Abstract
Background:
To determine whether a variable definition of biochemical recurrence (BCR) based on clincopathologic features facilitates early identification of patients likely to suffer from disease progression. The definition of BCR after radical prostatectomy (RP) bears important implications for patient counseling and management; however, there remains a significant debate regarding the appropriate definition.
Methods:
The study cohort consisted of 3619 men who underwent RP for localized prostate cancer from 1989 to 2007, with data abstracted from the Cancer of the Prostate Strategic Urologic Research Endeavor (CaPSURE) registry. Patients were stratified into three risk groups according to Cancer of the Prostate Risk Assessment post-Surgical (CAPRA-S) score. Three single threshold PSA cut-points for BCR were evaluated (PSA⩾0.05, ⩾0.2 and ⩾0.4 ng ml−1) as well as a variable cut-point defined by risk group. After reaching the cut-points, patients were followed for further PSA progression.
Results:
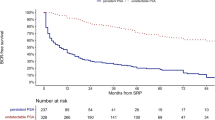
The proportion of patients with BCR differed by cut-point and risk group, ranging from 7 to 37% (low risk), 22 to 58% (intermediate risk) and 60 to 86% (high risk). The positive-predictive value (PPV) for predicting further PSA progression was 49% for the PSA⩾0.05 ng ml−1, 62% for the PSA⩾0.2 ng ml−1, 65% for the PSA⩾0.4 ng ml−1 and 68% for the risk-adjusted definition. Five-year progression-free survival was 39% for the risk-adjusted definition compared with 45–52% for the other definitions of BCR.
Conclusions:
These data suggest that a variable definition of BCR determined by clinicopathologic risk may improve the identification of early recurrence after RP without increasing the overdiagnosis of BCR. By using a risk-adjusted BCR definition, clinicians can better predict future PSA progression and more appropriately counsel patients regarding salvage therapies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Lange PH, Ercole CJ, Lightner DJ, Fraley EE, Vessella R . The value of serum prostate specific antigen determinations before and after radical prostatectomy. J Urol 1989; 141: 873–879.
Pound C, Partin AW, Eisenberger MA, Chan DW, Pearson JD, Walsh PC . Natural history of progression after PSA elevation following radical prostatectomy. JAMA 1999; 281: 1591–1597.
Freedland SJ, Humphreys EB, Mangold LA, Eisenberger M, Dorey FJ, Walsh PC et al. Risk of prostate cancer-specific mortality following biochemical recurrence after radical prostatectomy. JAMA 2005; 294: 433–439.
Stephenson AJ, Kattan MW, Eastham JA, Dotan ZA, Bianco Jr FJ, Lilja H et al. Defining biochemical recurrence of prostate cancer after radical prostatectomy: a proposal for a standardized definition. J Clin Oncol 2006; 24: 3973–3978.
Freedland SJ, Sutter ME, Dorey F, Aronson WJ . Defining the ideal cutpoint for determining PSA recurrence after radical prostatectomy. Prostate-specific antigen. Urology 2003; 61: 365–369.
Amling CL, Bergstralh EJ, Blute ML, Slezak JM, Zincke H . Defining prostate specific antigen progression after radical prostatectomy: what is the most appropriate cut point? J Urol 2001; 165: 1146–1151.
Boorjian SA, Thompson RH, Tollefson MK, Rangel LJ, Bergstralh EJ, Blute ML et al. Long-term risk of clinical progression after biochemical recurrence following radical prostatectomy: the impact of time from surgery to recurrence. Eur Urol 2011; 59: 893–899.
Nielsen ME, Makarov DV, Humphreys E, Mangold L, Partin AW, Walsh PC . Is it possible to compare PSA recurrence-free survival after surgery and radiotherapy using revised ASTRO criterion—‘nadir+2’? Urology 2008; 72: 389–393.
Cookson MS, Aus G, Burnett AL, Canby-Hagino ED, D'Amico AV, Dmochowski RR et al. Variation in the definition of biochemical recurrence in patients treated for localized prostate cancer: the American Urological Association Prostate Guidelines for Localized Prostate Cancer Update Panel report and recommendations for a standard in the reporting of surgical outcomes. J Urol 2007; 177: 540–545.
Stamey TA, Graves HC, Wehner N, Ferrari M, Freiha FS . Early detection of residual prostate cancer after radical prostatectomy by an ultrasensitive assay for prostate specific antigen. J Urol 1993; 149: 787–792.
Haese A, Huland E, Graefen M, Hammerer P, Noldus J, Huland H . Ultrasensitive detection of prostate specific antigen in the followup of 422 patients after radical prostatectomy. J Urol 1999; 161: 1206–1211.
Yu H, Diamandis EP, Wong PY, Nam R, Trachtenberg J . Detection of prostate cancer relapse with prostate specific antigen monitoring at levels of 0.001 to 0.1 microG./L. J Urol 1997; 157: 913–918.
Eisenberg ML, Davies BJ, Cooperberg MR, Cowan JE, Carroll PR . Prognostic implications of an undetectable ultrasensitive prostate-specific antigen level after radical prostatectomy. Eur Urol 2010; 57: 622–629.
