Abstract
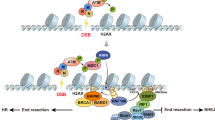
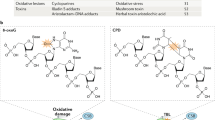
Oxidative genome damage is an unavoidable consequence of cellular metabolism. It arises at gene regulatory elements by epigenetic demethylation during transcriptional activation1,2. Here we show that promoters are protected from oxidative damage via a process mediated by the nuclear mitotic apparatus protein NuMA (also known as NUMA1). NuMA exhibits genomic occupancy approximately 100 bp around transcription start sites. It binds the initiating form of RNA polymerase II, pause-release factors and single-strand break repair (SSBR) components such as TDP1. The binding is increased on chromatin following oxidative damage, and TDP1 enrichment at damaged chromatin is facilitated by NuMA. Depletion of NuMA increases oxidative damage at promoters. NuMA promotes transcription by limiting the polyADP-ribosylation of RNA polymerase II, increasing its availability and release from pausing at promoters. Metabolic labelling of nascent RNA identifies genes that depend on NuMA for transcription including immediate–early response genes. Complementation of NuMA-deficient cells with a mutant that mediates binding to SSBR, or a mitotic separation-of-function mutant, restores SSBR defects. These findings underscore the importance of oxidative DNA damage repair at gene regulatory elements and describe a process that fulfils this function.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The original images for all gels and immunoblots are available in Supplementary Figure 1. Mass spectrometry data have been deposited to the ProteomeXchange Consortium via the PRIDE partner repository75 with the identifier number PXD018002. NGS data have been deposited in the Gene Expression Omnibus database under the SuperSeries accession code GSE147015. The SubSeries are: RPE-1 4sU-seq (GSE147012), RPE-1 OGAP-seq (GSE147013), MRC5 NuMA ChIP–seq (GSE147014), SH-SY5Y OGAP-seq (GSE201981), RPE-1 Pol2 ChIP–seq (GSE201982), SH-SY5Y NuMA CUT&RUN (GSE201983), RPE-1 NuMA and TDP1 CUT&RUN (GSE201984). Figures that have associated raw data (gel or blot images), mass spectrometry and bioinformatics analysis include: Figs. 1b,f–h,j–l, 2a–d,f–g,i,k–m, 3a–g, 4b–g, 5a–h and 6a,d,f–i and Extended Data Figs. 1, 3a,c–j, 4a–d, 5b–e,i, 6b,c,h,i, 7a,d, 8b, 9b,c,e and 10h.
Code availability
The code for the pausing ratio calculator is available at https://github.com/jdparker101/Pausing_Ratio_Notebook.
References
Kooistra, S. M. & Helin, K. Molecular mechanisms and potential functions of histone demethylases. Nat. Rev. Mol. Cell Biol. 13, 297–311 (2012).
Xu, G.-L. & Wong, J. Oxidative DNA demethylation mediated by Tet enzymes. Natl Sci. Rev. 2, 318–328 (2015).
Margolin, Y., Cloutier, J.-F., Shafirovich, V., Geacintov, N. E. & Dedon, P. C. Paradoxical hotspots for guanine oxidation by a chemical mediator of inflammation. Nat. Chem. Biol. 2, 365–366 (2006).
Pan, L. et al. Oxidized guanine base lesions function in 8-oxoguanine DNA glycosylase-1-mediated epigenetic regulation of nuclear factor κB-driven gene expression. J. Biol. Chem. 291, 25553–25566 (2016).
Fleming, A. M., Ding, Y. & Burrows, C. J. Oxidative DNA damage is epigenetic by regulating gene transcription via base excision repair. Proc. Natl Acad. Sci. USA 114, 2604–2609 (2017).
El-Khamisy, S. F. et al. Defective DNA single-strand break repair in spinocerebellar ataxia with axonal neuropathy-1. Nature 434, 108–113 (2005).
Nilsen, L., Forstrøm, R. J., Bjørås, M. & Alseth, I. AP endonuclease independent repair of abasic sites in Schizosaccharomyces pombe. Nucleic Acids Res. 40, 2000–2009 (2012).
Interthal, H., Chen, H. J. & Champoux, J. J. Human Tdp1 cleaves a broad spectrum of substrates, including phosphoamide linkages. J. Biol. Chem. 280, 36518–36528 (2005).
