Abstract
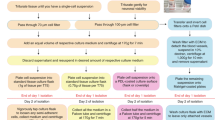
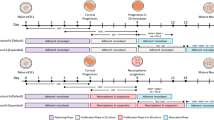
Here we present a protocol for extraction and culture of neurons from adult rat or mouse CNS. The method proscribes an optimized protease digestion of slices, control of osmolarity and pH outside the incubator with Hibernate and density gradient separation of neurons from debris. This protocol produces yields of millions of cortical, hippocampal neurons or neurosphere progenitors from each brain. The entire process of neuron isolation and culture takes less than 4 h. With suitable growth factors, adult neuron regeneration of axons and dendrites in culture proceeds over 1–3 weeks to allow controlled studies in pharmacology, electrophysiology, development, regeneration and neurotoxicology. Adult neurospheres can be collected in 1 week as a source of neuroprogenitors ethically preferred over embryonic or fetal sources. This protocol emphasizes two differences between neuron differentiation and neurosphere proliferation: adhesion dependence and the differentiating power of retinyl acetate.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Banker, G.A. & Cowan, W.M. Rat hippocampal neurons in dispersed cell culture. Brain Res. 126, 397–425 (1977).
Brewer, G.J., Torricelli, J.R., Evege, E.K. & Price, P.J. Optimized survival of hippocampal neurons in B27-supplemented Neurobasal, a new serum-free medium combination. J. Neurosci. Res. 35, 567–576 (1993).
Ahlemeyer, B. & Baumgart-Vogt, E. Optimized protocols for the simultaneous preparation of primary neuronal cultures of the neocortex, hippocampus and cerebellum from individual newborn (P0.5) C57Bl/6J mice. J. Neurosci. Methods 149, 110–120 (2005).
Brewer, G.J. Isolation and culture of adult rat hippocampal neurons. J. Neurosci. Methods 71, 143–155 (1997).
Brewer, G.J. Age-related toxicity to lactate, glutamate, and beta-amyloid in cultured adult neurons. Neurobiol. Aging 19, 561–568 (1998).
Brewer, G.J., Reichensperger, J.D. & Brinton, R.D. Prevention of age-related dysregulation of calcium dynamics by estrogen in neurons. Neurobiol. Aging 27, 306–317 (2006).
Nathan, B.P. et al. Apolipoprotein E4 inhibits, and apolipoprotein E3 promotes neurite outgrowth in cultured adult mouse cortical neurons through the low-density lipoprotein receptor-related protein. Brain Res. 928, 96–105 (2002).
Parihar, M.S. & Brewer, G.J. Simultaneous age-related depolarization of mitochondrial membrane potential and increased mitochondrial ROS production correlate with age-related glutamate excitotoxicity in rat hippocampal neurons. J Neurosci. Res. 85, 1018–1032 (2007).
Struble, R.G., Nathan, B.P., Cady, C., Cheng, X.Y. & McAsey, M. Estradiol regulation of astroglia and apolipoprotein E: an important role in neuronal regeneration. Exp. Gerontol. 42, 54–63 (2007).
Zhou, L.P. et al. Neuroprotection by estradiol: a role of aromatase against spine synapse loss after blockade of GABA(A) receptors. Exp. Neurol. 203, 72–81 (2007).
Yip, P.K. et al. Lentiviral vector expressing retinoic acid receptor beta 2 promotes recovery of function after corticospinal tract injury in the adult rat spinal cord. Hum. Mol. Genet. 15, 3107–3118 (2006).
Tyler, W.J. et al. BDNF increases release probability and the size of a rapidly recycling vesicle pool within rat hippocampal excitatory synapses. J. Physiol. 574, 787–803 (2006).
Hurst, R.S. et al. A novel positive allosteric modulator of the alpha 7 neuronal nicotinic acetylcholine receptor: in vitro and in vivo characterization. J. Neurosci. 25, 4396–4405 (2005).
