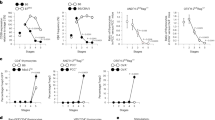
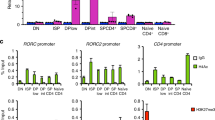
Abstract
Regulatory T cells (Treg cells) develop from progenitor thymocytes after the engagement of T cell antigen receptors (TCRs) with high-affinity ligands, but the underlying molecular mechanisms are still unclear. Here we show that the Nr4a nuclear receptors, which are encoded by immediate-early genes upregulated by TCR stimulation in thymocytes, have essential roles in Treg cell development. Mice that lacked all Nr4a factors could not produce Treg cells and died early owing to systemic autoimmunity. Nr4a receptors directly activated the promoter of the gene encoding the transcription factor Foxp3, and forced activation of Nr4a receptors bypassed low-strength TCR signaling to drive the Treg cell developmental program. Our results suggest that Nr4a receptors have key roles in determining CD4+ T cell fates in the thymus and thus contribute to immune homeostasis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Stritesky, G.L., Jameson, S.C. & Hogquist, K.A. Selection of self-reactive T cells in the thymus. Annu. Rev. Immunol. 30, 95–114 (2012).
Fontenot, J.D., Gavin, M.A. & Rudensky, A.Y. Foxp3 programs the development and function of CD4+CD25+ regulatory T cells. Nat. Immunol. 4, 330–336 (2003).
Hori, S., Nomura, T. & Sakaguchi, S. Control of regulatory T cell development by the transcription factor Foxp3. Science 299, 1057–1061 (2003).
Khattri, R., Cox, T., Yasayko, S.A. & Ramsdell, F. An essential role for Scurfin in CD4+CD25+ T regulatory cells. Nat. Immunol. 4, 337–342 (2003).
Bennett, C.L. et al. The immune dysregulation, polyendocrinopathy, enteropathy, X-linked syndrome (IPEX) is caused by mutations of FOXP3. Nat. Genet. 27, 20–21 (2001).
Sakaguchi, S. Naturally arising CD4+ regulatory t cells for immunologic self-tolerance and negative control of immune responses. Annu. Rev. Immunol. 22, 531–562 (2004).
Shevach, E.M. Regulatory T cells in autoimmmunity. Annu. Rev. Immunol. 18, 423–449 (2000).
Ruan, Q. et al. Development of Foxp3+ regulatory t cells is driven by the c-Rel enhanceosome. Immunity 31, 932–940 (2009).
Long, M., Park, S.G., Strickland, I., Hayden, M.S. & Ghosh, S. Nuclear factor-kappaB modulates regulatory T cell development by directly regulating expression of Foxp3 transcription factor. Immunity 31, 921–931 (2009).
Zheng, Y. et al. Role of conserved non-coding DNA elements in the Foxp3 gene in regulatory T-cell fate. Nature 463, 808–812 (2010).
Tone, Y. et al. Smad3 and NFAT cooperate to induce Foxp3 expression through its enhancer. Nat. Immunol. 9, 194–202 (2008).
Zorn, E. et al. IL-2 regulates FOXP3 expression in human CD4+CD25+ regulatory T cells through a STAT-dependent mechanism and induces the expansion of these cells in vivo. Blood 108, 1571–1579 (2006).
Kitoh, A. et al. Indispensable role of the Runx1-Cbfβ transcription complex for in vivo-suppressive function of FoxP3+ regulatory T cells. Immunity 31, 609–620 (2009).
Rudra, D. et al. Runx-CBFβ complexes control expression of the transcription factor Foxp3 in regulatory T cells. Nat. Immunol. 10, 1170–1177 (2009).
Bruno, L. et al. Runx proteins regulate Foxp3 expression. J. Exp. Med. 206, 2329–2337 (2009).
Ouyang, W. et al. Foxo proteins cooperatively control the differentiation of Foxp3+ regulatory T cells. Nat. Immunol. 11, 618–627 (2010).
Mouly, E. et al. The Ets-1 transcription factor controls the development and function of natural regulatory T cells. J. Exp. Med. 207, 2113–2125 (2010).