Lebedeva, N. A., Rechkunova, N. I., El-Khamisy, S. F. & Lavrik, O. I. Tyrosyl-DNA phosphodiesterase 1 initiates repair of apurinic/apyrimidinic sites. Biochimie 94, 1749–1753 (2012).
Whitehouse, C. J. et al. XRCC1 stimulates human polynucleotide kinase activity at damaged DNA termini and accelerates DNA single-strand break repair. Cell 104, 107–117 (2001).
El-Khamisy, S. F., Masutani, M., Suzuki, H. & Caldecott, K. W. A requirement for PARP-1 for the assembly or stability of XRCC1 nuclear foci at sites of oxidative DNA damage. Nucleic Acids Res. 31, 5526–5533 (2003).
Das, B. B. et al. PARP1–TDP1 coupling for the repair of topoisomerase I-induced DNA damage. Nucleic Acids Res. 42, 4435–4449 (2014).
Awwad, S. W., Abu-Zhayia, E. R., Guttmann-Raviv, N. & Ayoub, N. NELF-E is recruited to DNA double-strand break sites to promote transcriptional repression and repair. EMBO Rep. 18, 745–764 (2017).
Hoch, N. C. et al. XRCC1 mutation is associated with PARP1 hyperactivation and cerebellar ataxia. Nature 541, 87–91 (2017).
Takashima, H. et al. Mutation of TDP1, encoding a topoisomerase I–dependent DNA damage repair enzyme, in spinocerebellar ataxia with axonal neuropathy. Nat. Genet. 32, 267–272 (2002).
Giampetruzzi, A. et al. Modulation of actin polymerization affects nucleocytoplasmic transport in multiple forms of amyotrophic lateral sclerosis. Nat. Commun. 10, 3827 (2019).
Maynard, S. et al. Lamin A/C promotes DNA base excision repair. Nucleic Acids Res. 47, 11709–11728 (2019).
Lydersen, B. K. & Pettijohn, D. E. Human-specific nuclear protein that associates with the polar region of the mitotic apparatus: distribution in a human/hamster hybrid cell. Cell 22, 489–499 (1980).
Hueschen, C. L., Kenny, S. J., Xu, K. & Dumont, S. NuMA recruits dynein activity to microtubule minus-ends at mitosis. eLife 6, e29328 (2017).
Merdes, A., Ramyar, K., Vechio, J. D. & Cleveland, D. W. A complex of NuMA and cytoplasmic dynein is essential for mitotic spindle assembly. Cell 87, 447–458 (1996).
Merdes, A. & Cleveland, D. W. The role of NuMA in the interphase nucleus. J. Cell Sci. 111, 71–79 (1998).
Vidi, P.-A. et al. NuMA promotes homologous recombination repair by regulating the accumulation of the ISWI ATPase SNF2h at DNA breaks. Nucleic Acids Res. 42, 6365–6379 (2014).
Salvador-Moreno, N. et al. The nuclear structural protein NuMA is a negative regulator of 53BP1 in DNA double-strand break repair. Nucleic Acids Res. 47, 2703–2715 (2019).
Serra-Marques, A. et al. The mitotic protein NuMA plays a spindle-independent role in nuclear formation and mechanics. J. Cell Biol. 219, e202004202 (2020).
Bahrami, S. & Drabløs, F. Gene regulation in the immediate–early response process. Adv. Biol. Regul. 62, 37–49 (2016).
Tullai, J. W. et al. Immediate–early and delayed primary response genes are distinct in function and genomic architecture. J. Biol. Chem. 282, 23981–23995 (2007).
Samson, L. D. A target to suppress inflammation. Science 362, 748–749 (2018).
Visnes, T. et al. Small-molecule inhibitor of OGG1 suppresses proinflammatory gene expression and inflammation. Science 362, 834–839 (2018).
Dellino, G. I. et al. Release of paused RNA polymerase II at specific loci favors DNA double-strand-break formation and promotes cancer translocations. Nat. Genet. 47, 1–21 (2019).
Boehler, C. et al. Poly(ADP-ribose) polymerase 3 (PARP3), a newcomer in cellular response to DNA damage and mitotic progression. Proc. Natl Acad. Sci. USA 108, 2783–2788 (2011).
Chang, P., Coughlin, M. & Mitchison, T. J. Tankyrase-1 polymerization of poly(ADP-ribose) is required for spindle structure and function. Nat. Cell Biol. 7, 1133–1139 (2005).
