Abstract
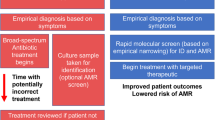
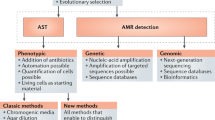
The best available data indicate that the world is heading towards a pandemic of extensively drug-resistant Neisseria gonorrhoeae. At the same time, clinical microbiology laboratories have moved away from using culture-based methods to diagnose gonorrhoea, thus undermining our ability to detect antimicrobial resistance (AMR) using current technologies. In this Opinion article, we discuss the problem of N. gonorrhoeae AMR, particularly emerging resistance to the cephalosporin ceftriaxone, outline current concerns about the surveillance of N. gonorrhoeae AMR and propose the use of molecular methods on a large scale to systematically enhance surveillance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Holmes, K. K. et al. in Sexually Transmitted Diseases 4th edn 627–645 (McGraw Hill, 2008).
Workowski, K. A., Berman, S. M. & Douglas, J. M. Emerging antimicrobial resistance in Neisseria gonorrhoeae: urgent need to strengthen prevention strategies. Ann. Intern. Med. 148, 606–613 (2008).
Barry, P. M. & Klausner, J. D. The use of cephalosporins for gonorrhea: the impending problem of resistance. Expert. Opin. Pharmacother. 10, 555–577 (2009).
Livermore, D. M. Has the era of untreatable infections arrived? J. Antimicrob. Chemother. 64, i29–i36 (2009).
Lewis, D. A. The Gonococcus fights back: is this time a knock out? Sex. Transm. Infect. 86, 415–421 (2010).
Kirkcaldy, R., Ballard, R. & Dowell, D. Gonococcal resistance: are cephalosporins next? Curr. Infect. Dis. Rep. 13, 196–204 (2011).
Tapsall, J. W. et al. Meeting the public health challenge of multidrug- and extensively drug-resistant Neisseria gonorrhoeae. Expert Rev. Anti Infect. Ther. 7, 821–834 (2009).
Ohnishi, M. et al. Is Neisseria gonorrhoeae initiating a future era of untreatable gonorrhea?: detailed characterization of the first strain with high-level resistance to ceftriaxone. Antimicrob. Agents Chemother. 55, 3538–3545 (2011).
Ohnishi, M. et al. High-level cefixime- and ceftriaxone-resistant Neisseria gonorrhoeae in France: novel penA mosaic allele in a successful international clone causes treatment failure. Antimicrob. Agents Chemother. 56, 1273–1280 (2012).
Ohnishi, M., Ono, E., Shimuta, K., Watanabe, H. & Okamura, N. Identification of TEM-135 β-lactamase in penicillinase-producing Neisseria gonorrhoeae strains in Japan. Antimicrob. Agents Chemother. 54, 3021–3023 (2010).
Nakayama, S.-I. et al. Molecular analyses of TEM genes and their corresponding penicillinase-producing Neisseria gonorrhoeae isolates in Bangkok, Thailand. Antimicrob. Agents Chemother. 56, 916–920 (2012).
Golparian, D. et al. In vitro activity of the new fluoroketolide solithromycin (CEM-101) against a large collection of clinical Neisseria gonorrhoeae isolates and international reference strains, including those with high-level antimicrobial resistance: potential treatment option for gonorrhea? Antimicrob. Agents Chemother. 56, 2739–2742 (2012).
Livermore, D. M. et al. Activity of ertapenem against Neisseria gonorrhoeae. J. Antimicrob. Chemother. 54, 280–281 (2004).
Ross, J. D. C. & Lewis, D. A. Cephalosporin resistant Neisseria gonorrhoeae: time to consider gentamicin? Sex. Transm. Infec. 88, 6–8 (2012).
Easmon, C. S. et al. Spectinomycin as initial treatment for gonorrhoea. BMJ 289, 1032–1034 (1984).
