Abstract
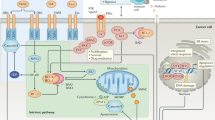
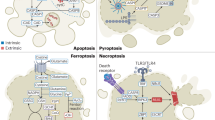
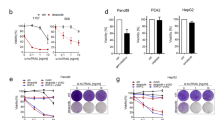
The inactivation of programmed cell death, or apoptosis, is central to the development of cancer. This disabling of apoptotic responses might be a major contributor both to treatment resistance and to the observation that, in many tumours, apoptosis is not the main mechanism for the death of cancer cells in response to common treatment regimens. Importantly, this suggests that other modes of cell death are involved in the response to therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Hanahan, D. & Weinberg, R. A. The hallmarks of cancer. Cell 100, 57–70 (2000).
Sarasin, A. An overview of the mechanisms of mutagenesis and carcinogenesis. Mutat. Res. 544, 99–106 (2003).
Levine, A. J. p53, the cellular gatekeeper for growth and division. Cell 88, 323–331 (1997).
Vousden, K. H. & Lu, X. Live or let die: the cell's response to p53. Nature Rev. Cancer 2, 594–604 (2002).
Attardi, L. D. The role of p53-mediated apoptosis as a crucial anti-tumor response to genomic instability: lessons from mouse models. Mutat. Res. 569, 145–157 (2005).
Schmitt, C. A. et al. A senescence program controlled by p53 and p16INK4a contributes to the outcome of cancer therapy. Cell 109, 335–346 (2002).
Hall, E. J. Radiobiology for the Radiologist (J. B. Lippincott Company, Philadelphia, 2000).
Wouters, B. G., Denko, N. C., Giaccia, A. J. & Brown, J. M. A p53 and apoptotic independent role for p21waf1 in tumour response to radiation therapy. Oncogene 18, 6540–6545 (1999).
Koike, M. et al. Dependence of chemotherapy response on p53 mutation status in a panel of human cancer lines maintained in nude mice. Cancer Sci. 95, 541–546 (2004).
Fichtner, I. et al. Anticancer drug response and expression of molecular markers in early-passage xenotransplanted colon carcinomas. Eur. J. Cancer 40, 298–307 (2004).
Lowe, S. W., Schmitt, E. M., Smith, S. W., Osborne, B. A. & Jacks, T. p53 is required for radiation-induced apoptosis in mouse thymocytes. Nature 362, 847–849 (1993).
Clarke, A. R. et al. Thymocyte apoptosis induced by p53-dependent and independent pathways. Nature 362, 849–852 (1993).
Lowe, S. W., Ruley, H. E., Jacks, T. & Housman, D. E. p53-dependent apoptosis modulates the cytotoxicity of anticancer agents. Cell 74, 957–967 (1993).
Lowe, S. W. et al. p53 status and the efficacy of cancer therapy in vivo. Science 266, 807–810 (1994).
Weinstein, J. N. et al. An information-intensive approach to the molecular pharmacology of cancer. Science 275, 343–349 (1997).
Amundson, S. A. et al. An informatics approach identifying markers of chemosensitivity in human cancer cell lines. Cancer Res. 60, 6101–6110 (2000).
Abend, M. Reasons to reconsider the significance of apoptosis for cancer therapy. Int. J. Radiat. Biol. 79, 927–941 (2003).
Okada, H. & Mak, T. W. Pathways of apoptotic and non-apoptotic death in tumour cells. Nature Rev. Cancer 4, 592–603 (2004).
Zong, W. X., Ditsworth, D., Bauer, D. E., Wang, Z. Q. & Thompson, C. B. Alkylating DNA damage stimulates a regulated form of necrotic cell death. Genes Dev. 18, 1272–1282 (2004).
Chan, T. A., Hermeking, H., Lengauer, C., Kinzler, K. W. & Vogelstein, B. 14-3-3σ is required to prevent mitotic catastrophe after DNA damage. Nature 401, 616–620 (1999).
Chu, K., Teele, N., Dewey, M. W., Albright, N. & Dewey, W. C. Computerized video time lapse study of cell cycle delay and arrest, mitotic catastrophe, apoptosis and clonogenic survival in irradiated 14-3-3σ and CDKN1A (p21) knockout cell lines. Radiat. Res. 162, 270–286 (2004).
Forrester, H. B., Albright, N., Ling, C. C. & Dewey, W. C. Computerized video time-lapse analysis of apoptosis of REC:Myc cells X-irradiated in different phases of the cell cycle. Radiat. Res. 154, 625–639 (2000).