Chen, W. et al. Conversion of peripheral CD4+CD25− naive T cells to CD4+CD25+ regulatory T cells by TGF-β induction of transcription factor Foxp3. J. Exp. Med. 198, 1875–1886 (2003).
Wang, Z. et al. Structure and function of Nurr1 identifies a class of ligand-independent nuclear receptors. Nature 423, 555–560 (2003).
Cheng, L.E., Chan, F.K., Cado, D. & Winoto, A. Functional redundancy of the Nur77 and Nor-1 orphan steroid receptors in T-cell apoptosis. EMBO J. 16, 1865–1875 (1997).
Sekiya, T. et al. The nuclear orphan receptor Nr4a2 induces Foxp3 and regulates differentiation of CD4+ T cells. Nat. Commun. 2, 269 (2011).
Moran, A.E. et al. T cell receptor signal strength in Treg and iNKT cell development demonstrated by a novel fluorescent reporter mouse. J. Exp. Med. 208, 1279–1289 (2011).
Hill, J.A. et al. Foxp3 transcription-factor-dependent and -independent regulation of the regulatory T cell transcriptional signature. Immunity 27, 786–800 (2007).
Wei, G. et al. Global mapping of H3K4me3 and H3K27me3 reveals specificity and plasticity in lineage fate determination of differentiating CD4+ T cells. Immunity 30, 155–167 (2009).
Lin, W. et al. Regulatory T cell development in the absence of functional Foxp3. Nat. Immunol. 8, 359–368 (2007).
Baldwin, T.A. & Hogquist, K.A. Transcriptional analysis of clonal deletion in vivo. J. Immunol. 179, 837–844 (2007).
Fassett, M.S., Jiang, W., D'Alise, A.M., Mathis, D. & Benoist, C. Nuclear receptor Nr4a1 modulates both regulatory T-cell (Treg) differentiation and clonal deletion. Proc. Natl. Acad. Sci. USA 109, 3891–3896 (2012).
Zhou, T. et al. Inhibition of Nur77/Nurr1 leads to inefficient clonal deletion of self-reactive T cells. J. Exp. Med. 183, 1879–1892 (1996).
Calnan, B.J., Szychowski, S., Chan, F.K., Cado, D. & Winoto, A. A role for the orphan steroid receptor Nur77 in apoptosis accompanying antigen-induced negative selection. Immunity 3, 273–282 (1995).
Lyon, M.F., Peters, J., Glenister, P.H., Ball, S. & Wright, E. The scurfy mouse mutant has previously unrecognized hematological abnormalities and resembles Wiskott-Aldrich syndrome. Proc. Natl. Acad. Sci. USA 87, 2433–2437 (1990).
Godfrey, V.L., Wilkinson, J.E. & Russell, L.B. X-linked lymphoreticular disease in the scurfy (sf) mutant mouse. Am. J. Pathol. 138, 1379–1387 (1991).
Barnes, M.J. & Powrie, F. Regulatory T cells reinforce intestinal homeostasis. Immunity 31, 401–411 (2009).
Brocker, T., Riedinger, M. & Karjalainen, K. Targeted expression of major histocompatibility complex (MHC) class II molecules demonstrates that dendritic cells can induce negative but not positive selection of thymocytes in vivo. J. Exp. Med. 185, 541–550 (1997).
Kirberg, J., Berns, A. & von Boehmer, H. Peripheral T cell survival requires continual ligation of the T cell receptor to major histocompatibility complex-encoded molecules. J. Exp. Med. 186, 1269–1275 (1997).
Mullican, S.E. et al. Abrogation of nuclear receptors Nr4a3 and Nr4a1 leads to development of acute myeloid leukemia. Nat. Med. 13, 730–735 (2007).
Lio, C.W. & Hsieh, C.S. A two-step process for thymic regulatory T cell development. Immunity 28, 100–111 (2008).
Proietto, A.I. et al. Dendritic cells in the thymus contribute to T-regulatory cell induction. Proc. Natl. Acad. Sci. USA 105, 19869–19874 (2008).
Sirin, O., Lukov, G.L., Mao, R., Conneely, O.M. & Goodell, M.A. The orphan nuclear receptor Nurr1 restricts the proliferation of haematopoietic stem cells. Nat. Cell Biol. 12, 1213–1219 (2010).
