Abstract
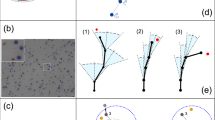
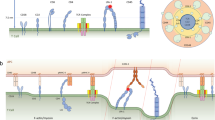
T cell migration is essential for T cell responses; it allows for the detection of cognate antigen at the surface of antigen-presenting cells and for interactions with other cells involved in the immune response. Although appearing random, growing evidence suggests that T cell motility patterns are strategic and governed by mechanisms that are optimized for both the activation stage of the cell and for environment-specific cues. In this Opinion article, we discuss how the combined effects of T cell-intrinsic and -extrinsic forces influence T cell motility patterns in the context of highly complex tissues that are filled with other cells involved in parallel motility. In particular, we examine how insights from 'search theory' can be used to describe T cell movement across an 'exploitation–exploration trade-off' in the context of activation versus effector function and lymph nodes versus peripheral tissues.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Blattman, J. N. et al. Estimating the precursor frequency of naive antigen-specific CD8 T cells. J. Exp. Med. 195, 657–664 (2002).
Jenkins, M. K., Chu, H. H., McLachlan, J. B. & Moon, J. J. On the composition of the preimmune repertoire of T cells specific for peptide-major histocompatibility complex ligands. Annu. Rev. Immunol. 28, 275–294 (2010).
Yu, W. et al. Clonal deletion prunes but does not eliminate self-specific αβ CD8+ T lymphocytes. Immunity 42, 929–941 (2015).
Bajenoff, M. et al. Highways, byways and breadcrumbs: directing lymphocyte traffic in the lymph node. Trends Immunol. 28, 346–352 (2007).
Fu, H., Wang, A., Mauro, C. & Marelli-Berg, F. T lymphocyte trafficking: molecules and mechanisms. Front. Biosci. (Landmark Ed.) 18, 422–440 (2013).
Ley, K. The second touch hypothesis: T cell activation, homing and polarization. F1000 Res. 3, 37 (2014).
Hills, T. T., Todd, P. M., Lazer, D., Redish, A. D. & Couzin, I. D. Exploration versus exploitation in space, mind, and society. Trends Cogn. Sci. 19, 46–54 (2015).
Codling, E. A., Plank, M. J. & Benhamou, S. Random walk models in biology. J. R. Soc. Interface 5, 813–834 (2008).
Miller, M. J., Wei, S. H., Cahalan, M. D. & Parker, I. Autonomous T cell trafficking examined in vivo with intravital two-photon microscopy. Proc. Natl Acad. Sci. USA 100, 2604–2609 (2003).
Cahalan, M. D. & Parker, I. Choreography of cell motility and interaction dynamics imaged by two-photon microscopy in lymphoid organs. Annu. Rev. Immunol. 26, 585–626 (2008).
Harris, T. H. et al. Generalized Levy walks and the role of chemokines in migration of effector CD8+ T cells. Nature 486, 545–548 (2012).
Worbs, T., Mempel, T. R., Bolter, J., von Andrian, U. H. & Forster, R. CCR7 ligands stimulate the intranodal motility of T lymphocytes in vivo. J. Exp. Med. 204, 489–495 (2007).
Witt, C. M., Raychaudhuri, S., Schaefer, B., Chakraborty, A. K. & Robey, E. A. Directed migration of positively selected thymocytes visualized in real time. PLoS Biol. 3, e160 (2005).
James, A., Plank, M. J. & Brown, R. Optimizing the encounter rate in biological interactions: ballistic versus Levy versus Brownian strategies. Phys. Rev. E 78, 051128 (2008).
Krummel, M. F., Sjaastad, M. D., Wulfing, C. & Davis, M. M. Differential clustering of CD4 and CD3ζ during T cell recognition. Science 289, 1349–1352 (2000).
Wulfing, C. et al. Kinetics and extent of T cell activation as measured with the calcium signal. J. Exp. Med. 185, 1815–1825 (1997).
Beauchemin, C., Dixit, N. M. & Perelson, A. S. Characterizing T cell movement within lymph nodes in the absence of antigen. J. Immunol. 178, 5505–5512 (2007).
