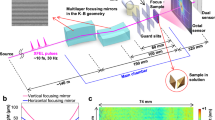
Abstract
Conventional Fourier-transform infrared (FTIR) microspectroscopic systems are limited by an inevitable trade-off between spatial resolution, acquisition time, signal-to-noise ratio (SNR) and sample coverage. We present an FTIR imaging approach that substantially extends current capabilities by combining multiple synchrotron beams with wide-field detection. This advance allows truly diffraction-limited high-resolution imaging over the entire mid-infrared spectrum with high chemical sensitivity and fast acquisition speed while maintaining high-quality SNR.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Wetzel, D.L. & LeVine, S.M. Science 285, 1224–1225 (1999).
Pezacki, J.P. et al. Nat. Chem. Biol. 7, 137–145 (2011).
Chalmers, J.M. & Griffiths, P.R. Handbook of Vibrational Spectroscopy (Wiley, 2002).
Stelzer, E.H.K. J. Microsc. 189, 15–24 (1998).
Lasch, P. & Naumann, D. Biochim. Biophys. Acta 1758, 814–829 (2006).
Carr, G.L. Rev. Sci. Instrum. 72, 1613–1619 (2001).
Dumas, P., Jamin, N., Teillaud, J.L., Miller, L.M. & Beccard, B. Faraday Discuss. 126, 289–302 (2004).
Bhargava, R. & Levin, I.W. Anal. Chem. 73, 5157–5167 (2001).
Lewis, E.N. et al. Anal. Chem. 67, 3377–3381 (1995).
Kidder, L.H., Levin, I.W., Lewis, E.N., Kleiman, V.D. & Heilweil, E.J. Opt. Lett. 22, 742–744 (1997).
Moss, D., Gasharova, B. & Mathis, Y. Infrared Phys. Technol. 49, 53–56 (2006).
Carr, G.L., Chubar, O. & Dumas, P. in Spectrochemical Analysis Using Infrared Multichannel Detectors 1st edn. (eds., Bhargava, R. & Levin, I.W.) 56–84 (Wiley-Blackwell, Oxford, 2005).
Miller, L.M. & Dumas, P. Biochim. Biophys. Acta 1758, 846–857 (2006).
Petibois, C., Cestelli-Guidi, M., Piccinini, M., Moenner, M. & Marcelli, A. Anal. Bioanal. Chem. 397, 2123–2129 (2010).
Fernandez, D.C., Bhargava, R., Hewitt, S.M. & Levin, I.W. Nat. Biotechnol. 23, 469–474 (2005).
Bhargava, R., Schwartz Perlman, R., Fernandez, D.C., Levin, I.W. & Bartick, E.G. Anal. Bioanal. Chem. 394, 2069–2075 (2009).
Li, Z.Q. et al. Nat. Phys. 4, 532–535 (2008).
Bunaciu, A.A., Aboul-Enein, H.Y. & Fleschin, S. Appl. Spectrosc. Rev. 45, 206–219 (2010).
Matveev, S. & Stachel, T. Geochim. Cosmochim. Acta 71, 5528–5543 (2007).
Prati, S., Joseph, E., Sciutto, G. & Mazzeo, R. Acc. Chem. Res. 43, 792–801 (2010).
Politi, Y., Arad, T., Klein, E., Weiner, S. & Addadi, L. Science 306, 1161–1164 (2004).
Martin, F.L. et al. Nat. Protoc. 5, 1748–1760 (2010).
Hazen, T.C. et al. Science 330, 204–208 (2010).
Walsh, M.J. et al. Stem Cells 26, 108–118 (2008).
Walsh, M.J. et al. Stem Cell Res. 3, 15–27 (2009).
Holman, H.N., Bechtel, H.A., Hao, Z. & Martin, M.C. Anal. Chem. 82, 8757–8765 (2010).
Kuzyk, A. et al. J. Biol. Chem. 285, 31202–31207 (2010).
Webster, G.T. et al. Anal. Chem. 81, 2516–2524 (2009).
Bhargava, R. Anal. Bioanal. Chem. 389, 1155–1169 (2007).
Centonze, V. & Pawley, J.B. in Handbook of Biological Confocal Microscopy 3rd edn. (ed. Pawley, J.B.) 627–649 (Springer, New York, 2006).
Murphy, D.B. Fundamentals of Light Microscopy and Electronic Imaging 1st edn. 233–258 (Wiley-Liss, New York, 2001).
Bobroff, N. Rev. Sci. Instrum. 57, 1152–1157 (1986).
Reffner, J.A., Martoglio, P.A. & Williams, G.P. Rev. Sci. Instrum. 66, 1298–1302 (1995).
Nasse, M.J., Reininger, R., Kubala, T., Janowski, S. & Hirschmugl, C. Nucl. Instrum. Methods Phys. Res. A 582, 107–110 (2007).
Park, K., Lee, J., Bhargava, R. & King, W.P. Anal. Chem. 80, 3221–3228 (2008).
Acknowledgements
We thank T. Kubala, S. Janowski and M. Fisher for their engineering work, and Z. El-Bayyari for his help during alignment of the beamline. This work was supported by the US National Science Foundation under awards CHE-0832298, CHE-0957849 and DMR-0619759, and by the Research Growth Initiative of the University of Wisconsin–Milwaukee. Part of this work is based on research conducted at the Synchrotron Radiation Center, University of Wisconsin–Madison, which is supported by the National Science Foundation under award DMR-0537588. The project described was also supported by award R01CA138882 from the US National Institutes of Health.
Author information
Authors and Affiliations
Contributions
M.J.N., R.R. and C.J.H. designed research; M.J.N., M.J.W. and E.C.M. performed research; M.J.W., A.K.-B., V.M. and R.B. contributed prostate samples; M.J.N., M.J.W., E.C.M., R.B. and C.J.H. analyzed data; and M.J.N., R.B. and C.J.H. wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
R.R. is an employee of Scientific Answers & Solutions and has received compensation for his scientific consultation related to this work.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7, Supplementary Table 1 and Supplementary Notes 1–3 (PDF 2100 kb)
Rights and permissions
About this article
Cite this article
Nasse, M., Walsh, M., Mattson, E. et al. High-resolution Fourier-transform infrared chemical imaging with multiple synchrotron beams. Nat Methods 8, 413–416 (2011). https://doi.org/10.1038/nmeth.1585
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth.1585
This article is cited by
-
Infrared spectroscopic laser scanning confocal microscopy for whole-slide chemical imaging
Nature Communications (2023)
-
Vibrational imaging for label-free cancer diagnosis and classification
La Rivista del Nuovo Cimento (2022)
-
Time lapse synchrotron IR chemical imaging for observing the acclimation of a single algal cell to CO2 treatment
Scientific Reports (2021)
-
Characterization of colorectal mucus using infrared spectroscopy: a potential target for bowel cancer screening and diagnosis
Laboratory Investigation (2020)
-
Origin of micro-scale heterogeneity in polymerisation of photo-activated resin composites
Nature Communications (2020)