Abstract
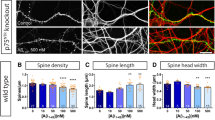
Amyloid-beta (Aβ) oligomers are thought to trigger Alzheimer's disease pathophysiology. Cellular prion protein (PrPC) selectively binds oligomeric Aβ and can mediate Alzheimer's disease–related phenotypes. We examined the specificity, distribution and signaling of Aβ-PrPC complexes, seeking to understand how they might alter the function of NMDA receptors (NMDARs) in neurons. PrPC is enriched in postsynaptic densities, and Aβ-PrPC interaction leads to Fyn kinase activation. Soluble Aβ assemblies derived from the brains of individuals with Alzheimer's disease interacted with PrPC to activate Fyn. Aβ engagement of PrPC-Fyn signaling yielded phosphorylation of the NR2B subunit of NMDARs, which was coupled to an initial increase and then a loss of surface NMDARs. Aβ-induced dendritic spine loss and lactate dehydrogenase release required both PrPC and Fyn, and human familial Alzheimer's disease transgene–induced convulsive seizures did not occur in mice lacking PrPC. These results delineate an Aβ oligomer signal transduction pathway that requires PrPC and Fyn to alter synaptic function, with deleterious consequences in Alzheimer's disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Lambert, M.P. et al. Diffusible, nonfibrillar ligands derived from Abeta1–42 are potent central nervous system neurotoxins. Proc. Natl. Acad. Sci. USA 95, 6448–6453 (1998).
Walsh, D.M. et al. Naturally secreted oligomers of amyloid beta protein potently inhibit hippocampal long-term potentiation in vivo. Nature 416, 535–539 (2002).
Shankar, G.M. et al. Amyloid-beta protein dimers isolated directly from Alzheimer's brains impair synaptic plasticity and memory. Nat. Med. 14, 837–842 (2008).
Lesné, S. et al. A specific amyloid-beta protein assembly in the brain impairs memory. Nature 440, 352–357 (2006).
Laurén, J., Gimbel, D.A., Nygaard, H.B., Gilbert, J.W. & Strittmatter, S.M. Cellular prion protein mediates impairment of synaptic plasticity by amyloid-beta oligomers. Nature 457, 1128–1132 (2009).
Zou, W.Q. et al. Amyloid-{beta}42 interacts mainly with insoluble prion protein in the alzheimer brain. J. Biol. Chem. 286, 15095–15105 (2011).
Balducci, C. et al. Synthetic amyloid-beta oligomers impair long-term memory independently of cellular prion protein. Proc. Natl. Acad. Sci. USA 107, 2295–2300 (2010).
Chen, S., Yadav, S.P. & Surewicz, W.K. Interaction between human prion protein and amyloid-beta (Abeta) oligomers: role of N-terminal residues. J. Biol. Chem. 285, 26377–26383 (2010).
Calella, A.M. et al. Prion protein and Abeta-related synaptic toxicity impairment. EMBO Mol. Med. 2, 306–314 (2010).
Freir, D.B. et al. Interaction between prion protein and toxic amyloid beta assemblies can be therapeutically targeted at multiple sites. Nat. Commun. 2, 336 (2011).
Cisse, M. et al. Ablation of cellular prion protein does not ameliorate abnormal neural network activity or cognitive dysfunction in the J20 line of human amyloid precursor protein transgenic mice. J. Neurosci. 31, 10427–10431 (2011).
Kessels, H.W., Nguyen, L.N., Nabavi, S. & Malinow, R. The prion protein as a receptor for amyloid-beta. Nature 466, E3–4 (2010).
Gimbel, D.A. et al. Memory impairment in transgenic Alzheimer mice requires cellular prion protein. J. Neurosci. 30, 6367–6374 (2010).
Chung, E. et al. Anti-PrPC monoclonal antibody infusion as a novel treatment for cognitive deficits in an Alzheimer's disease model mouse. BMC Neurosci. 11, 130 (2010).
Resenberger, U.K. et al. The cellular prion protein mediates neurotoxic signalling of beta-sheet-rich conformers independent of prion replication. EMBO J. 30, 2057–2070 (2011).
Barry, A.E. et al. Alzheimer's disease brain–derived amyloid-beta–mediated inhibition of LTP in vivo is prevented by immunotargeting cellular prion protein. J. Neurosci. 31, 7259–7263 (2011).
Bate, C. & Williams, A. Amyloid-beta–induced synapse damage is mediated via cross-linkage of cellular prion proteins. J. Biol. Chem. 286, 37955–37963 (2011).
You, H. et al. Abeta neurotoxicity depends on interactions between copper ions, prion protein and N-methyl-D-aspartate receptors. Proc. Natl. Acad. Sci. USA 109, 1737–1742 (2012).
