Abstract
Cancer progression is driven in part by genomic alterations1. The genomic characterization of cancers has shown interpatient heterogeneity regarding driver alterations2, leading to the concept that generation of genomic profiling in patients with cancer could allow the selection of effective therapies3,4. Although DNA sequencing has been implemented in practice, it remains unclear how to use its results. A total of 1,462 patients with HER2-non-overexpressing metastatic breast cancer were enroled to receive genomic profiling in the SAFIR02-BREAST trial. Two hundred and thirty-eight of these patients were randomized in two trials (nos. NCT02299999 and NCT03386162) comparing the efficacy of maintenance treatment5 with a targeted therapy matched to genomic alteration. Targeted therapies matched to genomics improves progression-free survival when genomic alterations are classified as level I/II according to the ESMO Scale for Clinical Actionability of Molecular Targets (ESCAT)6 (adjusted hazards ratio (HR): 0.41, 90% confidence interval (CI): 0.27–0.61, P < 0.001), but not when alterations are unselected using ESCAT (adjusted HR: 0.77, 95% CI: 0.56–1.06, P = 0.109). No improvement in progression-free survival was observed in the targeted therapies arm (unadjusted HR: 1.15, 95% CI: 0.76–1.75) for patients presenting with ESCAT alteration beyond level I/II. Patients with germline BRCA1/2 mutations (n = 49) derived high benefit from olaparib (gBRCA1: HR = 0.36, 90% CI: 0.14–0.89; gBRCA2: HR = 0.37, 90% CI: 0.17–0.78). This trial provides evidence that the treatment decision led by genomics should be driven by a framework of target actionability in patients with metastatic breast cancer.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
Genomic data and modalities for access are available at EGAS00001005584 and https://nextcloud.gustaveroussy.fr/s/JXLt7taZs8EtBF7.
References
Hanahan, D. & Weinberg, R. A. Hallmarks of cancer: the next generation. Cell. 144, 646–674 (2011).
Liu, J. et al. An integrated TCGA pan-cancer clinical data resource to drive high-quality survival outcome analytics. Cell. 173, 400–416 (2018).
Yates, L. R. et al. The European Society for Medical Oncology (ESMO) Precision Medicine Glossary. Ann. Oncol. 29, 30–35 (2018).
Arnedos, M. et al. Precision medicine for metastatic breast cancer—limitations and solutions. Nat. Rev. Clin. Oncol. 12, 693–704 (2015).
Gennari, A. et al. ESMO Clinical Practice Guideline for the diagnosis, staging and treatment of patients with metastatic breast cancer. Ann. Oncol. 32, 1475–1495 (2021).
Mateo, J. et al. A framework to rank genomic alterations as targets for cancer precision medicine: the ESMO Scale for Clinical Actionability of molecular Targets (ESCAT). Ann. Oncol. 29, 1895–1902 (2018).
André, F. et al. Comparative genomic hybridisation array and DNA sequencing to direct treatment of metastatic breast cancer: a multicentre, prospective trial (SAFIR01/UNICANCER). Lancet Oncol. 15, 267–274 (2014).
Massard, C. et al. High-throughput genomics and clinical outcome in hard-to-treat advanced cancers: results of the MOSCATO 01 trial. Cancer Discov. 7, 586–595 (2017).
Mosele, F. et al. Recommendations for the use of next generation sequencing in patients with metastatic cancers: a report from the ESMO precision medicine working group. Ann. Oncol. 31, 1491–1505 (2020).
Le Tourneau, C. et al. Molecularly targeted therapy based on tumour molecular profiling versus conventional therapy for advanced cancer (SHIVA): a multicentre, open-label, proof-of-concept, randomised, controlled phase 2 trial. Lancet Oncol. 16, 1324–1334 (2015).
Kingston, B. et al. Exceptional response to AKT inhibition in patients with breast cancer and germline PTEN mutations. JCO Precis. Oncol. 3, PO.19.00130 (2019).
Taniguchi, C. M. et al. The phosphoinositide 3-kinase regulatory subunit p85alpha can exert tumor suppressor properties through negative regulation of growth factor signaling. Cancer Res. 70, 5305–5315 (2010).
Curtis, C. The genomic and transcriptomic architecture of 2,000 breast tumours reveals novel subgroups. Nature 486, 346–352 (2012).
Li, M. M. et al. Standards and guidelines for the interpretation and reporting of sequence variants in cancer: a joint consensus recommendation of the Association for Molecular Pathology, American Society of Clinical Oncology, and College of American Pathologists. J. Mol. Diagn. 19, 4–23 (2017).
Chakravarty, D. et al. OncoKB: a precision oncology knowledge base. JCO Precis. Oncol. 2017, PO.17.00011 (2017).
Johnson, A. et al. Clinical use of precision oncology decision support. JCO Precis. Oncol. 2017, PO.17.00036 (2017).
Hyman, D. M., Taylor, B. S. & Baselga, J. Implementing genome-driven oncology. Cell. 168, 584–599 (2017).
Costello, J. C. et al. A community effort to assess and improve drug sensitivity prediction algorithms. Nat. Biotechnol. 32, 1202–1212 (2014).