Brewer, G.J. & Price, P.J. Viable cultured neurons in ambient carbon dioxide and hibernation storage for a month. NeuroReport 7, 1509–1512 (1996).
Brewer, G.J. et al. Culture and regeneration of human neurons after brain surgery. J. Neurosci. Methods 107, 15–23 (2001).
Xie, C., Markesbery, W.R. & Lovell, M.A. Survival of hippocampal and cortical neurons in a mixture of MEM+ and B27-supplemented neurobasal medium. Free Radic. Biol. Med. 28, 665–672 (2000).
Viel, J.J., McManus, D.Q., Smith, S.S. & Brewer, G.J. Age- and concentration-dependent neuroprotection and toxicity by TNF in cortical neurons from beta-amyloid. J. Neurosci. Res. 64, 454–465 (2001).
Eide, L. & McMurray, C.T. Culture of adult mouse neurons. BioTechniques 38, 99–104 (2005).
Kivell, B.M., McDonald, F.J. & Miller, J.H. Serum-free culture of rat post-natal and fetal brainstem neurons. Brain Res. Dev. Brain Res. 120, 199–210 (2000).
Zhang, W., Hu, Y., Newman, E.A. & Mulholland, M.W. Serum-free culture of rat postnatal neurons derived from the dorsal motor nucleus of the vagus. J. Neurosci. Methods 150, 1–7 (2006).
Price, P.J. & Brewer, G.J. Protocols for Neural Cell Culture, 3rd edn. (eds. Fedoroff, S. & Richardson, A.) 255–264 (Humana Press, Inc., Totowa, New Jersey, 2000).
Gage, F.H. et al. Survival and differentiation of adult neuronal progenitor cells transplanted to the adult brain. Proc. Natl. Acad. Sci. USA 92, 11879–11883 (1995).
Caldwell, M.A. et al. Growth factors regulate the survival and fate of cells derived from human neurospheres. Nat. Biotechnol. 19, 475–479 (2001).
Tatebayashi, Y., Iqbal, K. & Grundke-Iqbal, I. Dynamic regulation of expression and phosphorylation of tau by fibroblast growth factor-2 in neural progenitor cells from adult rat hippocampus. J. Neurosci. 19, 5245–5254 (1999).
Alexanian, A.R. & Nornes, H.O. Proliferation and regeneration of retrogradely labeled adult rat corticospinal neurons in culture. Exp. Neurol. 170, 277–282 (2001).
Palmer, T.D., Ray, J. & Gage, F.H. FGF-2—responsive neuronal progenitors reside in proliferation and quiescent regions of the adult rodent brain. Mol. Cell. Neurosci. 6, 474–486 (1995).
Zheng, T. et al. Transplantation of multipotent astrocytic stem cells into a rat model of neonatal hypoxic-ischemic encephalopathy. Brain Res. 1112, 99–105 (2006).
Shetty, A.K. & Turner, D.A. In vitro survival and differentiation of neurons derived from epidermal growth factor-responsive postnatal hippocampal stem cells: inducing effects of brain-derived neurotrophic factor. J. Neurobiol. 35, 395–425 (1998).
Gobbel, G.T., Choi, S.J., Beier, S. & Niranjan, A. Long-term cultivation of multipotential neural stem cells from adult rat subependyma. Brain Res. 980, 221–232 (2003).
Evans, J. et al. Characterization of mitotic neurons derived from adult rat hypothalamus and brain stem. J. Neurophysiol. 87, 1076–1085 (2002).
Brewer, G.J. & Cotman, C.W. Survival and growth of hippocampal neurons in defined medium at low density: advantages of a sandwich culture technique or low oxygen. Brain Res. 494, 65–74 (1989).
Kaplan, F.S. et al. Enhanced survival of rat neonatal cerebral cortical neurons at subatmospheric oxygen tensions in vitro . Brain Res. 384, 199–203 (1986).
Studer, L. et al. Enhanced proliferation, survival, and dopaminergic differentiation of CNS precursors in lowered oxygen. J. Neurosci. 20, 7377–7383 (2000).