Haren, L. & Merdes, A. Direct binding of NuMA to tubulin is mediated by a novel sequence motif in the tail domain that bundles and stabilizes microtubules. J. Cell Sci. 115, 1815–1824 (2002).
Hendriks, I. A., Larsen, S. C. & Nielsen, M. L. An advanced strategy for comprehensive profiling of ADP-ribosylation sites using mass spectrometry-based proteomics. Mol. Cell Proteomics 18, 1010–1026 (2019).
Palazzo, L. et al. Serine is the major residue for ADP-ribosylation upon DNA damage. eLife 7, e34334 (2018).
Páhi, Z. G., Borsos, B. N., Pantazi, V., Ujfaludi, Z. & Pankotai, T. PARylation during transcription: insights into the fine-tuning mechanism and regulation. Cancers 12, 183 (2020).
Ting, X. et al. USP11 acts as a histone deubiquitinase functioning in chromatin reorganization during DNA repair. Nucleic Acids Res. 47, 9721–9740 (2019).
Kuznetsov, N. A. et al. Pre-steady state kinetics of DNA binding and abasic site hydrolysis by tyrosyl-DNA phosphodiesterase 1. J. Biomol. Struct. Dyn. 35, 2314–2327 (2017).
Poetsch, A. R., Boulton, S. J. & Luscombe, N. M. Genomic landscape of oxidative DNA damage and repair reveals regioselective protection from mutagenesis. Genome Biol. 19, 215 (2018).
Wu, J., McKeague, M. & Sturla, S. J. Nucleotide-resolution genome-wide mapping of oxidative DNA damage by Click-Code-Seq. J. Am. Chem. Soc. 140, 9783–9787 (2018).
Mao, P. et al. Genome-wide maps of alkylation damage, repair, and mutagenesis in yeast reveal mechanisms of mutational heterogeneity. Genome Res. 27, 1674–1684 (2017).
Reid, D. A. et al. Incorporation of a nucleoside analog maps genome repair sites in postmitotic human neurons. Science 372, 91–94 (2021).
Wu, W. et al. Neuronal enhancers are hotspots for DNA single-strand break repair. Nature 593, 440–444 (2021).
Radulescu, A. E. & Cleveland, D. W. NuMA after 30 years: the matrix revisited. Trends Cell Biol. 20, 214–222 (2010).
Ohata, H. et al. NuMA is required for the selective induction of p53 target genes. Mol. Cell. Biol. 33, 2447–2457 (2013).
Jayaraman, S. et al. The nuclear mitotic apparatus protein NuMA controls rDNA transcription and mediates the nucleolar stress response in a p53-independent manner. Nucleic Acids Res. 45, 11725–11742 (2017).
Chang, W., Dynek, J. N. & Smith, S. NuMA is a major acceptor of poly(ADP-ribosyl)ation by tankyrase 1 in mitosis. Biochem. J. 391, 177–184 (2005).
Altmeyer, M. et al. Liquid demixing of intrinsically disordered proteins is seeded by poly(ADP-ribose). Nat. Commun. 6, 8088 (2015).
Nair, S. J. et al. Phase separation of ligand-activated enhancers licenses cooperative chromosomal enhancer assembly. Nat. Struct. Mol. Biol. 26, 193–203 (2019).
Endo, A., Moyori, A., Kobayashi, A. & Wong, R. W. Nuclear mitotic apparatus protein, NuMA, modulates p53-mediated transcription in cancer cells. Cell Death Dis. 4, e713–e713 (2013).
Perera, D. et al. Differential DNA repair underlies mutation hotspots at active promoters in cancer genomes. Nature 532, 259–263 (2016).
Janssens, D. & Henikoff, S. CUT&RUN: targeted in situ genome-wide profiling with high efficiency for low cell numbers. protocols.io https://doi.org/10.17504/protocols.io.zcpf2vn (2019).
Liu, N. Library prep for CUT&RUN with NEBNext UltraTM II DNA Library Prep Kit for Illumina (E7645) V.1. protocols.io https://doi.org/10.17504/protocols.io.wvgfe3w (2019).
Cox, J. & Mann, M. MaxQuant enables high peptide identification rates, individualized p.p.b.-range mass accuracies and proteome-wide protein quantification. Nat. Biotechnol. 26, 1367–1372 (2008).