Masters, J. R. & Koberle, B. Curing metastatic cancer: lessons from testicular germ-cell tumours. Nature Rev. Cancer 3, 517–525 (2003).
Taniguchi, T. et al. Disruption of the Fanconi anemia–BRCA pathway in cisplatin-sensitive ovarian tumors. Nature Med. 9, 568–574 (2003).
Gurubhagavatula, S. et al. XPD and XRCC1 genetic polymorphisms are prognostic factors in advanced non-small-cell lung cancer patients treated with platinum chemotherapy. J. Clin. Oncol. 22, 2594–2601 (2004).
Endlich, B., Radford, I. R., Forrester, H. B. & Dewey, W. C. Computerized video time-lapse microscopy studies of ionizing radiation-induced rapid-interphase and mitosis-related apoptosis in lymphoid cells. Radiat. Res. 153, 36–48 (2000).
Puck, T. T. & Markus, P. I. Action of x-rays on mammalian cells. J. Exper. Med. 103, 653–666 (1956).
Reinhold, H. S. & De Bree, C. Tumour cure rate and cell survival of a transplantable rat rhabdomyosarcoma following x-irradiation. Eur. J. Cancer 4, 367–374 (1968).
Durand, R. E. Cure, regression and cell survival: a comparison of common radiobiological endpoints using an in vitro tumour model. Br. J. Radiol. 48, 556–571 (1975).
Teicher, B. A. in Molecular Cancer Therapeutics: Strategies for Drug Discovery and Development (ed. Prendergast, G. C.) 7–40 (John Wiley and Sons, Hoboken, New Jersey, 2004).
Waldman, T. et al. Cell-cycle arrest versus cell death in cancer therapy. Nature Med. 3, 1034–1036 (1997).
Wouters, B. G., Giaccia, A. J., Denko, N. C. & Brown, J. M. Loss of p21Waf1/Cip1 sensitizes tumors to radiation by an apoptosis- independent mechanism. Cancer Res. 57, 4703–4706 (1997).
Bunz, F. et al. Disruption of p53 in human cancer cells alters the responses to therapeutic agents. J. Clin. Invest. 104, 263–269 (1999).
Brown, J. M. NCI's anticancer drug screening program may not be selecting for clinically active compounds. Oncol. Res. 9, 213–215 (1997).
Ruth, A. C. & Roninson, I. B. Effects of the multidrug transporter P-glycoprotein on cellular responses to ionizing radiation. Cancer Res. 60, 2576–2578 (2000).
Brown, J. M. & Giaccia, A. J. The unique physiology of solid tumors: opportunities (and problems) for cancer therapy. Cancer Res. 58, 1408–1416 (1998).
Schmitt, C. A., Rosenthal, C. T. & Lowe, S. W. Genetic analysis of chemoresistance in primary murine lymphomas. Nature Med. 6, 1029–1035 (2000).
Withers, H. R., Mason, K. A. & Thames, H. D. Jr. Late radiation response of kidney assayed by tubule-cell survival. Br. J. Radiol. 59, 587–595 (1986).
Tucker, S. L. et al. Direct analyses of in vivo colony survival after single and fractionated doses of radiation. Int. J. Radiat. Biol. 59, 777–795 (1991).
Hendry, J. H., Cai, W. B., Roberts, S. A. & Potten, C. S. p53 deficiency sensitizes clonogenic cells to irradiation in the large but not the small intestine. Radiat. Res. 148, 254–259 (1997).
Potten, C. S. & Grant, H. K. The relationship between ionizing radiation-induced apoptosis and stem cells in the small and large intestine. Br. J. Cancer 78, 993–1003 (1998).
Komarova, E. A. et al. Transgenic mice with p53-responsive lacZ: p53 activity varies dramatically during normal development and determines radiation and drug sensitivity in vivo. EMBO J. 16, 1391–1400 (1997).
Song, S. & Lambert, P. F. Different responses of epidermal and hair follicular cells to radiation correlate with distinct patterns of p53 and p21 induction. Am. J. Pathol. 155, 1121–1127 (1999).
Gudkov, A. V. & Komarova, E. A. The role of p53 in determining sensitivity to radiotherapy. Nature Rev. Cancer 3, 117–129 (2003).
Held, K. D. Radiation-induced apoptosis and its relationship to loss of clonogenic survival. Apoptosis 2, 265–282 (1997).
Aldridge, D. R., Arends, M. J. & Radford, I. R. Increasing the susceptibility of the rat 208F fibroblast cell line to radiation-induced apoptosis does not alter its clonogenic survival dose–response. Br. J. Cancer 71, 571–577 (1995).
