Abstract
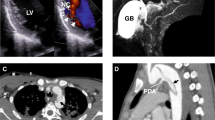
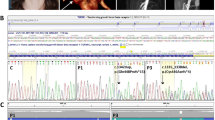
Elevated transforming growth factor (TGF)-β signaling has been implicated in the pathogenesis of syndromic presentations of aortic aneurysm, including Marfan syndrome (MFS) and Loeys-Dietz syndrome (LDS)1,2,3,4. However, the location and character of many of the causal mutations in LDS intuitively imply diminished TGF-β signaling5. Taken together, these data have engendered controversy regarding the specific role of TGF-β in disease pathogenesis. Shprintzen-Goldberg syndrome (SGS) has considerable phenotypic overlap with MFS and LDS, including aortic aneurysm6,7,8. We identified causative variation in ten individuals with SGS in the proto-oncogene SKI, a known repressor of TGF-β activity9,10. Cultured dermal fibroblasts from affected individuals showed enhanced activation of TGF-β signaling cascades and higher expression of TGF-β–responsive genes relative to control cells. Morpholino-induced silencing of SKI paralogs in zebrafish recapitulated abnormalities seen in humans with SGS. These data support the conclusions that increased TGF-β signaling is the mechanism underlying SGS and that high signaling contributes to multiple syndromic presentations of aortic aneurysm.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Dietz, H.C. et al. Marfan syndrome caused by a recurrent de novo missense mutation in the fibrillin gene. Nature 352, 337–339 (1991).
Loeys, B.L. et al. A syndrome of altered cardiovascular, craniofacial, neurocognitive and skeletal development caused by mutations in TGFBR1 or TGFBR2. Nat. Genet. 37, 275–281 (2005).
Habashi, J.P. et al. Losartan, an AT1 antagonist, prevents aortic aneurysm in a mouse model of Marfan syndrome. Science 312, 117–121 (2006).
Holm, T.M. et al. Noncanonical TGFβ signaling contributes to aortic aneurysm progression in Marfan syndrome mice. Science 332, 358–361 (2011).
Loeys, B.L. et al. Aneurysm syndromes caused by mutations in the TGF-β receptor. N. Engl. J. Med. 355, 788–798 (2006).
Shprintzen, R.J. & Goldberg, R.B. A recurrent pattern syndrome of craniosynostosis associated with arachnodactyly and abdominal hernias. J. Craniofac. Genet. Dev. Biol. 2, 65–74 (1982).
Greally, M.T. et al. Shprintzen-Goldberg syndrome: a clinical analysis. Am. J. Med. Genet. 76, 202–212 (1998).
Robinson, P.N. et al. Shprintzen-Goldberg syndrome: fourteen new patients and a clinical analysis. Am. J. Med. Genet. A 135, 251–262 (2005).
Akiyoshi, S. et al. c-Ski acts as a transcriptional co-repressor in transforming growth factor-β signaling through interaction with smads. J. Biol. Chem. 274, 35269–35277 (1999).
Luo, K. et al. The Ski oncoprotein interacts with the Smad proteins to repress TGFβ signaling. Genes Dev. 13, 2196–2206 (1999).
Massagué, J., Blain, S.W. & Lo, R.S. TGFβ signaling in growth control, cancer, and heritable disorders. Cell 103, 295–309 (2000).
Wotton, D. & Massagué, J. Smad transcriptional corepressors in TGF β family signaling. Curr. Top. Microbiol. Immunol. 254, 145–164 (2001).
Prunier, C. et al. The oncoprotein Ski acts as an antagonist of transforming growth factor-β signaling by suppressing Smad2 phosphorylation. J. Biol. Chem. 278, 26249–26257 (2003).
Reed, J.A. et al. Cytoplasmic localization of the oncogenic protein Ski in human cutaneous melanomas in vivo: functional implications for transforming growth factor β signaling. Cancer Res. 61, 8074–8078 (2001).
Nomura, T. et al. Ski is a component of the histone deacetylase complex required for transcriptional repression by Mad and thyroid hormone receptor. Genes Dev. 13, 412–423 (1999).
Derynck, R. & Zhang, Y.E. Smad-dependent and Smad-independent pathways in TGF-β family signalling. Nature 425, 577–584 (2003).
Pearson, G. et al. Mitogen-activated protein (MAP) kinase pathways: regulation and physiological functions. Endocr. Rev. 22, 153–183 (2001).
Neptune, E.R. et al. Dysregulation of TGF-β activation contributes to pathogenesis in Marfan syndrome. Nat. Genet. 33, 407–411 (2003).
Ng, C.M. et al. TGF-β–dependent pathogenesis of mitral valve prolapse in a mouse model of Marfan syndrome. J. Clin. Invest. 114, 1586–1592 (2004).