Tyanova, S. et al. The Perseus computational platform for comprehensive analysis of (prote)omics data. Nat. Methods 13, 731–740 (2016).
Li, D. et al. pFind: a novel database-searching software system for automated peptide and protein identification via tandem mass spectrometry. Bioinformatics 21, 3049–3050 (2005).
Wang, L. et al. pFind 2.0: a software package for peptide and protein identification via tandem mass spectrometry. Rapid Commun. Mass Spectrom. 21, 2985–2991 (2007).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Love, I. M., Anders, S. & Huber, W. Differential analysis of count data—the DESeq2 package. Genome Biol. 15, 550 (2014).
Patro, R., Duggal, G., Love, M. I., Irizarry, R. A. & Kingsford, C. Salmon provides fast and bias-aware quantification of transcript expression. Nat. Methods 14, 417–419 (2017).
Soneson, C., Love, M. I. & Robinson, M. D. Differential analyses for RNA-seq: transcript-level estimates improve gene-level inferences. F1000Res. 4, 1521 (2016).
Durinck, S., Spellman, P. T., Birney, E. & Huber, W. Mapping identifiers for the integration of genomic datasets with the R/Bioconductor package biomaRt. Nat. Protoc. 4, 1184–1191 (2009).
Giannakakis, A. et al. Contrasting expression patterns of coding and noncoding parts of the human genome upon oxidative stress. Sci Rep. 5, 9737 (2015).
Dellino, G. I. et al. Release of paused RNA polymerase II at specific loci favors DNA double-strand-break formation and promotes cancer translocations. Nat. Genet. 51, 1011–1023 (2019).
Quinlan, A. R. & Hall, I. M. BEDTools: a flexible suite of utilities for comparing genomic features. Bioinformatics 26, 841–842 (2010).
Sun, Z. & Chadwick, B. P. Loss of SETDB1 decompacts the inactive X chromosome in part through reactivation of an enhancer in the IL1RAPL1 gene. Epigenet. Chromatin 11, 45 (2018).
Henrich, K.-O. et al. Integrative genome-scale analysis identifies epigenetic mechanisms of transcriptional deregulation in unfavorable neuroblastomas. Cancer Res. 76, 5523–5537 (2016).
Li, H. & Durbin, R. Fast and accurate long-read alignment with Burrows–Wheeler transform. Bioinformatics 26, 589–595 (2010).
Sims, D. et al. CGAT: computational genomics analysis toolkit. Bioinformatics 30, 1290–1291 (2014).
Li, H. et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
Gaspar, J. M. Improved peak-calling with MACS2. Preprint at Biorxiv https://doi.org/10.1101/496521 (2018).
Zerbino, D. R. et al. Ensembl 2018. Nucleic Acids Res. 46, D754–D761 (2017).
Liao, Y., Smyth, G. K. & Shi, W. featureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 30, 923–930 (2014).
Hadley, W. ggplot2: Elegant Graphics for Data Analysis (Springer–Verlag, 2016).
Perez-Riverol, Y. et al. The PRIDE database and related tools and resources in 2019: improving support for quantification data. Nucleic Acids Res. 47, D442–D450 (2019).
Acknowledgements
We thank L. Ferraiuolo for advice on iPS cell-derived neurons, S. Dumont for providing the hTERT (RPE-1) cells containing the stably integrated spCas9 (Tet-On promoter) and NuMA sgRNA, the Sheffield Wolfson Light Microscopy facility for the imaging experiments and the Sheffield biOMICS Facility for the protein mass spectrometry experiments. This work was funded by a Wellcome Trust Investigator Award (103844) and a Lister Institute of Preventative Medicine Fellowship (137661) to S.F.E.-K.
Author information
Authors and Affiliations
Contributions
S.R. performed the protein interaction and in vitro transcription experiments. A.A.A. performed the PARylation experiments. S.R. and A.A.A. performed the imaging, DNA strand break repair, genome-wide occupancy and oxidative damage experiments. J.P. performed all the bioinformatics analyses with advice and support from I.S. K.L., N.M.P. and C.L. conducted comet assays, in vitro binding and ubiquitination. A.E.A.-M. performed and analysed the protein mass spectrometry experiments. C.D.S.S. generated the iPS cell-derived neurons. M.J. assisted with the comet assays and performed the cell cycle analysis. S.F.E.-K., S.R. and A.A.A wrote the manuscript. All authors edited the manuscript. S.F.E.-K. conceived the study, and led and managed the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature thanks Rami Aqeilan, Zhao-Qi Wang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Expression of SSBR genes in different brain regions.