Lubeck DP, Litwin MS, Henning JM, Stier DM, Mazonson P, Fisk R et al. The CaPSURE database: a methodology for clinical practice and research in prostate cancer. CaPSURE Research Panel. Cancer of the Prostate Strategic Urologic Research Endeavor. Urology 1996; 48: 773–777.
Cooperberg MR, Broering JM, Litwin MS, Lubeck DP, Mehta SS, Henning JM et al. The contemporary management of prostate cancer in the United States: lessons from the cancer of the prostate strategic urologic research endeavor (CapSURE), a national disease registry. J Urol 2004; 171: 1393–1401.
Cooperberg MR, Hilton JF, Carroll PR . The CAPRA-S score: a straightforward tool for improved prediction of outcomes after radical prostatectomy. Cancer 2011; 117: 5039–5046.
van Iersel MP, Thomas CM, Segers MF, Witjes WP, Debruyne FM, Oosterhof GO et al. The use of ‘ultrasensitive’ prostate-specific antigen assays in the detection of biochemical recurrence after radical prostatectomy. Br J Urol 1996; 77: 418–422.
Cronin AM, Godoy G, Vickers AJ . Definition of biochemical recurrence after radical prostatectomy does not substantially impact prognostic factor estimates. J Urol 2010; 183: 984–989.
Fowler JE, Brooks J, Pandey P, Seaver LE . Variable histology of anastomotic biopsies with detectable prostate specific antigen after radical prostatectomy. J Urol 1995; 153: 1011–1014.
Shah O, Melamed J, Lepor H . Analysis of apical soft tissue margins during radical retropubic prostatectomy. J Urol 2001; 165: 1943–1948.
Wood DP, Peretsman SJ, Seay TM . Incidence of benign and malignant prostate tissue in biopsies of the bladder neck after a radical prostatectomy. J Urol 1995; 154: 1443–1446.
Morgan TM, Lange PH, Porter MP, Lin DW, Ellis WJ, Gallaher IS et al. Disseminated tumor cells in prostate cancer patients after radical prostatectomy and without evidence of disease predicts biochemical recurrence. Clin Cancer Res 2009; 15: 677–683.
Iwakiri J, Granbois K, Wehner N, Graves HC, Stamey T . An analysis of urinary prostate specific antigen before and after radical prostatectomy: evidence for secretion of prostate specific antigen by the periurethral glands. J Urol 1993; 149: 783–786.
Levesque M, Hu H, D'Costa M, Diamandis EP . Prostate-specific antigen expression by various tumors. J Clin Lab Anal 1995; 9: 123–128.
Stephenson A, Scardino PT, Kattan MW, Pisansky TM, Slawin KM, Klein EA et al. Predicting the outcome of salvage radiation therapy for recurrent prostate cancer after radical prostatectomy. J Clin Oncol 2007; 25: 2035–2041.
Thompson IM, Tangen CM, Klein EA . Is there a standard of care for pathologic stage T3 prostate cancer? J Clin Oncol 2009; 27: 2898–2899.
Eastham JA, Kelly WK, Grossfeld GD, Small EJ, Cancer and Leukemia Group B. Cancer and Leukemia Group B (CALGB) 90203: a randomized phase 3 study of radical prostatectomy alone versus estramustine and docetaxel before radical prostatectomy for patients with high-risk localized disease. Urology 2003; 62 (Suppl 1): 55–62.
Montgomery B, Lavori P, Garzotto M, Lee K, Brophy M, Thaneemit-Chen S et al. Veterans Affairs Cooperative Studies Program study 553: Chemotherapy after prostatectomy, a phase III randomized study of prostatectomy versus prostatectomy with adjuvant docetaxel for patients with high-risk, localized prostate cancer. Urology 2008; 72: 474–480.
Sartor O . Endpoints in prostate cancer clinical trials. Urology 2002; 60: 101–107.
Katz MS, Zelefsky MJ, Venkatraman ES, Fuks Z, Hummer A, Leibel SA . Predictors of biochemical outcome with salvage conformal radiotherapy after radical prostatectomy for prostate cancer. J Clin Oncol 2003; 21: 483–489.
Stephenson AJ, Shariat SF, Zelefsky MJ, Kattan MW, Butler EB, Teh BS et al. Salvage radiotherapy for recurrent prostate cancer after radical prostatectomy. JAMA 2004; 291: 1325–1332.
Acknowledgements
The CaPSURE database is supported by the Department of Urology at the University of California, San Francisco.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Morgan, T., Meng, M., Cooperberg, M. et al. A risk-adjusted definition of biochemical recurrence after radical prostatectomy. Prostate Cancer Prostatic Dis 17, 174–179 (2014). https://doi.org/10.1038/pcan.2014.5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/pcan.2014.5
Keywords
This article is cited by
-
Adjuvant Versus Early Salvage Radiation Therapy Following Radical Prostatectomy for Men with Localized Prostate Cancer
Current Urology Reports (2017)