Abstract
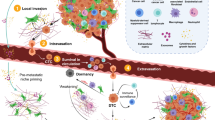
Stromal cells have been studied extensively in the primary tumor microenvironment. In addition, mesenchymal stromal cells may participate in several steps of the metastatic cascade. Studying this interaction requires methods to distinguish and target stromal cells originating from the primary tumor versus their counterparts in the metastatic site. Here we illustrate a model of human tumor stromal cell—mouse cancer cell coimplantation. This model can be used to selectively deplete human stromal cells (using diphtheria toxin, DT) without affecting mouse cancer cells or host-derived stromal cells. Establishment of novel genetic models (e.g., transgenic expression of the DT receptor in specific cells) may eventually allow analogous models using syngeneic cells. Studying the role of stromal cells in metastasis using the model outlined above may take 8 weeks.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Bhowmick, N.A. et al. TGF-β signaling in fibroblasts modulates the oncogenic potential of adjacent epithelia. Science 303, 848–851 (2004).
Jacobs, T.W., Byrne, C., Colditz, G., Connolly, J.L. & Schnitt, S.J. Radial scars in benign breast-biopsy specimens and the risk of breast cancer. N. Engl. J. Med. 340, 430–436 (1999).
Elenbaas, B. et al. Human breast cancer cells generated by oncogenic transformation of primary mammary epithelial cells. Genes Dev. 15, 50–65 (2001).
Bhowmick, N.A., Neilson, E.G. & Moses, H.L. Stromal fibroblasts in cancer initiation and progression. Nature 432, 332–337 (2004).
Olumi, A.F. et al. Carcinoma-associated fibroblasts direct tumor progression of initiated human prostatic epithelium. Cancer Res. 59, 5002–5011 (1999).
Orimo, A. et al. Stromal fibroblasts present in invasive human breast carcinomas promote tumor growth and angiogenesis through elevated SDF-1/CXCL12 secretion. Cell 121, 335–348 (2005).
Tlsty, T.D. Stromal cells can contribute oncogenic signals. Semin. Cancer Biol. 11, 97–104 (2001).
Kalluri, R. & Zeisberg, M. Fibroblasts in cancer. Nat. Rev. Cancer 6, 392–401 (2006).
O'Connell, J.T. et al. VEGF-A and Tenascin-C produced by S100A4+ stromal cells are important for metastatic colonization. Proc. Natl. Acad. Sci. USA 108, 16002–16007 (2011).
Podsypanina, K. et al. Seeding and propagation of untransformed mouse mammary cells in the lung. Science 321, 1841–1844 (2008).
Hiratsuka, S. et al. MMP9 induction by vascular endothelial growth factor receptor-1 is involved in lung-specific metastasis. Cancer Cell 2, 289–300 (2002).
Kaplan, R.N. et al. VEGFR1-positive haematopoietic bone marrow progenitors initiate the pre-metastatic niche. Nature 438, 820–827 (2005).
Kim, S. et al. Carcinoma-produced factors activate myeloid cells through TLR2 to stimulate metastasis. Nature 457, 102–106 (2009).
Deventer, K., Pozo, O.J., Van Eenoo, P. & Delbeke, F.T. Development and validation of an LC-MS/MS method for the quantification of ephedrines in urine. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 877, 369–374 (2009).
Al-Mehdi, A.B. et al. Intravascular origin of metastasis from the proliferation of endothelium-attached tumor cells: a new model for metastasis. Nat. Med. 6, 100–102 (2000).
Liotta, L.A., Saidel, M.G. & Kleinerman, J. The significance of hematogenous tumor cell clumps in the metastatic process. Cancer Res. 36, 889–894 (1976).
Fidler, I.J. The relationship of embolic homogeneity, number, size and viability to the incidence of experimental metastasis. Eur. J. Cancer 9, 223–227 (1973).
Ruiter, D.J., van Krieken, J.H., van Muijen, G.N. & deWaal, R.M. Tumour metastasis: is tissue an issue? Lancet Oncol. 2, 109–112 (2001).
Fidler, I.J. The pathogenesis of cancer metastasis: the 'seed and soil' hypothesis revisited. Nat. Rev. Cancer 3, 453–458 (2003).
Sahai, E. Illuminating the metastatic process. Nat. Rev. Cancer 7, 737–749 (2007).