Expression of TDP1, XRCC1 and PARP1 across different brain regions in the GTEx v8 dataset.
Extended Data Fig. 2 DRB treatment abolishes NuMA-dependent defect in SSBR.
siSCR and siNuMA-transfected MRC5 cells were pre-treated with DMSO or DRB for 2 h before H2O2 treatment and recovered for 30 and 60 min. Representative violin plots show the spread of comet tail moments from 150 nuclei, n = 3 biological replicates (two-sided, unpaired t-test).
Extended Data Fig. 3 NuMA promotes transcription following oxidative damage.
(a) Immunoblotting of lysates from RPE-1 cells for 4sU-Seq., n = 2 biological replicates. (b) A schematic for 4sU-Seq. (c) 4sU-Seq showing -log10 adjusted p-values plotted against Log2 fold change for genes differentially expressed in WT (+H2O2) versus (−H2O2) cells. (DEseq2, two-sided Wald test, Benjamini-Hochberg procedure adjusted p-values ≤ 0.05). Log2 fold changes with respect to WT (+H2O2) cells. (d) Overlap of paused genes with genes upregulated (left) and downregulated (right) in WT (+H2O2) versus (−H2O2) cells. (e) 4sU-Seq showing -log10 adjusted p-values against Log2 fold change for genes differentially expressed in KD versus WT (+H2O2) cells. (DEseq2, two-sided Wald test, Benjamini-Hochberg adjusted p-values ≤ 0.05). Log2 fold changes to KD cells. (f) Overlap of paused genes with genes upregulated (left) and downregulated (right) in KD versus WT (+H2O2) cells. (g) Overlap of genes containing fragile promoters (FPG) with NRGs. The background set is the total set of differentially expressed genes. (h) Distribution of average transcription levels in Log2 transcripts per million (TPM) for NRGs and genes differentially upregulated in WT(+H2O2) and are averaged across replicates. Only genes with TPM values ≥1 across WT(+H2O2) replicates were included in the analysis. In box plots, the centre line is the median, top and bottom hinges show the upper and lower quartiles, and the top and bottom whiskers indicate the largest and smallest values no further than 1.5 times interquartile range from the top and bottom hinges, respectively; two-sided Wilcoxon rank sum test. (i) Scatterplot of Log2 fold changes in transcription level in WT versus KD(+H2O2) cells against gene length in kb on a log10 scale. Correlation coefficient (R) by Spearman’s rank and significance using the asymptotic t approximation (two-sided). (j) Left; Fold change in nascent RNA transcripts for the indicated SSBR genes. Right; IGV snapshots of a representative track of the 4sU-Seq profiles of SSBR and DSBR genes.
Extended Data Fig. 4 Clustered proteins identified by mass spectrometry in GFP pull downs.
(a) HEK-293 cells transfected with GFP-empty or GFP-NuMA were treated with H2O2 and recovered for 10 min. Cell lysates were subjected to GFP-trap immunoprecipitation, on-bead trypsin digestion and subsequent analysis by mass spectrometry. Volcano plot showing quantitative enrichment of NuMA interacting proteins identified from statistical analysis of data sets using Student’s t-test. Solid lines indicate significant enrichment of interacting proteins after filtering for a false discovery rate of 0.05 and an artificial within groups variance S0 = 1. Components of the Pol2 and SSBR machineries are highlighted as red squares. (b) Heat map of clustered proteins identified by mass spectrometry in GFP pull downs. Hierarchical clustering of LFQ protein intensity shows two dominant sample clusters, GFP and GFP-NuMA-L (LTR), according to sample conditions. (c) HEK293 cells were treated with either DMSO or inhibitors of PARG (PDD00017273), PARP3 (ME0328) or PARP1 (Olaparib). Western immunoblotting was carried out to show poly-ADP ribosylation (PAR) levels, n = 3 biological replicates. (d) Heat map of clustered proteins identified by mass spectrometry in GFP pull downs. Hierarchical clustering of LFQ protein intensity shows three dominant sample clusters, GFP, GFP-NuMA-S (STR) and GFP-NuMA-L (LTR), according to sample conditions. On the right-hand side, zoomed heat map of NuMA-S (STR) and NuMA-L (LTR) intensity profiles across samples.
