemical and cellular properties. J. Biol. Chem. 278, 38740–38748 (2003).
Gardette, R., Courtois, M. & Bisconte, J.C. Prenatal development of mouse central nervous structures: time of neuron origin and gradients of neuronal production. A radioautographic study. J. Hirnforsch. 23, 415–431 (1982).
Uylings, H.B. Development of the cerebral cortex in rodents and man. Eur. J. Morphol. 38, 309–312 (2000).
Bystron, I., Blakemore, C. & Rakic, P. Development of the human cerebral cortex: Boulder Committee revisited. Nat. Rev. Neurosci. 9, 110–122 (2008).
Attardo, A., Calegari, F., Haubensak, W., Wilsch-Brauninger, W.M. & Huttner, W.B. Live imaging at the onset of cortical neurogenesis reveals differential appearance of the neuronal phenotype in apical versus basal progenitor progeny. PLoS ONE 3, e2388 (2008).
Noctor, S.C., Martinez-Cerdeno, V. & Kriegstein, A.R. Distinct behaviors of neural stem and progenitor cells underlie cortical neurogenesis. J. Comp. Neurol. 508, 28–44 (2008).
O'Rahilly, R. & Müller, F. Developmental Stages in Human Embryos: Revised and New Measurements. Cells Tissues Organs 192, 73–84 (2010).
Bilgüvar, K. et al. Whole-exome sequencing identifies recessive WDR62 mutations in severe brain malformations. Nature. published online, doi:10.1038/nature09327 (22 August 2010).
Barr, F.A. & Gruneberg, U. Cytokinesis: placing and making the final cut. Cell 131, 847–860 (2007).
Barr, A.R., Kilmartin, J.V. & Gergely, F. CDK5RAP2 functions in centrosome to spindle pole attachment and DNA damage response. J. Cell Biol. 189, 23–39 (2010).
Götz, M. & Huttner, W.B. The cell biology of neurogenesis. Nat. Rev. Mol. Cell Biol. 6, 777–788 (2005).
Kosodo, Y. et al. Asymmetric distribution of the apical plasma membrane during neurogenic divisions of mammalian neuroepithelial cells. EMBO J. 23, 2314–2324 (2004).
Acknowledgements
The authors would like to thank the research families for their participation in this project and the Wellcome Trust, Medical Research Council, Action Research and the Higher Education Commission of Pakistan for funding (to A.K.N., M.K., O.P.C., J.J.C., G.T. and E.R.). J.D. was supported by the Belgian Kids' Fund. M.A. was supported by grants from the Fonds Erasme and the Belgian Fonds de la Recherche Scientifique Médicale (FRSM). We thank S. Strollo for expert technical assistance. We thank the Medical Research Council (MRC)-Wellcome Trust Human Developmental Biology Resource (HDBR), Newcastle for providing the human tissue for the expression studies.
Author information
Authors and Affiliations
Contributions
The following authors contributed to the design of the study: A.K.N., M.K., J.J.C., F.G., E.R., M. Abramowicz and C.G.W. The following authors generated experimental data: A.K.N., M.K., J.D., O.P.C., G.T., R.K., M. Ansar, F.G., W.B.D., E.R. and C.G.W. Reagents were contributed by R.K., M. Abramowicz, W.A., A.L., S.P., J.-P.M., S.L., M. Abramowicz and C.G.W. The paper was written by A.K.N., M.K., O.P.C., J.J.C., W.A., S.L., F.G., W.B.D. and C.G.W.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Table 1 and Supplementary Figures 1–7 (PDF 796 kb)
Rights and permissions
About this article
Cite this article
Nicholas, A., Khurshid, M., Désir, J. et al. WDR62 is associated with the spindle pole and is mutated in human microcephaly. Nat Genet 42, 1010–1014 (2010). https://doi.org/10.1038/ng.682
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.682
This article is cited by
-
WDR62-deficiency Causes Autism-like Behaviors Independent of Microcephaly in Mice
Neuroscience Bulletin (2022)
-
Novel phenotype and genotype spectrum of WDR62 in two patients with associated primary autosomal recessive microcephaly
Irish Journal of Medical Science (1971 -) (2022)
-
Molecular evolutionary analysis of human primary microcephaly genes
BMC Ecology and Evolution (2021)
-
WDR62 is required for centriole duplication in spermatogenesis and manchette removal in spermiogenesis
Communications Biology (2021)
-
The journey of Zika to the developing brain
Molecular Biology Reports (2020)