Beltman, J. B., Henrickson, S. E., von Andrian, U. H., de Boer, R. J. & Maree, A. F. Towards estimating the true duration of dendritic cell interactions with T cells. J. Immunol. Methods 347, 54–69 (2009).
Preston, S. P., Waters, S. L., Jensen, O. E., Heaton, P. R. & Pritchard, D. I. T-cell motility in the early stages of the immune response modeled as a random walk amongst targets. Phys. Rev. E Stat. Nonlin. Soft Matter Phys. 74, 011910 (2006).
Textor, J. et al. Defining the quantitative limits of intravital two-photon lymphocyte tracking. Proc. Natl Acad. Sci. USA 108, 12401–12406 (2011).
Mrass, P., Petravic, J., Davenport, M. P. & Weninger, W. Cell-autonomous and environmental contributions to the interstitial migration of T cells. Semin. Immunopathol. 32, 257–274 (2010).
Miller, M. J., Wei, S. H., Parker, I. & Cahalan, M. D. Two-photon imaging of lymphocyte motility and antigen response in intact lymph node. Science 296, 1869–1873 (2002).
Benichou, O., Loverdo, C., Moreau, M. & Voituriez, R. Intermittent search strategies. Rev. Modern Phys. 83, 81–129 (2011).
Méndez, V., Campos, D. & Bartumeus, F. Stochastic Foundations in Movement Ecology (Springer, 2014).
Beltman, J. B., Maree, A. F., Lynch, J. N., Miller, M. J. & de Boer, R. J. Lymph node topology dictates T cell migration behavior. J. Exp. Med. 204, 771–780 (2007).
Woolf, E. et al. Lymph node chemokines promote sustained T lymphocyte motility without triggering stable integrin adhesiveness in the absence of shear forces. Nat. Immunol. 8, 1076–1085 (2007).
Lammermann, T. et al. Rapid leukocyte migration by integrin-independent flowing and squeezing. Nature 453, 51–55 (2008).
Katakai, T., Habiro, K. & Kinashi, T. Dendritic cells regulate high-speed interstitial T cell migration in the lymph node via LFA-1/ICAM-1. J. Immunol. 191, 1188–1199 (2013).
Banigan, E. J., Harris, T. H., Christian, D. A., Hunter, C. A. & Liu, A. J. Heterogeneous CD8+ T cell migration in the lymph node in the absence of inflammation revealed by quantitative migration analysis. PLoS Comput. Biol. 11, e1004058 (2015).
Bajenoff, M. et al. Stromal cell networks regulate lymphocyte entry, migration, and territoriality in lymph nodes. Immunity 25, 989–1001 (2006).
Katakai, T., Hara, T., Sugai, M., Gonda, H. & Shimizu, A. Lymph node fibroblastic reticular cells construct the stromal reticulum via contact with lymphocytes. J. Exp. Med. 200, 783–795 (2004).
Mempel, T. R., Junt, T. & von Andrian, U. H. Rulers over randomness: stroma cells guide lymphocyte migration in lymph nodes. Immunity 25, 867–869 (2006).
Malhotra, D. et al. Transcriptional profiling of stroma from inflamed and resting lymph nodes defines immunological hallmarks. Nat. Immunol. 13, 499–510 (2012).
Luther, S. A., Tang, H. L., Hyman, P. L., Farr, A. G. & Cyster, J. G. Coexpression of the chemokines ELC and SLC by T zone stromal cells and deletion of the ELC gene in the plt/plt mouse. Proc. Natl Acad. Sci. USA 97, 12694–12699 (2000).
Asperti-Boursin, F., Real, E., Bismuth, G., Trautmann, A. & Donnadieu, E. CCR7 ligands control basal T cell motility within lymph node slices in a phosphoinositide 3-kinase-independent manner. J. Exp. Med. 204, 1167–1179 (2007).
Katakai, T., Kondo, N., Ueda, Y. & Kinashi, T. Autotaxin produced by stromal cells promotes LFA-1-independent and Rho-dependent interstitial T cell motility in the lymph node paracortex. J. Immunol. 193, 617–626 (2014).
