Abstract
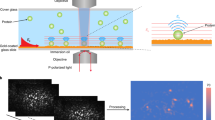
This protocol describes a single-molecule pull-down (SiMPull) assay for analyzing physiological protein complexes. The assay combines the conventional pull-down assay with single-molecule total internal reflection fluorescence (TIRF) microscopy and allows the probing of single macromolecular complexes directly from cell or tissue extracts. In this method, antibodies against the protein of interest are immobilized on a passivated microscope slide. When cell extracts are applied, the surface-tethered antibody captures the protein together with its physiological interaction partners. After washing away the unbound components, single-molecule fluorescence microscopy is used to probe the pulled-down proteins. Captured proteins are visualized through genetically encoded fluorescent protein tags or through antibody labeling. Compared with western blot analysis, this ultrasensitive assay requires considerably less time and reagents and provides quantitative data. Furthermore, SiMPull can distinguish between multiple association states of the same protein. SiMPull is generally applicable to proteins from a variety of cellular contexts and to endogenous proteins. Starting with the cell extracts and passivated slides, the assay requires 1.5–2.5 h for data acquisition and analysis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Alberts, B. The cell as a collection of protein machines: preparing the next generation of molecular biologists. Cell 92, 291–294 (1998).
Barrios-Rodiles, M. et al. High-throughput mapping of a dynamic signaling network in mammalian cells. Science 307, 1621–1625 (2005).
Yamada, T. & Bork, P. Evolution of biomolecular networks: lessons from metabolic and protein interactions. Nat. Rev. Mol. Cell Biol. 10, 791–803 (2009).
Puig, O. et al. The tandem affinity purification (TAP) method: a general procedure of protein complex purification. Methods 24, 218–229 (2001).
Gingras, A.C., Gstaiger, M., Raught, B. & Aebersold, R. Analysis of protein complexes using mass spectrometry. Nat. Rev. Mol. Cell Biol. 8, 645–654 (2007).
Fields, S. & Song, O. A novel genetic system to detect protein-protein interactions. Nature 340, 245–246 (1989).
Kerppola, T.K. Bimolecular fluorescence complementation (BiFC) analysis as a probe of protein interactions in living cells. Annu. Rev. Biophys. 37, 465–487 (2008).
Reyes-Lamothe, R., Sherratt, D.J. & Leake, M.C. Stoichiometry and architecture of active DNA replication machinery in Escherichia coli. Science 328, 498–501 (2010).
Li, H., Zhou, D., Browne, H., Balasubramanian, S. & Klenerman, D. Molecule by molecule direct and quantitative counting of antibody-protein complexes in solution. Anal. Chem. 76, 4446–4451 (2004).
Taguchi, H., Ueno, T., Tadakuma, H., Yoshida, M. & Funatsu, T. Single-molecule observation of protein-protein interactions in the chaperonin system. Nat. Biotechnol. 19, 861–865 (2001).
Kapanidis, A.N. et al. Fluorescence-aided molecule sorting: analysis of structure and interactions by alternating-laser excitation of single molecules. Proc. Natl. Acad. Sci. USA 101, 8936–8941 (2004).
Blosser, T.R., Yang, J.G., Stone, M.D., Narlikar, G.J. & Zhuang, X. Dynamics of nucleosome remodelling by individual ACF complexes. Nature 462, 1022–1027 (2009).
Jain, A. et al. Probing cellular protein complexes using single-molecule pull-down. Nature 473, 484–488 (2011).
Ulbrich, M.H. & Isacoff, E.Y. Subunit counting in membrane-bound proteins. Nat. Methods 4, 319–321 (2007).
Hoskins, A.A. et al. Ordered and dynamic assembly of single spliceosomes. Science 331, 1289–1295 (2011).
Yeom, K.H. et al. Single-molecule approach to immunoprecipitated protein complexes: insights into miRNA uridylation. EMBO Rep. 12, 690–696 (2011).
Roy, R., Hohng, S. & Ha, T. A practical guide to single-molecule FRET. Nat. Methods 5, 507–516 (2008).
Fleminger, G., Solomon, B., Wolf, T. & Hadas, E. Effect of polyethylene glycol on the non-specific adsorption of proteins to Eupergit C and agarose. J. Chromatogr. 510, 271–279 (1990).
Joo, C., Balci, H., Ishitsuka, Y., Buranachai, C. & Ha, T. Advances in single-molecule fluorescence methods for molecular biology. Annu. Rev. Biochem. 77, 51–76 (2008).
Rothenberg, E. & Ha, T. Single-molecule FRET analysis of helicase functions. Methods Mol. Biol. 587, 29–43 (2010).
Selvin, P.R. & Ha, T. Single-Molecule Techniques: A Laboratory Manual (Cold Spring Harbor Laboratory Press, 2008).
Acknowledgements
We thank B. Ramani, Y. Ishitsuka and K. Ragunathan for help with developing the protocol. This work was funded by US National Institutes of Health grants (AI083025, GM065367 to T.H.; HL082846 to Y.K.X.). Additional support was provided by National Science Foundation grants (0646550, 0822613 to T.H.). T.H. is an investigator with the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Contributions
A.J., Y.K.X. and T.H. designed the research. R.L. prepared the cell extracts. A.J. conducted single-molecule experiments and analyzed the data. A.J., Y.K.X. and T.H. wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Methods
Immunoprecipitation of endogenous proteins from mouse tissues. (DOC 61 kb)
Rights and permissions
About this article
Cite this article
Jain, A., Liu, R., Xiang, Y. et al. Single-molecule pull-down for studying protein interactions. Nat Protoc 7, 445–452 (2012). https://doi.org/10.1038/nprot.2011.452
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2011.452
This article is cited by
-
Mechanism of sensitivity modulation in the calcium-sensing receptor via electrostatic tuning
Nature Communications (2022)
-
Constructing a cost-efficient, high-throughput and high-quality single-molecule localization microscope for super-resolution imaging
Nature Protocols (2022)
-
Single-molecule analysis of specificity and multivalency in binding of short linear substrate motifs to the APC/C
Nature Communications (2022)
-
AAA + ATPase Thorase inhibits mTOR signaling through the disassembly of the mTOR complex 1
Nature Communications (2022)
-
Ligand functionalization of titanium nanopattern enables the analysis of cell–ligand interactions by super-resolution microscopy
Nature Protocols (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.