Tuveson, D. & Clevers, H. Cancer modeling meets human organoid technology. Science 364, 952–955 (2019).
Lee, S. H. et al. Tumor evolution and drug response in patient-derived organoid models of bladder cancer. Cell. 173, 515–528 (2018).
Robson, M. et al. Olaparib for metastatic breast cancer in patients with a germline BRCA mutation. N. Engl. J. Med. 377, 523–533 (2017).
Hyman, D. M. et al. HER kinase inhibition in patients with HER2- and HER3-mutant cancers. Nature 554, 189–194 (2018).
Bachelot, et al. Durvalumab compared to maintenance chemotherapy in metastatic breast cancer: the randomized phase II SAFIR02-BREAST IMMUNO trial. Nat. Med. 27, 250–255 (2021).
Commo, F. et al. rCGH: a comprehensive array-based genomic profile platform for precision medicine. Bioinformatics 32, 1402–1404 (2016).
Davies, H. et al. HRDetect is a predictor of BRCA1 and BRCA2 deficiency based on mutational signatures. Nat. Med. 23, 517–525 (2017).
Telli, M. L. et al. Homologous recombination deficiency (HRD) score predicts response to platinum-containing neoadjuvant chemotherapy in patients with triple-negative breast cancer. Clin. Cancer Res. 22, 3764–3773 (2016).
Carene, D. et al. Association between FGFR1 copy numbers, MAP3K1 mutations, and survival in axillary node-positive, hormone receptor-positive, and HER2-negative early breast cancer in the PACS04 and METABRIC studies. Breast Cancer Res. Treat. 179, 387–401 (2020).
Condorelli R et al, Genomic alterations in breast cancer: level of evidence for actionability according to ESMO Scale for Clinical Actionability of molecular Targets (ESCAT). Ann. Oncol. 30, 365–373 (2019)
Tung NM et al, TBCRC 048: Phase II Study of Olaparib for Metastatic Breast Cancer and Mutations in Homologous Recombination-Related Genes. J. Clin. Oncol. 38, 4274–4282 (2020)
Acknowledgements
We thank the patients and their families, as well as the investigators (especially V. Massard, T. l’Haridon, L. Venat-Bouvet, F. Del Piano, X. Tchiknavorian, N. Dohollou and C. Alliot) and their staff involved in SAFIR02-BREAST and SAFIR-PI3K. We also thank the UNICANCER team involved in both trials and C. Honfo Ga for data management. We also thank members of the IDMC (N. Turner, S. Loibl, S. Novello, E. Felip, S, Litiere and D. Beltram). The study was funded by Fondation ARC, AstraZeneca, the IHU-B programme (PRISM) and the Breast Cancer Research Foundation. AstraZeneca also supported targeted therapy (capivasertib, AZD2014, AZD8931, AZD4547, olaparib, selumetinib, bicalutamide and vandetanib) supply and distribution to the study sites. Novartis supported the supply of targeted therapy (alpelisib).
Author information
Authors and Affiliations
Contributions
F.A., T.B., T.F. and M.J. designed the study. F.A. and T.B. were the coordinators of the SAFIR02-BREAST trial. F.A. and A.G. were the coordinators of the SAFIR-PI3K trial. M.J. and A.J. operated the trial. T.B., M.A., F.D., M.-P.S., M.C., H.B., C.L.-P., W.J., F.C., J.-M.F., C.L.-P., M.-A.M.R., J.-C.T., N.I., A. Mege, P.B., B.Y., N.H. and A.G. included patients in the trial. I.B., L.L., E.R., S.B., V.A., I.S., M.K., C.L., N.S., P.G., L.L.C. and A. Morel performed genomic analyses. A.T.-D. collected genomic data and stored them on EGA. F.A., T.B., F. Mosele, T.F., A.L., I.B., M.J., F. Montemurro and A.J. wrote the manuscript. All authors have read and agreed on the content of the manuscript.