Masuyama, N. et al. Akt inhibits the orphan nuclear receptor Nur77 and T-cell apoptosis. J. Biol. Chem. 276, 32799–32805 (2001).
Kanangat, S. et al. Disease in the scurfy (sf) mouse is associated with overexpression of cytokine genes. Eur. J. Immunol. 26, 161–165 (1996).
Lahl, K. et al. Nonfunctional regulatory T cells and defective control of Th2 cytokine production in natural scurfy mutant mice. J. Immunol. 183, 5662–5672 (2009).
Paul, W.E. & Zhu, J. How are TH2-type immune responses initiated and amplified? Nat. Rev. Immunol. 10, 225–235 (2010).
Kuang, A.A., Cado, D. & Winoto, A. Nur77 transcription activity correlates with its apoptotic function in vivo. Eur. J. Immunol. 29, 3722–3728 (1999).
Koonpaew, S., Shen, S., Flowers, L. & Zhang, W. LAT-mediated signaling in CD4+CD25+ regulatory T cell development. J. Exp. Med. 203, 119–129 (2006).
Wan, Y.Y., Chi, H., Xie, M., Schneider, M.D. & Flavell, R.A. The kinase TAK1 integrates antigen and cytokine receptor signaling for T cell development, survival and function. Nat. Immunol. 7, 851–858 (2006).
Medoff, B.D. et al. Differential requirement for CARMA1 in agonist-selected T-cell development. Eur. J. Immunol. 39, 78–84 (2009).
Schmidt-Supprian, M. et al. Differential dependence of CD4+CD25+ regulatory and natural killer-like T cells on signals leading to NF-κB activation. Proc. Natl. Acad. Sci. USA 101, 4566–4571 (2004).
Schmidt-Supprian, M. et al. Mature T cells depend on signaling through the IKK complex. Immunity 19, 377–389 (2003).
Gupta, S. et al. Differential requirement of PKC-θ in the development and function of natural regulatory T cells. Mol. Immunol. 46, 213–224 (2008).
Kadkhodaei, B. et al. Nurr1 is required for maintenance of maturing and adult midbrain dopamine neurons. J. Neurosci. 29, 15923–15932 (2009).
Acknowledgements
We thank N. Shimizu, S. Tsuruta, N. Shiino, Y. Noguchi and M. Asakawa for technical assistance, and Y. Ushijima for manuscript preparation. Foxp3-hCD2-hCD52-KI mice were originally from the laboratory of S. Hori (Research Center for Allergy and Immunology, RIKEN), and the Nr4a1fl/fl and Nr4a2fl/fl mice were from H. Ichinose (Tokyo Institute of Technology). Supported by the Ministry of Education, Culture, Sports, Science and Technology of Japan, the Japan Society of the Promotion of Science, the Takeda Science Foundation, the Uehara Memorial Foundation, Mochida Memorial Foundation and the SENSHIN Medical Research Foundation.
Author information
Authors and Affiliations
Contributions
T.S. and A.Y. designed the research and analyzed data; T.S., I.K., R.Y., T.F., A.K. and R.M. did the experiments; H.I., D.M. and P.C. provided the Nr4a2fl and Nr4a1fl mouse strain, plasmids and feedback on the manuscript; and T.S. and A.Y. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 (PDF 591 kb)
Rights and permissions
About this article
Cite this article
Sekiya, T., Kashiwagi, I., Yoshida, R. et al. Nr4a receptors are essential for thymic regulatory T cell development and immune homeostasis. Nat Immunol 14, 230–237 (2013). https://doi.org/10.1038/ni.2520
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.2520
This article is cited by
-
Potential Roles of Nr4a3-Mediated Inflammation in Immunological and Neurological Diseases
Molecular Neurobiology (2024)
-
Recently activated CD4 T cells in tuberculosis express OX40 as a target for host-directed immunotherapy
Nature Communications (2023)
-
A role of NR4A2 in Graves’ disease: regulation of Th17/Treg
Endocrine (2023)
-
Regulation of effector and memory CD8 + T cell differentiation: a focus on orphan nuclear receptor NR4A family, transcription factor, and metabolism
Immunologic Research (2023)
-
Exhaustion of CAR T cells: potential causes and solutions
Journal of Translational Medicine (2022)