Extended Data Fig. 5 NuMA-L promotes SSBR and transcription recovery functions.
(a) A schematic of human NuMA isoforms. (b) GFP- immunoprecipitates from GFP-NuMA-L and NuMA-S-transfected HEK-293 cells, treated with H2O2 and subjected to mass spectrometric analysis. Annotated tandem mass spectra of NuMA-L (top) and NuMA-S (bottom) specific peptides were identified in the immunoprecipitates. Precursor mass deviation was 2.07 ppm for the peptide LTAQVEQLEVFQR and 0.66 ppm for the peptide LTAQVEELSK. (c) Quantitative enrichment of interacting proteins between NuMA-L and NuMA-S from (b). NuMA-interacting proteins were identified from statistical analysis using Student t-test. Solid lines indicate enriched interacting proteins after filtering for FDR of 0.05 and an artificial within groups variance, S0 = 1. Annotated blue and red dots indicate NuMA-S (STR), NuMA-L (LTR) and additional proteins of interest. Non-annotated pale red dots represent proteins significantly enriched in each NuMA pull down compared to GFP controls. Green dots are proteins enriched in both NuMA-S and NuMA-L pull-downs and red dots are proteins enriched only in NuMA-L pull-downs. (d) Immunoblotting of GFP- immunoprecipitates from GFP-empty, GFP-NuMA-L and GFP-NuMA-S-transfected HEK-293 cells, treated with H2O2, n = 3 biological replicates. (e) Immunoblotting of transfected MRC5 cells., n = 5 biological replicates. (f) MRC5 cells were transfected as in (e), treated with H2O2 and recovered in media. A representative plot showing spread of comet tail moments from 250 nuclei (two-sided, unpaired, t-test). (g) Bars represent percentage DNA strand breaks remaining. Error bars (±s.e.m,), n = 5 biological replicates (two-sided, unpaired t-test). (h) Clonogenic survival of MRC5 cells transfected as in (e) and presented on a semi-log scale, n = 3 biological replicates (two-sided, unpaired t-test). (i) Immunoblotting of transfected MRC5 cells. (j) MRC5 cells were transfected as in (i) and nascent transcripts labelled with EU. Scatter plots show fold change of average fluorescence. Error bars (±s.e.m.), n = 3 biological replicates (two-sided, unpaired t-test).
Extended Data Fig. 6 NuMA C-terminal tail or a mitotic separation-of-function mutant promotes SSBR.
(a) Schematic of the truncation mutants generated from NuMA-FL with the C-terminal tail (aa 1700-2115) designated as globular domain (GD) and the microtubule-binding domain (aa 1866-1936) as MD. (b) HEK293 cells were co-transfected with full-length or different truncations of NuMA as in (a), alongside myc-TDP1. GFP-immunoprecipitants were analysed by immunoblotting, n = 3 biological replicates. (c) Immunoblotting of MRC5 cells transfected with siSCR or siNuMA followed by complementation with targeting resistant GFP-tagged NuMA-FL, NuMA-GD, or EV. (d) Representative violin plot showing spread of comet tail moments from 150 MRC5 cells from 3 biological replicates transfected as in (c), post-H2O2 treatment and recovered at indicated time points (two-sided, unpaired t-test). (e) Bars represent percentage breaks remaining after removal of H2O2 and recovery in complete medium from (d). Error bars (±s.e.m), n = 3 biological replicates, (two-sided, unpaired t-test). (f) HEK293 cells transfected with either NuMA-FL or NuMA-MD followed by nocodazole treatment before harvest. Representative image showing NuMA (green), α-tubulin (red), nuclei (DAPI/blue), n = 6 biological replicates, Scale bar, 10 μm. (g) The percentage of cells with NuMA at the mitotic poles was quantified. Error bars (±s.e.m.), n = 100 cells from 6 biological replicates (two-sided, unpaired t-test). (h) Immunoblotting of GFP-immunoprecipitates from HEK-293 cells transfected with GFP-empty, NuMA-FL or NuMA-MD, alongside myc-TDP1, and treated with H2O2, n = 3 biological replicates. (i) Immunoblotting of lysates from MRC5 cells transfected with siSCR or siNuMA and complemented with targeting-resistant GFP-tagged NuMA-FL, NuMA-MD, or EV. (j) MRC5 cells were transfected as in (i), treated with H2O2 and recovered in media at the indicated time points. Representative violin plot showing spread of comet tail moments from 150 cells from 3 biological replicates (two-sided, unpaired t-test). (k) Bars represent percentage strand breaks remaining after removal of H2O2 and recovery in complete medium. Error bars (±s.e.m.), n = 3 biological replicates (two-sided, unpaired t-test).
