Abstract
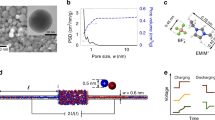
A single nanopore structure that embeds all components of an electrochemical storage device could bring about the ultimate miniaturization in energy storage. Self-alignment of electrodes within each nanopore may enable closer and more controlled spacing between electrodes than in state-of-art batteries. Such an ‘all-in-one’ nanopore battery array would also present an alternative to interdigitated electrode structures that employ complex three-dimensional geometries with greater spatial heterogeneity. Here, we report a battery composed of an array of nanobatteries connected in parallel, each composed of an anode, a cathode and a liquid electrolyte confined within the nanopores of anodic aluminium oxide, as an all-in-one nanosize device. Each nanoelectrode includes an outer Ru nanotube current collector and an inner nanotube of V2O5 storage material, forming a symmetric full nanopore storage cell with anode and cathode separated by an electrolyte region. The V2O5 is prelithiated at one end to serve as the anode, with pristine V2O5 at the other end serving as the cathode, forming a battery that is asymmetrically cycled between 0.2 V and 1.8 V. The capacity retention of this full cell (relative to 1 C values) is 95% at 5 C and 46% at 150 C, with a 1,000-cycle life. From a fundamental point of view, our all-in-one nanopore battery array unveils an electrochemical regime in which ion insertion and surface charge mechanisms for energy storage become indistinguishable, and offers a testbed for studying ion transport limits in dense nanostructured electrode arrays.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Rhodes, C., Long, J., Pettigrew, K., Stroud, R. & Rolison, D. Architectural integration of the components necessary for electrical energy storage on the nanoscale and in 3D. Nanoscale 3, 1731–1740 (2011).
Long, J., Dunn, B., Rolison, D. & White, H. Three-dimensional battery architectures. Chem. Rev. 104, 4463–4492 (2004).
Sun, K. et al. 3D printing of interdigitated Li-ion microbattery architectures. Adv. Mater. 4539–4543 (2013).
Pikul, J. H., Zhang, H. G., Cho, J., Braun, P. V. & King, W. P. High-power lithium ion microbatteries from interdigitated three-dimensional bicontinuous nanoporous electrodes. Nature Commun. 4, 1732 (2013).
Gowda, S. R., Reddy, A. L. L. M., Zhan, X. & Ajayan, P. M. Building energy storage device on a single nanowire. Nano Lett. 11, 3329–3333 (2011).
Banerjee, P., Perez, I., Henn-Lecordier, L., Lee, S. B. & Rubloff, G. W. Nanotubular metal–insulator–metal capacitor arrays for energy storage. Nature Nanotech. 4, 292–296 (2009).
Haspert, L. C., Lee, S. B. & Rubloff, G. W. Nano-engineering strategies for metal–insulator–metal electrostatic nanocapacitors. ACS Nano 6, 3528–3536 (2012).
Haspert, L. C., Gillette, E., Lee, S. B. & Rubloff, G. W. Perspective: hybrid systems combining electrostatic and electrochemical nanostructures for ultrahigh power energy storage. Energy Environ. Sci. 6, 2578–2590 (2013).
Ruzmetov, D. et al. Electrolyte stability determines scaling limits for solid-state 3D Li ion batteries. Nano Lett. 12, 505–511 (2011).
Vahur, Z. & Daniel, B. Modelling polymer electrolytes for 3D-microbatteries using finite element analysis. Electrochim. Acta 57, 237–243 (2011).
Arthur, T. S. et al. Three-dimensional electrodes and battery architectures. MRS Bull. 36, 523–531 (2011).
Gerasopoulos, K. et al. Hierarchical three-dimensional microbattery electrodes combining bottom-up self-assembly and top-down micromachining. ACS Nano 6, 6422–6432 (2012).
Gnerlich, M. et al. Solid flexible electrochemical supercapacitor using Tobacco mosaic virus nanostructures and ALD ruthenium oxide. J. Micromech. Microeng. 23, 114014 (2013).
White, R. & White, H. Electrochemistry in nanometer-wide electrochemical cells. Langmuir 24, 2850–2855 (2008).
Matt, K. P., Revati, K., Henry, S. W. & Gregory, A. V. A computationally efficient treatment of polarizable electrochemical cells held at a constant potential. J. Chem. Phys. C 116, 4903–4912 (2012).
Ryan, W. H., Henry, S. W., Bruce, D. & Debra, R. R. 3-D microbatteries. Electrochem Commun. 5, 120–123 (2003).
US Department of Energy Basic Energy Sciences Advisory Committee Mesoscale Science Committee W. From Quanta to the Continuum: Opportunities for Mesoscale Science (Government Printing Office, 2012).
Gregorczyk, K., Banerjee, P. & Rubloff, G. W. Conduction in ultrathin ruthenium electrodes prepared by atomic layer deposition. Mater. Lett. 73, 43–46 (2012).