Graw, F. & Regoes, R. R. Influence of the fibroblastic reticular network on cell-cell interactions in lymphoid organs. PLoS Comput. Biol. 8, e1002436 (2012).
Gerard, A. et al. Detection of rare antigen-presenting cells through T cell-intrinsic meandering motility, mediated by Myo1g. Cell 158, 492–505 (2014).
Bartumeus, F., Raposo, E. P., Viswanathan, G. M. & da Luz, M. G. Stochastic optimal foraging: tuning intensive and extensive dynamics in random searches. PLoS ONE 9, e106373 (2014).
Mandl, J. N. et al. Quantification of lymph node transit times reveals differences in antigen surveillance strategies of naive CD4+ and CD8+ T cells. Proc. Natl Acad. Sci. USA 109, 18036–18041 (2012).
Friedl, P. & Gunzer, M. Interaction of T cells with APCs: the serial encounter model. Trends Immunol. 22, 187–191 (2001).
Gunzer, M. et al. Antigen presentation in extracellular matrix: interactions of T cells with dendritic cells are dynamic, short lived, and sequential. Immunity 13, 323–332 (2000).
Moreau, H. D. et al. Signal strength regulates antigen-mediated T-cell deceleration by distinct mechanisms to promote local exploration or arrest. Proc. Natl Acad. Sci. USA 112, 12151–12156 (2015).
Joshi, N. S. et al. Inflammation directs memory precursor and short-lived effector CD8+ T cell fates via the graded expression of T-bet transcription factor. Immunity 27, 281–295 (2007).
Badovinac, V. P., Messingham, K. A., Jabbari, A., Haring, J. S. & Harty, J. T. Accelerated CD8+ T-cell memory and prime-boost response after dendritic-cell vaccination. Nat. Med. 11, 748–756 (2005).
Kaech, S. M. & Wherry, E. J. Heterogeneity and cell-fate decisions in effector and memory CD8+ T cell differentiation during viral infection. Immunity 27, 393–405 (2007).
Gerner, M. Y., Torabi-Parizi, P. & Germain, R. N. Strategically localized dendritic cells promote rapid T cell responses to lymph-borne particulate antigens. Immunity 42, 172–185 (2015).
Woodruff, M. C. et al. Trans-nodal migration of resident dendritic cells into medullary interfollicular regions initiates immunity to influenza vaccine. J. Exp. Med. 211, 1611–1621 (2014).
Gerard, A. et al. Secondary T cell-T cell synaptic interactions drive the differentiation of protective CD8+ T cells. Nat. Immunol. 14, 356–363 (2013).
Sabatos, C. A. et al. A synaptic basis for paracrine interleukin-2 signaling during homotypic T cell interaction. Immunity 29, 238–248 (2008).
Ferguson, A. R. & Engelhard, V. H. CD8 T cells activated in distinct lymphoid organs differentially express adhesion proteins and coexpress multiple chemokine receptors. J. Immunol. 184, 4079–4086 (2010).
Sallusto, F. et al. Switch in chemokine receptor expression upon TCR stimulation reveals novel homing potential for recently activated T cells. Eur. J. Immunol. 29, 2037–2045 (1999).
Mueller, S. N. et al. Regulation of homeostatic chemokine expression and cell trafficking during immune responses. Science 317, 670–674 (2007).
Castellino, F. et al. Chemokines enhance immunity by guiding naive CD8+ T cells to sites of CD4+ T cell-dendritic cell interaction. Nature 440, 890–895 (2006).
Hugues, S. et al. Dynamic imaging of chemokine-dependent CD8+ T cell help for CD8+ T cell responses. Nat. Immunol. 8, 921–930 (2007).
Hickman, H. D. et al. Chemokines control naive CD8+ T cell selection of optimal lymph node antigen presenting cells. J. Exp. Med. 208, 2511–2524 (2011).
Eickhoff, S. et al. Robust anti-viral immunity requires multiple distinct T cell-dendritic cell interactions. Cell 162, 1322–1337 (2015).
Hor, J. L. et al. Spatiotemporally distinct interactions with dendritic cell subsets facilitates CD4 and CD8 T cell activation to localized viral infection. Immunity 43, 554–565 (2015).