Cohn, R.D. et al. Angiotensin II type 1 receptor blockade attenuates TGF-β–induced failure of muscle regeneration in multiple myopathic states. Nat. Med. 13, 204–210 (2007).
Mizuguchi, T. et al. Heterozygous TGFBR2 mutations in Marfan syndrome. Nat. Genet. 36, 855–860 (2004).
van de Laar, I.M. et al. Mutations in SMAD3 cause a syndromic form of aortic aneurysms and dissections with early-onset osteoarthritis. Nat. Genet. 43, 121–126 (2011).
Lindsay, M.E. et al. Loss-of-function mutations in TGFB2 cause a syndromic presentation of thoracic aortic aneurysm. Nat. Genet. 44, 922–927 (2012).
Adzhubei, I.A. et al. A method and server for predicting damaging missense mutations. Nat. Methods 7, 248–249 (2010).
Ng, P.C. & Henikoff, S. SIFT: predicting amino acid changes that affect protein function. Nucleic Acids Res. 31, 3812–3814 (2003).
Nomura, N. et al. Isolation of human cDNA clones of ski and the ski-related gene, sno. Nucleic Acids Res. 17, 5489–5500 (1989).
Qin, B.Y., Lam, S.S., Correia, J.J. & Lin, K. Smad3 allostery links TGF-β receptor kinase activation to transcriptional control. Genes Dev. 16, 1950–1963 (2002).
Dahl, R., Wani, B. & Hayman, M.J. The Ski oncoprotein interacts with Skip, the human homolog of Drosophila Bx42. Oncogene 16, 1579–1586 (1998).
Chen, W. et al. Competition between Ski and CREB-binding protein for binding to Smad proteins in transforming growth factor-β signaling. J. Biol. Chem. 282, 11365–11376 (2007).
Wilson, J.J., Malakhova, M., Zhang, R., Joachimiak, A. & Hegde, R.S. Crystal structure of the dachshund homology domain of human SKI. Structure 12, 785–792 (2004).
Zhang, Y. I-TASSER server for protein 3D structure prediction. BMC Bioinformatics 9, 40 (2008).
Ueki, N. & Hayman, M.J. Direct interaction of Ski with either Smad3 or Smad4 is necessary and sufficient for Ski-mediated repression of transforming growth factor-β signaling. J. Biol. Chem. 278, 32489–32492 (2003).
Wang, L.H. et al. Inactivation of SMAD4 tumor suppressor gene during gastric carcinoma progression. Clin. Cancer Res. 13, 102–110 (2007).
Berk, M., Desai, S.Y., Heyman, H.C. & Colmenares, C. Mice lacking the ski proto-oncogene have defects in neurulation, craniofacial, patterning, and skeletal muscle development. Genes Dev. 11, 2029–2039 (1997).
Lyons, G.E. et al. Protooncogene c-ski is expressed in both proliferating and postmitotic neuronal populations. Dev. Dyn. 201, 354–365 (1994).
Colmenares, C. & Stavnezer, E. The ski oncogene induces muscle differentiation in quail embryo cells. Cell 59, 293–303 (1989).
Le Goff, C. et al. Mutations at a single codon in Mad homology 2 domain of SMAD4 cause Myhre syndrome. Nat. Genet. 44, 85–88 (2012).
Heilstedt, H.A. et al. Physical map of 1p36, placement of breakpoints in monosomy 1p36, and clinical characterization of the syndrome. Am. J. Hum. Genet. 72, 1200–1212 (2003).
Battaglia, A. et al. Further delineation of deletion 1p36 syndrome in 60 patients: a recognizable phenotype and common cause of developmental delay and mental retardation. Pediatrics 121, 404–410 (2008).
Sood, S., Eldadah, Z.A., Krause, W.L., McIntosh, I. & Dietz, H.C. Mutation in fibrillin-1 and the Marfanoid-craniosynostosis (Shprintzen-Goldberg) syndrome. Nat. Genet. 12, 209–211 (1996).
Kosaki, K. et al. Molecular pathology of Shprintzen-Goldberg syndrome. Am. J. Med. Genet. A. 140, 104–108 author reply 109–110 (2006).
Lindsay, M.E. & Dietz, H.C. Lessons on the pathogenesis of aneurysm from heritable conditions. Nature 473, 308–316 (2011).
Brooke, B.S. et al. Angiotensin II blockade and aortic-root dilation in Marfan's syndrome. N. Engl. J. Med. 358, 2787–2795 (2008).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
Li, H. et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
Wang, K., Li, M. & Hakonarson, H. ANNOVAR: functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res. 38, e164 (2010).
McKenna, A. et al. The Genome Analysis Toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res. 20, 1297–1303 (2010).
DePristo, M.A. et al. A framework for variation discovery and genotyping using next-generation DNA sequencing data. Nat. Genet. 43, 491–498 (2011).