Haynes, L.W. (ed.) The Neuron in Tissue Culture (John Wiley & Sons, New York, 1999).
Banker, G. & Goslin, K. (eds.) Culturing Nerve Cells 2nd edn. (The Bradford Book, MIT Press, Cambridge, Massachusetts, 1998).
Meberg, P.J. & Miller, M.W. Culturing hippocampal and cortical neurons. Methods Cell Biol. 71, 111–127 (2003).
Brewer, G.J., Lim, A., Capps, N.G. & Torricelli, J.R. Age-related calcium changes, oxyradical damage, caspase activation and nuclear condensation in hippocampal neurons in response to glutamate and beta-amyloid. Exp. Gerontol. 40, 426–437 (2005).
Evans, M.S., Collings, M.A. & Brewer, G.J. Electrophysiology of embryonic, adult and aged rat hippocampal neurons in serum-free culture. J. Neurosci. Methods 79, 37–46 (1998).
Patel, J.R. & Brewer, G.J. Age-related changes in neuronal glucose uptake in response to glutamate and beta-amyloid. J. Neurosci. Res. 72, 527–536 (2003).
Oddo, S. et al. Triple-transgenic model of Alzheimer's disease with plaques and tangles: intracellular Abeta and synaptic dysfunction. Neuron 39, 409–421 (2003).
Davis, A.A. & Temple, S. A self-renewing multipotential stem cell in the embryonic rat cerebral cortex. Nature 372, 263–266 (1994).
Temple, S. & Alvarez-Buylla, A. Stem cells in the adult mammalian central nervous system. Curr. Opin. Neurobiol. 9, 135–141 (1999).
Ostenfeld, T. et al. Regional specification of rodent and human neurospheres. Brain Res. Dev. Brain Res. 134, 43–55 (2002).
Mayer-Proschel, M., Kalyani, A.J., Mujtaba, T. & Rao, M.S. Isolation of lineage-restricted neuronal precursors from multipotent neuroepithelial stem cells. Neuron 19, 773–785 (1997).
Wohl, C.A. & Weiss, S. Retinoic acid enhances neuronal proliferation and astroglial differentiation in cultures of CNS stem cell-derived precursors. J. Neurobiol. 37, 281–290 (1998).
Takahashi, J., Palmer, T.D. & Gage, F.H. Retinoic acid and neurotrophins collaborate to regulate neurogenesis in adult-derived neural stem cell cultures. J. Neurobiol. 38, 65–81 (1999).
Laywell, E.D. et al. Neuron-to-astrocyte transition: Phenotypic fluidity and the formation of hybrid asterons in differentiating neurospheres. J. Comp. Neurol. 493, 321–333 (2005).
Acknowledgements
This work was supported by a Temple Foundation award from the Alzheimer Association and the National Institute on Aging.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Southern Illinois University receives royalties from Invitrogen for Neurobasal™ and B27™ products. In partnership with his wife, Brewer is the founder and owner of BrainBits LLC, maker of Hibernate™.
Rights and permissions
About this article
Cite this article
Brewer, G., Torricelli, J. Isolation and culture of adult neurons and neurospheres. Nat Protoc 2, 1490–1498 (2007). https://doi.org/10.1038/nprot.2007.207
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2007.207
This article is cited by
-
Disposable facemask waste combustion emits neuroactive smoke particulate matter
Scientific Reports (2023)
-
Aerobic glycolysis is the predominant means of glucose metabolism in neuronal somata, which protects against oxidative damage
Nature Neuroscience (2023)
-
Genome-wide pooled CRISPR screening in neurospheres
Nature Protocols (2023)
-
Culture of cerebrospinal fluid-contacting neurons from neonatal mouse spinal cord
Cell and Tissue Banking (2023)
-
LONRF2 is a protein quality control ubiquitin ligase whose deficiency causes late-onset neurological deficits
Nature Aging (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.