Duda, D.G. et al. Malignant cells facilitate lung metastasis by bringing their own soil. Proc. Natl. Acad. Sci. USA 107, 21677–21682 (2010).
Duyverman, A.M.M.J., Kohno, M., Duda, D.G., Jain, R.K. & Fukumura, D. A transient parabiosis skin transplantation model in mice. Nat. Protoc. 7, 763–770 (2012).
Duyverman, A.M.M.J. et al. An isolated tumor perfusion model in mice. Nat. Protoc. 7, 749–755 (2012).
Arbiser, J.L. et al. Isolation of mouse stromal cells associated with a human tumor using differential diphtheria toxin sensitivity. Am. J. Pathol. 155, 723–729 (1999).
Padera, T.P. et al. Pathology: cancer cells compress intratumour vessels. Nature 427, 695 (2004).
Padera, T.P. et al. Lymphatic metastasis in the absence of functional intratumor lymphatics. Science 296, 1883–1886 (2002).
Dawson, M.R., Duda, D.G., Fukumura, D. & Jain, R.K. VEGFR1-activity-independent metastasis formation. Nature 461, E4–E5 (2009).
Hiratsuka, S. et al. C-X-C receptor type 4 promotes metastasis by activating p38 mitogen-activated protein kinase in myeloid differentiation antigen (Gr-1)-positive cells. Proc. Natl. Acad. Sci. USA 108, 302–307 (2011).
Tian, B., Han, L., Kleidon, J. & Henke, C. An HSV-TK transgenic mouse model to evaluate elimination of fibroblasts for fibrosis therapy. Am. J. Pathol. 163, 789–801 (2003).
Kassen, A. et al. Stromal cells of the human prostate: initial isolation and characterization. Prostate 28, 89–97 (1996).
Mazzocca, A. et al. Tumor-secreted lysophostatidic acid accelerates hepatocellular carcinoma progression by promoting differentiation of peritumoral fibroblasts in myofibroblasts. Hepatology 54, 920–930 (2011).
Welch, D.R. Technical considerations for studying cancer metastasis in vivo. Clin. Exp. Metastasis 15, 272–306 (1997).
Acknowledgements
The work of the authors is supported by US National Cancer Institute grants P01-CA80124, R01-CA115767, R01-CA85140, R01-CA126642 and T32-CA73479 (R.K.J.), R01-CA96915 (D.F.), R21-CA139168 and R01-CA159258 (D.G.D.) and Federal Share Proton Beam Program grants (R.K.J., D.F. and D.G.D.); Department of Defense Innovator Award W81XWH-10-1-0016 (R.K.J.) and Predoctoral Fellowship W81XWH-06-1-0781 (A.M.M.J.D.); American Cancer Society grant RSG-11-073-01TBG (D.G.D.); and Stichting Michael Van Vloten Fonds and the Stichting Jo Kolk (A.M.M.J.D.). We acknowledge the outstanding technical assistance of J. Kahn, S. Roberge and P. Huang with animal models.
Author information
Authors and Affiliations
Contributions
D.G.D., D.F. and R.K.J. designed the studies; A.M.M.J.D. and E.J.A.S. performed the experiments; D.G.D., D.F., A.M.M.J.D. and R.K.J. analyzed the data; and A.M.M.J.D., R.K.J. and D.G.D. edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Duyverman, A., Steller, E., Fukumura, D. et al. Studying primary tumor–associated fibroblast involvement in cancer metastasis in mice. Nat Protoc 7, 756–762 (2012). https://doi.org/10.1038/nprot.2012.031
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2012.031
This article is cited by
-
Extracellular vesicle-mediated transfer of miRNA-1 from primary tumors represses the growth of distant metastases
Experimental & Molecular Medicine (2024)
-
Cancer-associated fibroblasts as key regulators of the breast cancer tumor microenvironment
Cancer and Metastasis Reviews (2018)
-
An orthotopic xenograft model with survival hindlimb amputation allows investigation of the effect of tumor microenvironment on sarcoma metastasis
Clinical & Experimental Metastasis (2015)
-
An isolated tumor perfusion model in mice
Nature Protocols (2012)
-
A transient parabiosis skin transplantation model in mice
Nature Protocols (2012)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.