Alier, K., Ma, L., Yang, J., Westaway, D. & Jhamandas, J.H. Abeta inhibition of ionic conductance in mouse basal forebrain neurons is dependent upon the cellular prion protein PrPC. J. Neurosci. 31, 16292–16297 (2011).
Kudo, W. et al. Cellular prion protein is essential for oligomeric amyloid-beta–induced neuronal cell death. Hum. Mol. Genet. 21, 1138–1144 (2012).
Shankar, G.M. et al. Natural oligomers of the Alzheimer amyloid-beta protein induce reversible synapse loss by modulating an NMDA-type glutamate receptor–dependent signaling pathway. J. Neurosci. 27, 2866–2875 (2007).
Lacor, P.N. et al. Abeta oligomer-induced aberrations in synapse composition, shape and density provide a molecular basis for loss of connectivity in Alzheimer's disease. J. Neurosci. 27, 796–807 (2007).
Snyder, E.M. et al. Regulation of NMDA receptor trafficking by amyloid-beta. Nat. Neurosci. 8, 1051–1058 (2005).
Pantera, B. et al. PrPc activation induces neurite outgrowth and differentiation in PC12 cells: role for caveolin-1 in the signal transduction pathway. J. Neurochem. 110, 194–207 (2009).
Mouillet-Richard, S. et al. Cellular prion protein signaling in serotonergic neuronal cells. Ann. NY Acad. Sci. 1096, 106–119 (2007).
Williamson, R., Usardi, A., Hanger, D.P. & Anderton, B.H. Membrane-bound beta-amyloid oligomers are recruited into lipid rafts by a fyn-dependent mechanism. FASEB J. 22, 1552–1559 (2008).
Málaga-Trillo, E. et al. Regulation of embryonic cell adhesion by the prion protein. PLoS Biol. 7, e55 (2009).
Bizat, N. et al. Neuron dysfunction is induced by prion protein with an insertional mutation via a Fyn kinase and reversed by sirtuin activation in Caenorhabditis elegans. J. Neurosci. 30, 5394–5403 (2010).
Suzuki, T. & Okumura-Noji, K. NMDA receptor subunits epsilon 1 (NR2A) and epsilon 2 (NR2B) are substrates for Fyn in the postsynaptic density fraction isolated from the rat brain. Biochem. Biophys. Res. Commun. 216, 582–588 (1995).
Grant, S.G. et al. Impaired long-term potentiation, spatial learning, and hippocampal development in fyn mutant mice. Science 258, 1903–1910 (1992).
Nakazawa, T. et al. Characterization of Fyn-mediated tyrosine phosphorylation sites on GluR epsilon 2 (NR2B) subunit of the N-methyl-D-aspartate receptor. J. Biol. Chem. 276, 693–699 (2001).
Chin, J. et al. Fyn kinase induces synaptic and cognitive impairments in a transgenic mouse model of Alzheimer's disease. J. Neurosci. 25, 9694–9703 (2005).
Renner, M. et al. Deleterious effects of amyloid beta oligomers acting as an extracellular scaffold for mGluR5. Neuron 66, 739–754 (2010).
Collins, M.O. et al. Molecular characterization and comparison of the components and multiprotein complexes in the postsynaptic proteome. J. Neurochem. 97 Suppl 1, 16–23 (2006).
Stuermer, C.A. et al. PrPc capping in T cells promotes its association with the lipid raft proteins reggie-1 and reggie-2 and leads to signal transduction. FASEB J. 18, 1731–1733 (2004).
Salter, M.W. & Kalia, L.V. Src kinases: a hub for NMDA receptor regulation. Nat. Rev. Neurosci. 5, 317–328 (2004).
Zhang, Y. et al. Genetic reduction of striatal-enriched tyrosine phosphatase (STEP) reverses cognitive and cellular deficits in an Alzheimer's disease mouse model. Proc. Natl. Acad. Sci. USA 107, 19014–19019 (2010).
Prybylowski, K. et al. The synaptic localization of NR2B-containing NMDA receptors is controlled by interactions with PDZ proteins and AP-2. Neuron 47, 845–857 (2005).
Chen, P., Gu, Z., Liu, W. & Yan, Z. Glycogen synthase kinase 3 regulates N-methyl-D-aspartate receptor channel trafficking and function in cortical neurons. Mol. Pharmacol. 72, 40–51 (2007).
Scheff, S.W., DeKosky, S.T. & Price, D.A. Quantitative assessment of cortical synaptic density in Alzheimer's disease. Neurobiol. Aging 11, 29–37 (1990).
Palop, J.J. et al. Aberrant excitatory neuronal activity and compensatory remodeling of inhibitory hippocampal circuits in mouse models of Alzheimer's disease. Neuron 55, 697–711 (2007).