Robinson, J.T. et al. Integrative genomics viewer. Nat. Biotechnol. 29, 24–26 (2011).
Norris, R.A. et al. Expression of the familial cardiac valvular dystrophy gene, filamin-A, during heart morphogenesis. Dev. Dyn. 239, 2118–2127 (2010).
Westerfield, M. The Zebrafish Book. A Guide for the Laboratory Use of Zebrafish (Danio rerio) (University of Oregon Press, Eugene, Oregon, 1995).
Cross, L.M., Cook, M.A., Lin, S., Chen, J.N. & Rubinstein, A.L. Rapid analysis of angiogenesis drugs in a live fluorescent zebrafish assay. Arterioscler. Thromb. Vasc. Biol. 23, 911–912 (2003).
Cui, Z., Clark, K.J., Kaufman, C.D. & Hackett, P.B. Inhibition of skiA and skiB gene expression ventralizes zebrafish embryos. Genesis 30, 149–153 (2001).
Walker, M.B. & Kimmel, C.B. A two-color acid-free cartilage and bone stain for zebrafish larvae. Biotech. Histochem. 82, 23–28 (2007).
Acknowledgements
The authors would like to thank E. Gerber and H. Bjornsson for their contributions to the cell culture experiments. This work was funded by grants to H.C.D. from the US National Institutes of Health (RO1-AR41135 and PO1-AR049698), the Bloomberg Fund of the National Marfan Foundation, the Smilow Center for Marfan Syndrome Research, the Howard Hughes Medical Institute and the Baylor-Hopkins Center for Mendelian Genetics (1U54HG006542). A.J.D. was supported as a Research Associate by the Howard Hughes Medical Institute. J.J.D. was supported by a Victor A. McKusick Fellowship from the National Marfan Foundation. M.E.L. was supported by an NHLBI K08 Award (HL107738-01) and by a Fellow-to-Faculty Award from the National Marfan Foundation. A.S.M. was supported in part by the NHLBI (1R01HL111267). M.J.C. was supported by the National Institute for Health Research (NIHR) through Barts NIHR Cardiovascular Biomedical Research Unit. This study was also supported in part by funding from the Fund for Scientific Research, Flanders (Belgium) (G.0458.09 and G.0221.12), a European Grant Fighting Aneurysmal Disease (EC-FP7) and the Special Research Fund of Ghent University (BOF10/GOA/005). B.L.L. is a Senior Clinical Investigator of the Fund for Scientific Research, Flanders (FWO, Belgium). D.S. was supported by a Ph.D. grant from the Agency for Innovation by Science and Technology.
Author information
Authors and Affiliations
Contributions
H.C.D., B.L.L., G.M., T.H., A.D., R.C.H., P.H.A. and C.J.C. recruited participants for the study. H.C.D., A.J.D., J.J.D. and M.E.L. were instrumental in the experimental design and interpretation of the data. A.J.D. performed bioinformatics analysis, with the assistance of D.W.M. and A.F.S., and carried out DNA sequencing and analysis, receiving guidance and supervision from M.J.C. and H.C.D. J.J.D. performed all cell culture experiments and protein blot and quantitative PCR analyses, with the assistance of N.D.H. S.L.B. and S.M. performed all zebrafish experiments under the supervision of A.S.M. B.L.L. and L.V.L. supervised D.S. and E.G., who performed DNA sequencing and analysis. K.S. and R.A.N. performed the mouse developmental survey. D.L. performed structural prediction analyses. A.J.D. and J.J.D. drafted and are co-lead authors for the manuscript. H.C.D., B.L.L. and L.V.L. critically revised and gave final approval for the manuscript to be published.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5 and Supplementary Tables 1 and 2 (PDF 2749 kb)
Rights and permissions
About this article
Cite this article
Doyle, A., Doyle, J., Bessling, S. et al. Mutations in the TGF-β repressor SKI cause Shprintzen-Goldberg syndrome with aortic aneurysm. Nat Genet 44, 1249–1254 (2012). https://doi.org/10.1038/ng.2421
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.2421
This article is cited by
-
The roles and regulatory mechanisms of TGF-β and BMP signaling in bone and cartilage development, homeostasis and disease
Cell Research (2024)
-
Nucleotide substitutions at the p.Gly117 and p.Thr180 mutational hot-spots of SKI alter molecular dynamics and may affect cell cycle
Journal of Human Genetics (2024)
-
Aortic aneurysms: current pathogenesis and therapeutic targets
Experimental & Molecular Medicine (2023)
-
Genome-wide identification and phenotypic characterization of seizure-associated copy number variations in 741,075 individuals
Nature Communications (2023)
-
SMAD6-deficiency in human genetic disorders
npj Genomic Medicine (2022)