Kirkcaldy, B. Treatment of gonorrhea in an era of emerging cephalosporin resistance and results of a randomized trial of new potential treatment options. Sex. Trans. Infect. 89, A14 (2013).
Centers for Disease, Control and Prevention. Ceftriaxone-resistant Neisseria gonorrhoeae public health response plan [online], (2012).
Department of Reproductive Health and Research, World Health Organization. Global action plan to control the spread and impact of antimicrobial resistance in Neisseria gonorrhoeae [online], (2012).
Centers for Disease Control and Prevention. Update to CDC's sexually transmitted diseases treatment guidelines, 2010: oral cephalosporins no longer a recommended treatment for gonococcal infections. MMWR 61, 590–594 (2012).
Bignell, C. & Fitzgerald, M. UK national guideline for the management of gonorrhoea in adults, 2011. Int. J. STD AIDS. 22, 541–547 (2011).
Bignell, C. et al. 2012 European guideline on the diagnosis and treatment of gonorrhoea in adults. Int. J. STD AIDS 24, 85–92 (2013).
Tapsall, J. W. Antibiotic Resistance in Neisseria gonorrhoeae (WHO, 2001).
Zhao, S. et al. Genetics of chromosomally mediated intermediate resistance to ceftriaxone and cefixime in Neisseria gonorrhoeae. Antimicrob. Agents Chemother. 53, 3744–3751 (2009).
Ameyama, S. et al. Mosaic-like structure of penicillin-binding protein 2 gene (penA) in clinical isolates of Neisseria gonorrhoeae with reduced susceptibility to cefixime. Antimicrob. Agents Chemother. 46, 3744–3749 (2002).
Tomberg, J., Unemo, M., Davies, C. & Nicholas, R. A. Molecular and structural analysis of mosaic variants of penicillin-binding protein 2 conferring decreased susceptibility to expanded-spectrum cephalosporins in Neisseria gonorrhoeae: role of epistatic mutations. Biochemistry 49, 8062–8070 (2010).
Tomberg, J. et al. Identification of amino acids conferring high-level resistance to expanded-spectrum cephalosporins in the penA gene from Neisseria gonorrhoeae strain H041. Antimicrob. Agents Chemother. 57, 3029–3036 (2013).
Unemo, M. & Nicholas, R. A. Emergence of multidrug-resistant, extensively drug-resistant and untreatable gonorrhea. Future Microbiol. 7, 1401–1422 (2012).
Akasaka, S. et al. Emergence of cephem- and aztreonam-high-resistant Neisseria gonorrhoeae that does not produce β-lactamase. J. Infect. Chemother. 7, 49–50 (2001).
Tanaka, M. et al. Analysis of mutations within multiple genes associated with resistance in a clinical isolate of Neisseria gonorrhoeae with reduced ceftriaxone susceptibility that shows a multidrug-resistant phenotype. Int. J. Antimicrob. Agents 27, 20–26 (2006).
de Vries, H. J., van der Helm, J. J., Schim van der Loeff, M. F. & van Dam, A. P. Multidrug-resistant Neisseria gonorrhoeae with reduced cefotaxime susceptibility is increasingly common in men who have sex with men, Amsterdam, the Netherlands. Euro. Surveill. 17, 19330 (2009).
Australian Gonococcal Surveillance Programme. Annual report of Australian Gonococcal Surveillance Programme 2008. Commun. Dis. Intell. 34, 89–95 (2009).
Cole, M. J. et al. European surveillance of antimicrobial resistance in Neisseria gonorrhoeae. Sex. Transm. Infect. 86, 427–432 (2010).
Golparian, D., Hellmark, B., Fredlund, H. & Unemo, M. Emergence, spread and characteristics of Neisseria gonorrhoeae isolates with in vitro decreased susceptibility and resistance to extended-spectrum cephalosporins in Sweden. Sex. Transm. Infect. 86, 454–460 (2010).
Martin, I. et al. Trends in antimicrobial resistance in Neisseria gonorrhoeae isolated in Canada: 2000–2009. Sex. Transm. Dis. 38, 892–898 (2011).