Corresponding author
Ethics declarations
Competing interests
F.A. received research funding and served as a speaker/advisor (compensated by the hospital) for Pfizer, Roche, Lilly, Daiichi Sankyo, AstraZeneca and Novartis. T.B. received research funding and served as a speaker/advisor (compensated by the hospital) for Roche, Novartis, Pfizer, Seattle Genetics, Lilly and AstraZeneca. M.A. received research funding and served as a speaker/advisor (compensated by the hospital) for Novartis, AstraZeneca, Seattle Genetics, Abvie and Pfizer. M.C. received research funding and served as a speaker/advisor (compensated by the hospital) for AstraZeneca, Novartis, Abbvie, Sanofi, Lilly, Pfizer, Sandoz, ACCORD, G1 Therapeutic, Pierre Fabre Oncology, Servier, Roche, Daiichi and Gilead. F.D. received research funding and served as a speaker/advisor (compensated by the hospital) for Roche, Novartis, Lilly, Pfizer, Eisai, MSD and AstraZeneca. C.L.-P. received research funding and served as a speaker/advisor (compensated by the hospital) for AstraZeneca, Roche and Pfizer. A.G. received research funding and served as a speaker/advisor (compensated by the hospital) for AstraZeneca, Pfizer, Novartis, Roche and MSD. M.-A.M.R. received research funding and served as a speaker/advisor for Pfizer, Novartis, Lilly, Roche, MSD and Myriad. W.J. received research funding and served as a speaker/advisor for AstraZeneca, BMS, Daiichi Sankyo, Eisai, Lilly France, MSD, Novartis, Pfizer and Roche. B.Y. received research funding and served as a speaker/advisor for MSD, AstraZeneca, GSK-TESARO, BAYER, Roche-Genentech, ECS Progastrine, Novartis, LEK, Amgen, Clovis Oncology, Merck Serono, BMS, SEAGEN and Myriad. P.B. served as a speaker/advisor for Roche, BMS, IPSEN, MSD, Bayer, Amgen, Esai, Janssen Cilag, Pfizer, Novartis and Astellas and received honoraria from Seagen. N.H. received research funding from Novartis and Pfizer, and served as a consultant for AstraZeneca, Roche, Novartis and Pfizer. N.I. received research funding and served as a speaker/advisor (compensated by the hospital) for Ipsen and Transgene. J.-C.T. received research funding and served as a speaker/advisor (compensated by the hospital) for Pfizer and AstraZeneca. J.-M.F. received research funding and served as a speaker/advisor for Pfizer and Esai. T.F. consulted for Cellectis (compensated by the hospital). The following authors declared no competing interests: F. Mosele, L.L., G.E., A. Mege, F.C., M.-P.S., I.B., E.R., I.S., V.A., S.B., A. Morel, A.T.-D., A.L., H.B., M.J., A.J., L.L.C., M.K., P.G. and N.S. C.L. participated in advisory boards for MSD, BMS, Merck Serono, GSK, AstraZeneca, Nanobiotix, Roche, Rakuten, Seattle Genetics and Celgene. F. Montemurro received consultancy fees from Roche, AstraZeneca, Daiichi Sankyo, SeaGen, MSD, Eli Lilly, Pierre Fabre and Novartis, and travel grants from Roche.
Peer review
Peer review information
Nature thanks Alicia Okines and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
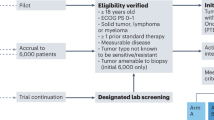
Extended Data Fig. 1
CONSORT diagram of the trial.
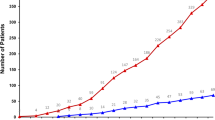
Extended Data Fig. 2 Genomic alterations identification in patients with HR+/Her2- (left panel) or TNBC (right panel).
The analysis focuses on the 50 genes that were included in the first panel and on the copy number analyses.
Extended Data Fig. 3 Subgroup analysis regarding efficacy of targeted therapy on Progression free survival, in patients presenting an ESCAT I/II alteration.
The figure reports unadjusted Hazard Ratio (diamonds) and 90% confidence intervals (error bars) estimated using a Cox proportional hazard model in each subgroup for progression or death according to clinical and biological variables. P-value for interaction between treatment arm and each variable was estimated using a Cox proportional hazard model fitted with the treatment arm, the variable and an interaction term between treatment arm and variable. All statistical tests were two sided. No adjustment was made for multiple comparisons. *: A: tyrosine kinase, B: PI3K/mTOR pathway, C: MEK pathway, D: DNA repair.
Extended Data Fig. 4 Subgroup analysis regarding efficacy of targeted therapy on progression free survival, in the intent-to-treat population.
The figure reports unadjusted Hazard Ratio (diamonds) and 95% confidence intervals (error bars) estimated using a Cox proportional hazard model in each subgroup for progression or death according to clinical and biological variables. P-value for interaction between treatment arm and each variable was estimated using a Cox proportional hazard model fitted with the treatment arm, the variable and an interaction term between treatment arm and variable. All statistical tests were two sided. No adjustment was made for multiple comparisons. *: A: tyrosine kinase, B: PI3K/mTOR pathway, C: MEK pathway, D: DNA repair.
Supplementary information
Supplementary Data 1
Statistical report for the trial and ancillary analyses.
Supplementary Data 2
Report regarding TP53, HRD and PIK3CA mutations.
Supplementary Data 3
Original protocol for SAFIR02-BREAST.
Supplementary Data 4
Final version of the SAFIR02-BREAST protocol.
Supplementary Data 5
SAFIR-PI3K protocol.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Andre, F., Filleron, T., Kamal, M. et al. Genomics to select treatment for patients with metastatic breast cancer. Nature 610, 343–348 (2022). https://doi.org/10.1038/s41586-022-05068-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-022-05068-3
This article is cited by
-
Deep learning in cancer genomics and histopathology
Genome Medicine (2024)
-
Circulating tumor DNA validity and potential uses in metastatic breast cancer
npj Breast Cancer (2024)
-
Genomic spectrum of actionable alterations in serial cell free DNA (cfDNA) analysis of patients with metastatic breast cancer
npj Breast Cancer (2024)
-
Forget lung, breast or prostate cancer: why tumour naming needs to change
Nature (2024)
-
A new treatment for breast cancer using a combination of two drugs: AZD9496 and palbociclib
Scientific Reports (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.