Minkeviciene, R. et al. Amyloid beta–induced neuronal hyperexcitability triggers progressive epilepsy. J. Neurosci. 29, 3453–3462 (2009).
McDonald, J.M. et al. The presence of sodium dodecyl sulphate-stable Abeta dimers is strongly associated with Alzheimer-type dementia. Brain 133, 1328–1341 (2010).
Steinerman, J.R. et al. Distinct pools of beta-amyloid in Alzheimer disease–affected brain: a clinicopathologic study. Arch. Neurol. 65, 906–912 (2008).
Kuo, Y.M. et al. Water-soluble Abeta (N-40, N-42) oligomers in normal and Alzheimer disease brains. J. Biol. Chem. 271, 4077–4081 (1996).
Peng, J. et al. Semiquantitative proteomic analysis of rat forebrain postsynaptic density fractions by mass spectrometry. J. Biol. Chem. 279, 21003–21011 (2004).
Yoshimura, Y. et al. Molecular constituents of the postsynaptic density fraction revealed by proteomic analysis using multidimensional liquid chromatography-tandem mass spectrometry. J. Neurochem. 88, 759–768 (2004).
Li, S. et al. Soluble A{beta} oligomers inhibit long-term potentiation through a mechanism involving excessive activation of extrasynaptic NR2B-containing NMDA receptors. J. Neurosci. 31, 6627–6638 (2011).
Kojima, N., Ishibashi, H., Obata, K. & Kandel, E.R. Higher seizure susceptibility and enhanced tyrosine phosphorylation of N-methyl-D-aspartate receptor subunit 2B in fyn transgenic mice. Learn. Mem. 5, 429–445 (1998).
Ittner, L.M. et al. Dendritic function of tau mediates amyloid-beta toxicity in Alzheimer's disease mouse models. Cell 142, 387–397 (2010).
Stein, P.L., Lee, H.M., Rich, S. & Soriano, P. pp59fyn mutant mice display differential signaling in thymocytes and peripheral T cells. Cell 70, 741–750 (1992).
Zahn, R., von Schroetter, C. & Wuthrich, K. Human prion proteins expressed in Escherichia coli and purified by high-affinity column refolding. FEBS Lett. 417, 400–404 (1997).
Acknowledgements
H.B.N. is an Ellison Medical Foundation American Federation for Aging Research Postdoctoral Fellow and S.M.S. is a member of the Kavli Institute for Neuroscience at Yale University. We acknowledge support from the US National Institutes of Health (R01AG034924, R37NS033020, R01NS074319 and P30DA018343), the Falk Medical Research Trust and the Alzheimer's Association (S.M.S.), and from the US National Institutes of Health (R01NS47433 and R01NS073502; T.W.).
Author information
Authors and Affiliations
Contributions
J.W.U., H.B.N., J.K.H., M.A.K., M.S., A.V., E.C.G. and S.M.S. designed various aspects of the research. J.W.U., H.B.N., J.K.H., M.S. and E.C.G. performed the research. J.W.U., H.B.N., J.K.H., M.S., T.W., E.C.G. and S.M.S. analyzed various parts of the data and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
S.M.S. is a co-founder of Axerion Therapeutics, which seeks to develop NgR- and PrP-based therapeutics.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–10 (PDF 28592 kb)
Supplementary Movie 1
Example of NMDA-induced intracellular calcium signal. (MOV 28335 kb)
Supplementary Movie 2
Example of three-dimensional reconstruction of images of dendritic spines. (MOV 4446 kb)
Supplementary Movie 3
Example of APP/PSen Transgenic Mouse Seizure. (WMV 10234 kb)
Supplementary Movie 4
Second Example of APP/PSen Transgenic Mouse Seizure. (WMV 9450 kb)
Rights and permissions
About this article
Cite this article
Um, J., Nygaard, H., Heiss, J. et al. Alzheimer amyloid-β oligomer bound to postsynaptic prion protein activates Fyn to impair neurons. Nat Neurosci 15, 1227–1235 (2012). https://doi.org/10.1038/nn.3178
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3178
This article is cited by
-
Fyn Kinase in Alzheimer’s Disease: Unraveling Molecular Mechanisms and Therapeutic Implications
Molecular Neurobiology (2024)
-
Aberrant palmitoylation caused by a ZDHHC21 mutation contributes to pathophysiology of Alzheimer’s disease
BMC Medicine (2023)
-
Synaptic proteasome is inhibited in Alzheimer’s disease models and associates with memory impairment in mice
Communications Biology (2023)
-
Amyloid β-based therapy for Alzheimer’s disease: challenges, successes and future
Signal Transduction and Targeted Therapy (2023)
-
Fyn nanoclustering requires switching to an open conformation and is enhanced by FTLD-Tau biomolecular condensates
Molecular Psychiatry (2023)