Chen, X. et al. Ozone-based atomic layer deposition of crystalline V2O5 films for high performance electrochemical energy storage. Chem. Mater. 24, 1255–1261 (2012).
Lu, Z. et al. Basic electroanalytical characterization of lithium insertion into thin, well-crystallized V2O5 films. J. Electroanal. Chem. 491, 211–221 (2000).
Zhou, C. F., Kumar, S., Doyle, C. D. & Tour, J. M. Functionalized single wall carbon nanotubes treated with pyrrole for electrochemical supercapacitor membranes. Chem. Mater. 17, 1997–2002 (2005).
Wang, S. et al. Porous monodisperse V2O5 microspheres as cathode materials for lithium-ion batteries. J. Mater. Chem. 21, 6365–6369 (2011).
Liu, Y. et al. V2O5 nano-electrodes with high power and energy densities for thin film Li-ion batteries. Adv. Energy Mater. 1, 194–202 (2011).
Ardizzone, S., Fregonara, G. & Trasatti, S. ‘Inner’ and ‘outer’ active surface of RuO2 electrodes. Electrochim. Acta 35, 263–267 (1990).
Harris, S. J., Timmons, A., Baker, D. R. & Monroe, C. Direct in situ measurements of Li transport in Li-ion battery negative electrodes. Chem. Phys. Lett. 485, 265–274 (2010).
Harris, S. J. & Lu, P. Effects of inhomogeneities—nanoscale to mesoscale—on the durability of Li-ion batteries. J. Phys. Chem. C 117, 6481–6492 (2013).
Cleveland, E. R., Banerjee, P., Perez, I., Lee, S. B. & Rubloff, G. W. Profile evolution for conformal atomic layer deposition over nanotopography. ACS Nano 4, 4637–4644 (2010).
Karki, K. et al. Hoop-strong nanotubes for battery electrodes. ACS Nano 7, 8295–8302 (2013).
Lei, W. et al. Real-time observation and optimization of tungsten atomic layer deposition process cycle. J. Vac. Sci. Technol. B 24, 780–789 (2006).
Grubbs, R. K., Steinmetz, N. J. & George, S. M. Gas phase reaction products during tungsten atomic layer deposition using WF6 and Si2H6 . J. Vac. Sci. Technol. B 22, 1811–1821 (2004).
Kim, W-H. et al. Atomic layer deposition of Ni thin films and application to area-selective deposition. J. Electrochem. Soc. 158, D1–D5 (2011).
Gregorczyk, K., Henn-Lecordier, L., Gatineau, J., Dussarrat, C. & Rubloff, G. Atomic layer deposition of ruthenium using the novel precursor bis(2,6,6-trimethyl-cyclohexadienyl)ruthenium. Chem. Mater. 23, 2650–2656 (2011).
Acknowledgements
This work was supported by Nanostructures for Electrical Energy Storage (NEES), an Energy Frontier Research Center funded by the US Department of Energy, Office of Science, Office of Basic Energy Sciences (award no. DESC0001160). The authors acknowledge support from the Maryland NanoCenter and the NispLab, and thank C. Wang for providing electrolyte, M. Noked, Y. Xu and C. Sun for discussions regarding electrochemistry and J. Provine for valuable suggestions for noble-metal ALD.
Author information
Authors and Affiliations
Contributions
C.L., X.C., S.B.L. and G.W.R. conceived and designed the experiments. C.L. and X.C. performed the half-cell experiments. C.L. performed the full-cell experiment. E.I.G. conducted the COMSOL simulation. A.J.P. carried out XPS characterization. K.E.G., A.C.K. and M.A.S. contributed material fabrication tools. C.L., E.I.G., S.B.L. and G.W.R. co-wrote the paper. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary Information (PDF 1951 kb)
Rights and permissions
About this article
Cite this article
Liu, C., Gillette, E., Chen, X. et al. An all-in-one nanopore battery array. Nature Nanotech 9, 1031–1039 (2014). https://doi.org/10.1038/nnano.2014.247
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nnano.2014.247
This article is cited by
-
Fabrication of metal-oxide arrays: mechanism of solvent-mediated metal infiltration into block copolymer nanopatterns
Clean Technologies and Environmental Policy (2023)
-
Well-defined nanostructuring with designable anodic aluminum oxide template
Nature Communications (2022)
-
Influence of different dopants and redox forms of PANI in its crystal structure, morphology, electrochemical energy storage to variable extent, unique properties and kinetics
Bulletin of Materials Science (2022)
-
Regional and functional division of functional elements of solid-state nanochannels for enhanced sensitivity and specificity of biosensing in complex matrices
Nature Protocols (2021)
-
Towards explicit regulating-ion-transport: nanochannels with only function-elements at outer-surface
Nature Communications (2021)