Kelner, G. S. et al. Lymphotactin: a cytokine that represents a new class of chemokine. Science 266, 1395–1399 (1994).
Dorner, B. G. et al. Selective expression of the chemokine receptor XCR1 on cross-presenting dendritic cells determines cooperation with CD8+ T cells. Immunity 31, 823–833 (2009).
Lei, Y. & Takahama, Y. XCL1 and XCR1 in the immune system. Microbes Infect. 14, 262–267 (2012).
Groom, J. R. et al. CXCR3 chemokine receptor-ligand interactions in the lymph node optimize CD4+ T helper 1 cell differentiation. Immunity 37, 1091–1103 (2012).
Hu, J. K., Kagari, T., Clingan, J. M. & Matloubian, M. Expression of chemokine receptor CXCR3 on T cells affects the balance between effector and memory CD8 T-cell generation. Proc. Natl Acad. Sci. USA 108, E118–E127 (2011).
Kurachi, M. et al. Chemokine receptor CXCR3 facilitates CD8+ T cell differentiation into short-lived effector cells leading to memory degeneration. J. Exp. Med. 208, 1605–1620 (2011).
Campbell, D. J., Kim, C. H. & Butcher, E. C. Separable effector T cell populations specialized for B cell help or tissue inflammation. Nat. Immunol. 2, 876–881 (2001).
Reif, K. et al. Balanced responsiveness to chemoattractants from adjacent zones determines B-cell position. Nature 416, 94–99 (2002).
Okada, T. et al. Antigen-engaged B cells undergo chemotaxis toward the T zone and form motile conjugates with helper T cells. PLoS Biol. 3, e150 (2005).
Mempel, T. R., Henrickson, S. E. & Von Andrian, U. H. T-cell priming by dendritic cells in lymph nodes occurs in three distinct phases. Nature 427, 154–159 (2004).
Tan, J. T. & Surh, C. D. T cell memory. Curr. Top. Microbiol. Immunol. 311, 85–115 (2006).
Kastenmuller, W. et al. Peripheral prepositioning and local CXCL9 chemokine- mediated guidance orchestrate rapid memory CD8+ T cell responses in the lymph node. Immunity 38, 502–513 (2013).
Sung, J. H. et al. Chemokine guidance of central memory T cells is critical for antiviral recall responses in lymph nodes. Cell 150, 1249–1263 (2012).
Vergassola, M., Villermaux, E. & Shraiman, B. I. 'Infotaxis' as a strategy for searching without gradients. Nature 445, 406–409 (2007).
Griffith, J. W., Sokol, C. L. & Luster, A. D. Chemokines and chemokine receptors: positioning cells for host defense and immunity. Annu. Rev. Immunol. 32, 659–702 (2014).
Marelli-Berg, F. M., Cannella, L., Dazzi, F. & Mirenda, V. The highway code of T cell trafficking. J. Pathol. 214, 179–189 (2008).
Bromley, S. K., Mempel, T. R. & Luster, A. D. Orchestrating the orchestrators: chemokines in control of T cell traffic. Nat. Immunol. 9, 970–980 (2008).
Guo, L. et al. Innate immunological function of TH2 cells in vivo. Nat. Immunol. 16, 1051–1059 (2015).
Broz, M. L. et al. Dissecting the tumor myeloid compartment reveals rare activating antigen-presenting cells critical for T cell immunity. Cancer Cell 26, 638–652 (2014).
Chang, T. T., Jabs, C., Sobel, R. A., Kuchroo, V. K. & Sharpe, A. H. Studies in B7-deficient mice reveal a critical role for B7 costimulation in both induction and effector phases of experimental autoimmune encephalomyelitis. J. Exp. Med. 190, 733–740 (1999).
Krummel, M. F., Heath, W. R. & Allison, J. Differential coupling of second signals for cytotoxicity and proliferation in CD8+ T cell effectors: amplification of the lytic potential by B7. J. Immunol. 163, 2999–3006 (1999).
Lindsay, R. S. et al. Antigen recognition in the islets changes with progression of autoimmune islet infiltration. J. Immunol. 194, 522–530 (2015).
