Abstract
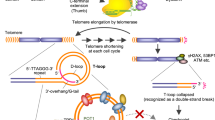
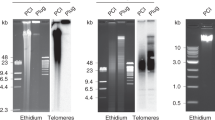
Nucleic acids containing stretches of tandem guanines can fold into four-stranded structures called G-quadruplexes. The existence of such sequences in genomic DNA suggests the occurrence of these motifs in cells, with potential implications in a number of biological processes relevant to cancer. Small molecules have proven to be valuable tools to dissect cell circuitry. Here, we describe a synthetic small molecule derived from an N,N′-bis(2-quinolinyl)pyridine-2,6-dicarboxamide, which is designed to mediate the selective isolation of G-quadruplex nucleic acids. The methodology was successfully applied to a range of DNA and RNA G-quadruplexes in vitro. We demonstrate the general applicability of the method by isolating telomeric DNA-containing G-quadruplex motifs from cells. We show that telomeres are targets for the probe, providing further evidence of the formation of G-quadruplexes in human cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Taunton, J., Hassig, C. A. & Schreiber, S. L. A mammalian histone deacetylase related to the yeast transcriptional regulator Rpd3p. Science 272, 408–411 (1996).
Kielkopf, C. L. et al. A structural basis for recognition of A·T and T·A base pairs in the minor groove of B-DNA. Science 282, 111–115 (1998).
Schaeffer, C. et al. The fragile X mental retardation protein binds specifically to its mRNA via a purine quartet motif. EMBO J. 20, 4803–4813 (2001).
Siddiqui-Jain, A., Grand, C. L., Bearss, D. J. & Hurley, L. H. Direct evidence for a G-quadruplex in a promoter region and its targeting with a small molecule to repress c-MYC transcription. Proc. Natl Acad. Sci. USA 99, 11593–11598 (2002).
Zahler, A. M., Williamson, J. R., Cech, T. R. & Prescott, D. M. Inhibition of telomerase by G-quartet DNA structures. Nature 350, 718–720 (1991).
Rizzo, A. et al. Stabilization of quadruplex DNA perturbs telomere replication leading to the activation of an ATR-dependent ATM signalling pathway. Nucleic Acids Res. 37, 5353–5364 (2009).
Huppert, J. L. & Balasubramanian, S. Prevalence of quadruplexes in the human genome. Nucleic Acids Res. 33, 2908–2916 (2005).
Barski, A. et al. High-resolution profiling of histone methylation in the human genome. Cell 129, 823–837 (2007).
Johnson, D. S., Mortazavi, A., Myers, R. M. & Wold, B. Genome-wide mapping of in vivo protein–DNA interactions. Science 316, 1497–1502 (2007).
Bentley, D. R. et al. Accurate whole human genome sequencing using reversible terminator chemistry. Nature 456, 53–59 (2008).
Rodriguez, R. et al. A novel small molecule that alters shelterin integrity and triggers a DNA-damage response at telomeres. J. Am. Chem. Soc. 130, 15758–15759 (2008).
Buguat, A., Rodriguez, R., Kumari, S., Hsu, S.-T. D. & Balasubramanian, S. Small molecule-mediated inhibition of translation by targeting a native RNA G-quadruplex. Org. Biomol. Chem. 8, 2771–2776 (2010).
d'Adda di Fagagna, F. et al. A DNA damage checkpoint response in telomere-initiated senescence. Nature 426, 194–198 (2003).
Hunter, C. A. & Sanders, J. K. M. The nature of π–π interactions. J. Am. Chem. Soc. 112, 5525–5534 (1990).
Müller, S., Pantoş, G. D., Rodriguez, R. & Balasubramanian, S. Controlled-folding of a small molecule modulates DNA G-quadruplex recognition. Chem. Commun. 80–82 (2009).
Berl, B., Huc, I., Khoury, R. G., Krische, M. J. & Lehn, J.-M. Interconversion of single and double helices formed from synthetic molecular strands. Nature 402, 720–723 (2000).