Extended Data Fig. 7 NuMA promotes the enrichment of TDP1 at damaged chromatin.
(a) Top; The knockdown efficiency of NuMA was analysed by immunoblotting using anti-NuMA antibodies. Bottom; The expression levels of GFP–TDP1 was measured in cells used in photo-bleaching experiments at time 0 (sec). Error bars (± s.e.m.), n = 3 biological replicates. (b) MRC5 cells were plated onto glass-bottom dishes and co-transfected with GFP-TDP1 and siSCR or siNuMA. Cells were pre-incubated with DMSO or olaparib. Cells expressing similar total GFP signal were locally irradiated with an ultraviolet A laser (405 nm), and GFP–TDP1 accumulation at the site of damage was quantified for the indicated time points. Scale bar-5 µm. (c) Data are plotted as the average percentage fluorescence (arbitrary units) in micro-irradiated tracks ± s.e.m from ∼30 cells measured, n = 3 biological replicates (two-sided, unpaired t-test). (d) Lysates of MRC5 cells transfected with siSCR or siNuMA and siTDP1 either individually or in combination was analysed by immunoblotting, n = 3 biological replicates. (e) MRC5 cells transfected as in (d), treated with H2O2 and recovered at indicated time points. Representative violin plot showing spread of comet tail moments from 150 cells, n = 3 biological replicates. (f) Bars represent percentage breaks remaining after removal of H2O2 and recovery in complete medium. Error bars (±s.e.m.), n = 3 biological replicates (two-sided, unpaired t-test). (g) Survival of MRC5 cells transfected as in (d) was compared using the indicated doses of H2O2. Results are presented on a semi-log scale and represent the average of three biological replicates ± s.e.m. ns - not significant (two-sided, unpaired t-test).
Extended Data Fig. 8 NuMA interacts with PAF1.
(a) HEK-293 cells were transfected with plasmids encoding GFP or GFP-NuMA-L, treated with H2O2 and cell lysates analysed by mass spectrometry. Volcano plot showing quantitative enrichment of NuMA interacting proteins identified from statistical analysis of data sets using Student’s t-test. Solid lines indicate significant enrichment of interacting proteins after filtering for a false discovery rate of 0.05 and an artificial within groups variance S0 = 1. Components of the PAF1 complex are highlighted as blue squares. (b) HEK-293 cells were treated with H2O2 and fractionated into insoluble chromatin bound fraction and soluble nucleoplasmic fraction. Endogenous RNA Pol2 was immunoprecipitated using Pol2 pS5 antibody. Immunoprecipitated complexes were analysed by immunoblotting using antibodies against Pol2 pS5, NuMA and PAF1, n = 3 biological replicates.
Extended Data Fig. 9 NuMA deficiency increases 8-oxoG levels and decreases TDP1 occupancy at promoters, while oxidative stress increases NuMA and TDP1 availability at promoters.
(a) RPE-1 cells were either untreated, treated with H2O2 or KBrO3. Genomic DNA was extracted and the levels of 8-oxoG per 1,000,000 deoxyguanosine (dG) was quantified by HPLC-QQQ mass spectrometry. Error bars (± s.e.m.), n = 3 biological replicates, (two-sided, unpaired t-test) (b and c) Metaprofiles of fold change in counts per million over the TSS relative to IgG for (b) NuMA and (c) TDP1 CUT&RUN in RPE-1 cells. (d) RPE-1 cells were transfected with Flag-TDP1 and ChIP-qPCR was conducted at the promoters of FOS, CCN2 and SRF. Error bars (±s.e.m.), n = 3 biological replicates (two-sided, unpaired t-test). (e) Metaprofile of fold change in counts per million over enhancer regions relative to IgG for NuMA CUT&RUN in RPE-1 cells under unperturbed conditions. The regions flanking the enhancers are 2,500 bp.
Extended Data Fig. 10 NuMA-deficient neuronal cells exhibit defective SSBR and transcription recovery.