Han, J. W., Dionne, C. A., Kedersha, N. L. & Goldmacher, V. S. p53 status affects the rate of the onset but not the overall extent of doxorubicin-induced cell death in rat-1 fibroblasts constitutively expressing c-Myc. Cancer Res. 57, 176–182 (1997).
Lock, R. B. & Stribinskiene, L. Dual modes of death induced by etoposide in human epithelial tumor cells allow Bcl-2 to inhibit apoptosis without affecting clonogenic survival. Cancer Res. 56, 4006–4012 (1996).
Yin, D. X. & Schimke, R. T. BCL-2 expression delays drug-induced apoptosis but does not increase clonogenic survival after drug treatment in HeLa cells. Cancer Res. 55, 4922–4928 (1995).
Tannock, I. F. & Lee, C. Evidence against apoptosis as a major mechanism for reproductive cell death following treatment of cell lines with anti-cancer drugs. Br. J. Cancer 84, 100–105 (2001).
Ekert, P. G. et al. Apaf-1 and caspase-9 accelerate apoptosis, but do not determine whether factor-deprived or drug-treated cells die. J. Cell Biol. 165, 835–842 (2004).
Westphal, C. H. et al. atm and p53 cooperate in apoptosis and suppression of tumorigenesis, but not in resistance to acute radiation toxicity. Nature Genet. 16, 397–401 (1997).
Merritt, A. J., Allen, T. D., Potten, C. S. & Hickman, J. A. Apoptosis in small intestinal epithelial from p53-null mice: evidence for a delayed, p53-independent G2/M-associated cell death after γ-irradiation. Oncogene 14, 2759–2766 (1997).
Kemp, C. J., Sun, S. & Gurley, K. E. p53 induction and apoptosis in response to radio- and chemotherapy in vivo is tumor-type-dependent. Cancer Res. 61, 327–332 (2001).
Ling, V. Multidrug resistance: molecular mechanisms and clinical relevance. Cancer Chemother. Pharmacol. 40 (Suppl.), 3–8 (1997).
Tannock, I. F. Tumor physiology and drug resistance. Cancer Metastasis Rev. 20, 123–132 (2001).
Townsend, D. M. & Tew, K. D. The role of glutathione-S-transferase in anti-cancer drug resistance. Oncogene 22, 7369–7375 (2003).
Brown, J. M. & Wilson, G. Apoptosis genes and resistance to cancer therapy: what do the experimental and clinical data tell us? Cancer Biol. Ther. 2, 477–490 (2003).
Altieri, D. C. The molecular basis and potential role of survivin in cancer diagnosis and therapy. Trends Mol. Med. 7, 542–547 (2001).
Thames, H. D., Petersen, C., Petersen, S., Nieder, C. & Baumann, M. Immunohistochemically detected p53 mutations in epithelial tumors and results of treatment with chemotherapy and radiotherapy. A treatment-specific overview of the clinical data. Strahlenther. Onkol. 178, 411–421 (2002).
Schmitt, C. A. & Lowe, S. W. Apoptosis and therapy. J. Pathol. 187, 127–137 (1999).
Kovacs, M. S., Yudoh, K., Evans, J. W., Menke, D. & Brown, J. M. Stable translocations detected by fluorescence in situ hybridization: a rapid surrogate end point to evaluate the efficacy of a potentiator of tumor response to radiotherapy. Cancer Res. 57, 672–677 (1997).
Castedo, M. et al. Cell death by mitotic catastrophe: a molecular definition. Oncogene 23, 2825–2837 (2004).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Brown, J., Attardi, L. The role of apoptosis in cancer development and treatment response. Nat Rev Cancer 5, 231–237 (2005). https://doi.org/10.1038/nrc1560
Issue Date:
DOI: https://doi.org/10.1038/nrc1560
This article is cited by
-
Apoptosis induction in cancer cell lines and anti-inflammatory and anti-pathogenic properties of proteinaceous metabolites secreted from potential probiotic Enterococcus faecalis KUMS-T48
Scientific Reports (2023)
-
Valrubicin decreases cell proliferation in gastric cancer through the apoptosis induction
Molecular & Cellular Toxicology (2023)
-
Repositioning of simvastatin for diabetic colon cancer: role of CDK4 inhibition and apoptosis
Molecular and Cellular Biochemistry (2023)
-
Valproic acid radiosensitizes anaplastic thyroid cells through a decrease of the DNA damage repair capacity
Journal of Endocrinological Investigation (2023)
-
RNF7 inhibits apoptosis and sunitinib sensitivity and promotes glycolysis in renal cell carcinoma via the SOCS1/JAK/STAT3 feedback loop
Cellular & Molecular Biology Letters (2022)