Jain, S. L. et al. New pyridine carboxamide ligands and their complexation to copper(II). X-ray crystal structure of mono-, di, tri- and tetranuclear copper complexes. Dalton Trans. 862–871 (2004).
Parkinson, G. N., Lee, M. P. H. & Neidle, S. Crystal structure of parallel quadruplexes from human telomeric DNA. Nature 417, 876–880 (2002).
Phan, A. T., Kuryavyi, V., Burge, S., Neidle, S. & Patel, D. J. Structure of an unprecedented G-quadruplex scaffold in the human c-kit promoter. J. Am. Chem. Soc. 129, 4386–4392 (2007).
Hsu, S.-T. D. et al. A G-rich sequence within the c-kit oncogene promoter forms a parallel G-quadruplex having asymmetric G-tetrad dynamics. J. Am. Chem. Soc. 131, 13399–13409 (2009).
Kumari, S., Bugaut, A., Huppert, J. L. & Balasubramanian, S. An RNA G-quadruplex in the 5′ UTR of the NRAS proto-oncogene modulates translation. Nature Chem. Biol. 3, 218–221 (2007).
Rodriguez, R., Pantoş, G. D., Gonçalves, D. P. N., Sanders, J. K. M. & Balasubramanian, S. Ligand-driven G-quadruplex conformational switching by using an unusual mode of interaction. Angew. Chem. Int. Ed. 46, 5405–5407 (2007).
Temime-Smaali, N. et al. The G-quadruplex ligand telomestatin impairs binding of topoisomerase IIIα to G-quadruplex-forming oligonucleotides and uncaps telomeres in ALT cells. PloS One 4, e6919 (2009).
d'Adda di Fagagna, F. Living on a break: cellular senescence as a DNA-damage response. Nature Rev. Cancer 8, 512–522 (2008).
Frye, S. V. The art of the chemical probe. Nature Chem. Biol. 6, 159–161 (2010).
Cawthon, C. Telomere measurement by quantitative PCR. Nucleic Acids Res. 30, e47 (2002).
Paeschke, K., Simonsson, T., Postberg, J., Rhodes, D. & Lipps, H. J. Telomeres end-binding proteins control the formation of G-quadruplex DNA structures in vivo. Nature Struct. Mol. Biol. 12, 847–854 (2005).
Acknowledgements
The authors thank D.A. Sanders, R. Kranaster, J.–F. Riou, D. Gomez, A.R. Venkitaraman, A.J. Bannister and K. Holmes for fruitful discussions and advice. The authors also acknowledge Cancer Research UK for a studentship (S.M.) and programme funding, and the Biotechnology and Biological Sciences Research Council for project funding. R.R. is a Herchel Smith Research Fellow.
Author information
Authors and Affiliations
Contributions
R.R. and S.B. conceptualized the study. R.R. is the inventor of pyridostatin and related analogues. All the authors designed the experiments and analysed the data. S.M. and S.K. performed the experiments. R.R. and S.B wrote the manuscript with contributions from S.M. and S.K.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 1914 kb)
Rights and permissions
About this article
Cite this article
Müller, S., Kumari, S., Rodriguez, R. et al. Small-molecule-mediated G-quadruplex isolation from human cells. Nature Chem 2, 1095–1098 (2010). https://doi.org/10.1038/nchem.842
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.842
This article is cited by
-
Chem-map profiles drug binding to chromatin in cells
Nature Biotechnology (2023)
-
Stress promotes RNA G-quadruplex folding in human cells
Nature Communications (2023)
-
High-throughput techniques enable advances in the roles of DNA and RNA secondary structures in transcriptional and post-transcriptional gene regulation
Genome Biology (2022)
-
Global mapping of RNA G-quadruplexes (G4-RNAs) using G4RP-seq
Nature Protocols (2022)
-
Facile induction and stabilization of intramolecular antiparallel G-quadruplex of d(TTAGGG)n on interaction with triazine-2-imidazole ethyl amine compound and its Cu(II), Zn(II) complexes under no-salt conditions
Journal of Chemical Sciences (2022)