(a) Brightfield image showing differentiation of SH-SY5Y cells to neuronal morphology (similar to primary neurons with long processes) after growth in low-serum media supplemented with retinoic acid (RA). Scale bar, 100 µm. (b) Violin plots showing quantification of neurite outgrowth following RA differentiation, quantified using Fiji (ImageJ) software with NeuronJ plugin, n = 16/condition (two-sided, unpaired t-test). (c) MAP2 immunofluorescence images of undifferentiated and differentiated SH-SY5Y cells, n = 2 biological replicates, scale bar, 10 µm. (d) Representative violin plots showing spread of comet tail moments from 200 differentiated SH-SY5Y cells from four biological replicates immediately after or during recovery from H2O2 (two-sided, unpaired t-test). (e) Brightfield image showing human iPSC-derived motor neurons, n = 3 biological replicates, scale bar, 100 µm. (f) Representative immunofluorescence images of human shSCR or shNuMA-transduced iPSC-derived motor neurons showing relative levels of NuMA (green), n = 2 biological replicates, scale bar, 10 µm. (g) Violin plot showing NuMA fluorescence intensity n = 20 cells/condition (two-sided, unpaired t-test). (h) Immunoblot of lysates from shSCR and shNuMA-transduced neurons, n = 2 biological replicates. (i) Bars represent percentage breaks remaining after removal of H2O2 and recovery in complete medium, as quantified by the alkaline comet assay. Error bars represent the range, n = 2 biological replicates. (j) Representative violin plots showing spread of comet tail moments from 100 cells immediately after or during recovery from H2O2 (two-tailed, unpaired t-test) (k) shSCR or shNuMA-transduced motor neurons were treated with H2O2 and nascent transcripts were labelled by EU. Scatter plots shows fold change of average fluorescence. Error bars (±s.e.m.), n = 4 separate cover slips (two-sided, unpaired t-test). (l) PLA was performed on motor neurons that were either mock-treated or H2O2-treated. For each sample, a representative image, which is a merge of the red and blue channels, is shown. Violin plots show mean signal intensity of Pol2 pS5-NuMA complex/nuclei, n = 125 cells from three separate wells, (two-sided, unpaired t-test), scale bar, 10 µm.
Extended Data Fig. 11 Depicting the function of NuMA in response to oxidative stress.
(a) Distribution of PARP1/NuMA expression across brain tissues of the GTEx v8 dataset. (b) A schematic diagram depicting NuMA’s role in response to oxidative stress. NuMA is enriched at the promoters and interacts with RNA Polymerase 2 (Pol2) and the SSBR proteins, TDP1 and PARP1. As a result of the enrichment of this complex, cells are efficient at repairing oxidative damage at promoters. NuMA acts as a PAR sink, ensuring the physiological levels of Pol2 PARylation that takes place in the nucleoplasm and thus increases the availability of Pol2 at promoters and enhances its release from pausing. This results in increasing the transcription of NuMA regulated genes (NRGs) (c) Decreased levels of NuMA increases the oxidative damage at promoters. Moreover, the absence of NuMA increases Pol2 PARylation in the nucleoplasm, thereby decreasing Pol2 availability at promoters, resulting in a reduction of the transcription of NRGs.
Supplementary information
Supplementary Information
This file contains Supplementary Tables 1–6 for siRNA and gRNA sequences, antibodies, primers, plasmids, oligonucleotides and transition and retention times used for 8-oxoG mass spectrometry. It also contains uncropped images for gels and western blots, with the red boxes showing the cropped bands.
Supplementary Data 1
This file contains the complete list of proteins represented in the volcano plot GFP vs NuMA-L.
Supplementary Data 2
This file contains the complete list of proteins represented in the volcano plot NuMA-S vs NuMA-L.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ray, S., Abugable, A.A., Parker, J. et al. A mechanism for oxidative damage repair at gene regulatory elements. Nature 609, 1038–1047 (2022). https://doi.org/10.1038/s41586-022-05217-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-022-05217-8
This article is cited by
-
A comparative study of different antiviral treatment protocols in HCV related cryoglobulinemic vasculitis
Scientific Reports (2024)
-
NUMA1 modulates apoptosis of esophageal squamous cell carcinoma cells through regulating ASK1-JNK signaling pathway
Cellular and Molecular Life Sciences (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.