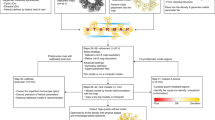
Abstract
We describe an algorithm for phasing protein crystal X-ray diffraction data that identifies, retrieves, refines and exploits general tertiary structural information from small fragments available in the Protein Data Bank. The algorithm successfully phased, through unspecific molecular replacement combined with density modification, all-helical, mixed alpha-beta, and all-beta protein structures. The method is available as a software implementation: Borges.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Change history
05 May 2014
In the version of this article initially published, the authors did not acknowledge all of the people involved in the generation of the crystallographic data set of AF1503 from A. fulgidus. These diffraction data were originated by M. Hulko, A. Ursinus, K. Bär, J. Martin, K.Z. and A.N. Lupas at the Max Planck Institute for Developmental Biology, Tübingen. M. Hulko, A. Ursinus, K. Bär, J. Martin and A.N. Lupas have kindly given their retroactive permission to use the data. Their report on the AF1503 structure was published in the Journal of Structural Biology (doi:10.1016/j.jsb.2014.02.008) and PDB 4CQ4. The error has been corrected in the HTML and PDF versions of the article.
References
Bernstein, F.C. et al. J. Mol. Biol. 112, 535–542 (1977).
Qian, B. et al. Nature 450, 259–264 (2007).
Huber, R. Acta Crystallogr. 19, 353–356 (1965).
Rossmann, M.G. The Molecular Replacement Method (Gordon and Breach, 1972).
DiMaio, F. et al. Nature 473, 540–543 (2011).
Rodríguez, D.D. et al. Nat. Methods 6, 651–653 (2009).
McCoy, A.J. et al. J. Appl. Crystallogr. 40, 658–674 (2007).
Sheldrick, G.M. Acta Crystallogr. D66, 479–485 (2010).
Tannenbaum, T., Wright, D., Miller, K. & Livny, M. in Beowulf Cluster Computing with Linux (ed. Sterling, T.) Ch. 14, 307–350 (The MIT Press, 2002).
Burla, M.C., Carrozzini, B., Cascarano, G.L., Giacovazzo, C. & Polidori, G. J. Appl. Crystallogr. 44, 1143–1151 (2011).
Miller, R. et al. Science 259, 1430–1433 (1993).
Joosten, R.P. et al. Nucleic Acids Res. 39, D411–D419 (2011).
Oldfield, T.J. Acta Crystallogr. D57, 1421–1427 (2001).
Cowtan, K. Acta Crystallogr. D68, 328–335 (2012).
Nicholls, R.A., Long, F. & Murshudov, G.N. Acta Crystallogr. D Biol. Crystallogr. 68, 404–417 (2012).
Linder, J.U. & Schultz, J.E. Methods Enzymol. 471, 115–123 (2010).
Brünger, A.T. Methods Enzymol. 276, 558–580 (1997).
Grosse-Kunstleve, R.W. & Adams, P.D. Acta Crystallogr. D57, 1390–1396 (2001).
Sampietro, J. et al. Mol. Cell 24, 293–300 (2006).
Hopcroft, J. & Tarjan, R. Commun. ACM 16, 372–378 (1973).
Kabsch, W. Acta Crystallogr. D Biol. Crystallogr. 66, 125–132 (2010).
Acknowledgements
This work was supported by the Spanish Ministerio de Ciencia e Innovación-Ministerio de Economia y Competitividad, Centro de Desarrollo Tecnológico Industrial and Consejo Superior de Investigaciones Científicas (grants BIO2009-10576; IDC-2010-1173; BFU2012-35367; BFU2012-32847; predoctoral grants to D.D.R., I.D.M. and I.M.d.I.; JdC to K.M.; RyC to R.M.B.); Generalitat de Catalunya (2009SGR-1036); VW-Stiftung Niedersachsenprofessur to G.M.S. We also acknowledge beam time on the Swiss Light Source beamline X10SA and computing time at the FCSCL. We thank M. Hulko, A. Ursinus, K. Bär, J. Martin and A.N. Lupas at the Max Planck Institute for Developmental Biology, Tübingen, for permission to use the diffraction data of AF1503 from A. fulgidus.
Author information
Authors and Affiliations
Contributions
All authors contributed extensively to the work presented in this paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1 and 2, Supplementary Tables 1 and 2, Supplementary Results and Supplementary Notes 1 and 2 (PDF 6443 kb)
Rights and permissions
About this article
Cite this article
Sammito, M., Millán, C., Rodríguez, D. et al. Exploiting tertiary structure through local folds for crystallographic phasing. Nat Methods 10, 1099–1101 (2013). https://doi.org/10.1038/nmeth.2644
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth.2644
This article is cited by
-
Ab initio phasing macromolecular structures using electron-counted MicroED data
Nature Methods (2022)
-
Structure and mechanism of a bacterial host-protein citrullinating virulence factor, Porphyromonas gingivalis peptidylarginine deiminase
Scientific Reports (2015)
-
Erratum: Corrigendum: Exploiting tertiary structure through local folds for crystallographic phasing
Nature Methods (2014)