Friedman, R. S. et al. An evolving autoimmune microenvironment regulates the quality of effector T cell restimulation and function. Proc. Natl Acad. Sci. USA 111, 9223–9228 (2014).
Thornton, E. E. et al. Spatiotemporally separated antigen uptake by alveolar dendritic cells and airway presentation to T cells in the lung. J. Exp. Med. 209, 1183–1199 (2012).
Overstreet, M. G. et al. Inflammation-induced interstitial migration of effector CD4+ T cells is dependent on integrin αV. Nat. Immunol. 14, 949–958 (2013).
Honda, T. et al. Tuning of antigen sensitivity by T cell receptor-dependent negative feedback controls T cell effector function in inflamed tissues. Immunity 40, 235–247 (2014).
Sorokin, L. The impact of the extracellular matrix on inflammation. Nat. Rev. Immunol. 10, 712–723 (2010).
DiMilla, P. A., Barbee, K. & Lauffenburger, D. A. Mathematical model for the effects of adhesion and mechanics on cell migration speed. Biophys. J. 60, 15–37 (1991).
Baron, J. L., Reich, E. P., Visintin, I. & Janeway, C. A. Jr. The pathogenesis of adoptive murine autoimmune diabetes requires an interaction between α4- integrins and vascular cell adhesion molecule-1. J. Clin. Invest. 93, 1700–1708 (1994).
Ray, S. J. et al. The collagen binding α1β1 integrin VLA-1 regulates CD8 T cell-mediated immune protection against heterologous influenza infection. Immunity 20, 167–179 (2004).
Richter, M. et al. Collagen distribution and expression of collagen-binding α1β1 (VLA-1) and α2β1 (VLA-2) integrins on CD4 and CD8 T cells during influenza infection. J. Immunol. 178, 4506–4516 (2007).
Natsuaki, Y. et al. Perivascular leukocyte clusters are essential for efficient activation of effector T cells in the skin. Nat. Immunol. 15, 1064–1069 (2014).
Hickman, H. D. et al. CXCR3 chemokine receptor enables local CD8+ T cell migration for the destruction of virus-infected cells. Immunity 42, 524–537 (2015).
Groom, J. R. & Luster, A. D. CXCR3 in T cell function. Exp. Cell Res. 317, 620–631 (2011).
Ariotti, S. et al. Subtle CXCR3-dependent chemotaxis of CTLs within infected tissue allows efficient target localization. J. Immunol. 195, 5285–5295 (2015).
Wilson, E. H. et al. Behavior of parasite-specific effector CD8+ T cells in the brain and visualization of a kinesis-associated system of reticular fibers. Immunity 30, 300–311 (2009).
Herz, J. et al. In vivo imaging of lymphocytes in the CNS reveals different behaviour of naive T cells in health and autoimmunity. J. Neuroinflamm. 8, 131 (2011).
Bartholomaus, I. et al. Effector T cell interactions with meningeal vascular structures in nascent autoimmune CNS lesions. Nature 462, 94–98 (2009).
Kim, J. V. et al. Two-photon laser scanning microscopy imaging of intact spinal cord and cerebral cortex reveals requirement for CXCR6 and neuroinflammation in immune cell infiltration of cortical injury sites. J. Immunol. Methods 352, 89–100 (2010).
Kawakami, N. et al. Live imaging of effector cell trafficking and autoantigen recognition within the unfolding autoimmune encephalomyelitis lesion. J. Exp. Med. 201, 1805–1814 (2005).
Engelhardt, J. J. et al. Marginating dendritic cells of the tumor microenvironment cross-present tumor antigens and stably engage tumor-specific T cells. Cancer Cell 21, 402–417 (2012).
Joyce, J. A. & Fearon, D. T. T cell exclusion, immune privilege, and the tumor microenvironment. Science 348, 74–80 (2015).
Mrass, P. et al. Random migration precedes stable target cell interactions of tumor-infiltrating T cells. J. Exp. Med. 203, 2749–2761 (2006).
Salmon, H. et al. Matrix architecture defines the preferential localization and migration of T cells into the stroma of human lung tumors. J. Clin. Invest. 122, 899–910 (2012).