Carannante, A. et al. Cefixime and ceftriaxone susceptibility of Neisseria gonorrhoeae in Italy from 2006 to 2010. Clin. Microbiol. Infect. 18, 558–564 (2012).
US Centers for Disease Control and Prevention. Cephalosporin susceptibility among Neisseria gonorrhoeae isolates — United States, 2000–2010. MMWR 60, 873–877 (2011).
Chisholm, S. A. et al. Emergence of a Neisseria gonorrhoeae clone showing decreased susceptibility to cefixime in England and Wales. J. Antimicrob. Chemother. 66, 2509–2512 (2011).
Whiley, D. M. et al. Reduced susceptibility to ceftriaxone in Neisseria gonorrhoeae is spread internationally by genetically distinct gonococcal populations. J. Antimicrob. Chemother. 66, 1186–1187 (2011).
Cámara, J. et al. Molecular characterization of two high-level ceftriaxone-resistant Neisseria gonorrhoeae isolates detected in Catalonia, Spain. J. Antimicrob. Chemother. 67, 1858–1860 (2012).
Bissessor, M. et al. Differing Neisseria gonorrhoeae bacterial loads in the pharynx and rectum in men who have sex with men: implications for gonococcal detection, transmission, and control. J. Clin. Microbiol. 49, 4304–4306 (2011).
Department of Reproductive Health and Research, World Health Organization. Strategies and laboratory methods for strengthening surveillance of sexually transmitted infection 2012. (WHO, 2012).
Martin, I. M. C. et al. Rapid sequence-based identification of gonococcal transmission clusters in a large metropolitan area. J. Infect. Dis. 189, 1497–1505 (2004).
Unemo, M. & Dillon, J. A. Review and international recommendation of methods for typing Neisseria gonorrhoeae isolates and their implications for improved knowledge of gonococcal epidemiology, treatment, and biology. Clin. Microbiol. Rev. 24, 447–458 (2011).
Palmer, H. M., Young, H., Graham, C. & Dave, J. Prediction of antibiotic resistance using Neisseria gonorrhoeae multi-antigen sequence typing. Sex. Transm. Infect. 84, 280–284 (2008).
Buono, S. et al. Using the Neisseria gonorrhoeae multi-antigen sequence-typing method to assess strain diversity and antibiotic resistance in San Francisco, California. Microb. Drug Resist. 18, 510–517 (2012).
Chisholm, S. A. et al. Molecular epidemiological typing within the European Gonococcal Antimicrobial Resistance Surveillance Programme reveals predominance of a multidrug-resistant clone. Euro Surveill. 18, 20358 (2013).
Whiley, D. M. et al. Neisseria gonorrhoeae multi-antigen sequence typing using non-cultured clinical specimens. Sex. Transm. Infect. 1, 51–55 (2010).
Siedner, M. J. et al. Real-time PCR assay for detection of quinolone-resistant Neisseria gonorrhoeae in urine samples. J. Clin. Microbiol. 45, 1250–1254 (2007).
Su, X. & Lind, I. Molecular basis of high-level ciprofloxacin resistance in Neisseria gonorrhoeae strains isolated in Denmark from 1995 to 1998. Antimicrob. Agents Chemother. 45, 117–123 (2001).
Magooa, M. P., Müller, E. E., Gumede, L. & Lewis, D. A. Determination of Neisseria gonorrhoeae susceptibility to ciprofloxacin in clinical specimens from men using a real-time PCR assay. Int. J. Antimicrob. Agents 42, 63–67 (2013).
Fluit, A. C., Visser, M. R. & Schmitz, F.-J. Molecular detection of antimicrobial resistance. Clin. Microbiol. Rev. 14, 836–871 (2001).
Goire, N. et al. Enhancing gonococcal antimicrobial resistance surveillance: a real-time PCR assay for detection of penicillinase-producing Neisseria gonorrhoeae by use of noncultured clinical samples. J. Clin. Microbiol. 2, 513–518 (2011).
Speers, D. J,, Fisk, R. E, Goire, N, & Mak, D. B. Non-culture Neisseria gonorrhoeae molecular penicillinase production surveillance demonstrates the long-term success of empirical dual therapy and informs gonorrhoea management guidelines in a highly endemic setting. J. Antimicrob. Chemother. http://dx.doi.org/10.1093/jac/dkt501; (2013).
Goire, N. et al. Enhanced gonococcal antimicrobial surveillance in the era of ceftriaxone resistance: a real-time PCR assay for direct detection of the Neisseria gonorrhoeae H041 strain. J. Antimicrob. Chemother. 67, 902–905 (2012).
Goire, N. et al. Polymerase chain reaction-based screening for the ceftriaxone-resistant Neisseria gonorrhoeae F89 strain. Euro Surveill. 18, 20444 (2013).
Gose, S. et al. Neisseria gonorrhoeae and extended-spectrum cephalosporins in California: surveillance and molecular detection of mosaic penA. BMC Infect. Dis. 13, 570 (2013).
Katz, A. R. et al. Neisseria gonorrhoeae with high-level resistance to azithromycin: case report of the first isolate identified in the United States. Clin. Infect. Dis. 54, 841–843 (2012).
Balashov, S., Mordechai, E., Adelson, M. E. & Gygax, S. E. Multiplex bead suspension array for screening Neisseria gonorrhoeae antibiotic resistance genetic determinants in noncultured clinical samples. J. Mol. Diagn. 15, 116–129 (2012).
Grad, Y. H. et al. Genomic epidemiology of Neisseria gonorrhoeae with reduced susceptibility to cefixime in the USA: a retrospective observational study. Lancet Infect. Dis. http:dx.doi.org/10.1016/S1473-3099(13)70693-5 (2014).
Tabrizi, S. N. et al. Analytical evaluation of GeneXpert CT/NG, the first genetic point-of-care assay for simultaneous detection of Neisseria gonorrhoeae and Chlamydia trachomatis. J. Clin. Microbiol. 51, 1945–1947 (2013).
Spizz, G. et al. Rheonix CARD® technology: an innovative and fully automated molecular diagnostic device. Point Care 11, 42–51 (2012).
Doseeva, V. et al. Multiplex isothermal helicase-dependent amplification assay for detection of Chlamydia trachomatis and Neisseria gonorrhoeae. Diagn. Microbiol. Infect. Dis. 71, 354–365 (2011).
Acknowledgements
This article results from the Gonorrhoea Resistance Assessment by Nucleic acid Detection (GRAND) study supported by an Australian National Health and Medical Research Council (NHMRC) Project Grant (APP1025517).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
DATABASES
Rights and permissions
About this article
Cite this article
Goire, N., Lahra, M., Chen, M. et al. Molecular approaches to enhance surveillance of gonococcal antimicrobial resistance. Nat Rev Microbiol 12, 223–229 (2014). https://doi.org/10.1038/nrmicro3217
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrmicro3217
This article is cited by
-
Antimicrobial resistance of Neisseria gonorrhoeae isolated from patients attending sexually transmitted infection clinics in Urban Hospitals, Lusaka, Zambia
BMC Infectious Diseases (2022)
-
Azithromycin resistant gonococci: a literature review
Antimicrobial Resistance & Infection Control (2020)
-
Gonorrhoea
Nature Reviews Disease Primers (2019)
-
Antimicrobial-resistant sexually transmitted infections: gonorrhoea and Mycoplasma genitalium
Nature Reviews Urology (2017)
-
A multicentre double-blind randomised controlled trial evaluating the efficacy of daily use of antibacterial mouthwash against oropharyngeal gonorrhoea among men who have sex with men: the OMEGA (Oral Mouthwash use to Eradicate GonorrhoeA) study protocol
BMC Infectious Diseases (2017)