Mrass, P. et al. CD44 mediates successful interstitial navigation by killer T cells and enables efficient antitumor immunity. Immunity 29, 971–985 (2008).
Feig, C. et al. Targeting CXCL12 from FAP-expressing carcinoma-associated fibroblasts synergizes with anti-PD-L1 immunotherapy in pancreatic cancer. Proc. Natl Acad. Sci. USA 110, 20212–20217 (2013).
Boissonnas, A. et al. CD8+ tumor-infiltrating T cells are trapped in the tumor-dendritic cell network. Neoplasia 15, 85–94 (2013).
de Jager, M. et al. How superdiffusion gets arrested: ecological encounters explain shift from Levy to Brownian movement. Proc. R. Soc. B 281, 20132605 (2013).
Bartumeus, F. Behavioural ecology cannot turn its back on Levy walk research: comment on “Liberating Levy walk research from the shackles of optimal foraging” by A. M. Reynolds. Phys. Life Rev. 14, 84–86 (2015).
Letendre, K., Donnadieu, E., Moses, M. E. & Cannon, J. L. Bringing statistics up to speed with data in analysis of lymphocyte motility. PLoS ONE 10, e0126333 (2015).
Dusenbery, D. B. Sensory ecology: how organisms acquire and respond to information. (W. H. Freeman, 1992).
Mischiati, M. et al. Internal models direct dragonfly interception steering. Nature 517, 333–338 (2015).
Lima, S. L. & Zollner, P. A. Towards a behavioral ecology of ecological landscapes. Trends Ecol. Evol. 11, 131–135 (1996).
Nathan, R. et al. A movement ecology paradigm for unifying organismal movement research. Proc. Natl Acad. Sci. USA 105, 19052–19059 (2008).
Campos, D., Bartumeus, F. & Mendez, V. Search times with arbitrary detection constraints. Phys. Rev. E 88, 022101 (2013).
Viswanathan, G. M., da Luz, M. G. E., Raposo, E. P. & Stanley, H. E. The Physics of Foraging (Cambridge Univ. Press, 2011).
Dieterich, P., Klages, R., Preuss, R. & Schwab, A. Anomalous dynamics of cell migration. Proc. Natl Acad. Sci. USA 105, 459–463 (2008).
Acknowledgements
The authors thank E. Roberts for critical reading of the manuscript and the Advanced Study Group at Max-Planck-Institut für Physik komplexer Systeme (MPIPKS) on Statistical Physics and Anomalous Dynamics in Foraging for insightful discussions on anomalous processes. This work was supported by grants from US NIH (R01AI052116 and R01AI114787 to M.F.K.; R03AI119220 to A.G.) and the Human Frontier Science Program (RGY0084/2011 to F.B.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- Chemokinesis
-
Migration driven by soluble chemokines, without any cue gradient to provide a directional bias.
- Haptokinesis
-
Migration along a surface, utilizing immobilized ligands such as chemokines or integrins, without any cue gradient to provide a directional bias.
- Chemotaxis
-
Migration driven by a gradient of soluble chemokines, which occurs when an asymmetry in chemoattractant (such as rate and density) exists and local cue gradients can be followed.
- Haptotaxis
-
Migration along a surface that is guided by a gradient of immobilized chemoattractants or adhesion receptor ligands, which provides a directional bias.
Rights and permissions
About this article
Cite this article
Krummel, M., Bartumeus, F. & Gérard, A. T cell migration, search strategies and mechanisms. Nat Rev Immunol 16, 193–201 (2016). https://doi.org/10.1038/nri.2015.16
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri.2015.16
This article is cited by
-
Motility and tumor infiltration are key aspects of invariant natural killer T cell anti-tumor function
Nature Communications (2024)
-
Biomaterials to enhance adoptive cell therapy
Nature Reviews Bioengineering (2024)
-
Identification of a clinically efficacious CAR T cell subset in diffuse large B cell lymphoma by dynamic multidimensional single-cell profiling
Nature Cancer (2024)
-
Flow rate resonance of actively deforming particles
Scientific Reports (2023)
-
Localization, tissue biology and T cell state — implications for cancer immunotherapy
Nature Reviews Immunology (2023)