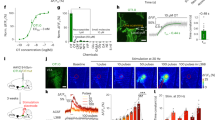
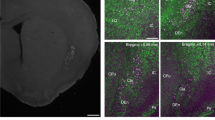
Abstract
Neuromodulatory control by oxytocin is essential to a wide range of social1,2, parental3 and stress-related behaviours4. Autism spectrum disorders (ASD) are associated with deficiencies in oxytocin levels5 and with genetic alterations of the oxytocin receptor (OXTR)6. Thirty years ago, Mühlethaler et al.7 found that oxytocin increases the firing of inhibitory hippocampal neurons, but it remains unclear how elevated inhibition could account for the ability of oxytocin to improve information processing in the brain. Here we describe in mammalian hippocampus a simple yet powerful mechanism by which oxytocin enhances cortical information transfer while simultaneously lowering background activity, thus greatly improving the signal-to-noise ratio. Increased fast-spiking interneuron activity not only suppresses spontaneous pyramidal cell firing, but also enhances the fidelity of spike transmission and sharpens spike timing. Use-dependent depression at the fast-spiking interneuron–pyramidal cell synapse is both necessary and sufficient for the enhanced spike throughput. We show the generality of this novel circuit mechanism by activation of fast-spiking interneurons with cholecystokinin or channelrhodopsin-2. This provides insight into how a diffusely delivered neuromodulator can improve the performance of neural circuitry that requires synapse specificity and millisecond precision.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Domes, G., Heinrichs, M., Michel, A., Berger, C. & Herpertz, S. C. Oxytocin improves “mind-reading” in humans. Biol. Psychiatry 61, 731–733 (2007)
Ferguson, J. N., Aldag, J. M., Insel, T. R. & Young, L. J. Oxytocin in the medial amygdala is essential for social recognition in the mouse. J. Neurosci. 21, 8278–8285 (2001)
Tomizawa, K. et al. Oxytocin improves long-lasting spatial memory during motherhood through MAP kinase cascade. Nature Neurosci. 6, 384–390 (2003)
Knobloch, H. S. et al. Evoked axonal oxytocin release in the central amygdala attenuates fear response. Neuron 73, 553–566 (2012)
Modahl, C. et al. Plasma oxytocin levels in autistic children. Biol. Psychiatry 43, 270–277 (1998)
Wu, S. et al. Positive association of the oxytocin receptor gene (OXTR) with autism in the Chinese Han population. Biol. Psychiatry 58, 74–77 (2005)
Mühlethaler, M., Charpak, S. & Dreifuss, J. J. Contrasting effects of neurohypophysial peptides on pyramidal and non-pyramidal neurones in the rat hippocampus. Brain Res. 308, 97–107 (1984)
Pouille, F. & Scanziani, M. Enforcement of temporal fidelity in pyramidal cells by somatic feed-forward inhibition. Science 293, 1159–1163 (2001)
Buzsáki, G. & Eidelberg, E. Commissural projection to the dentate gyrus of the rat: evidence for feed-forward inhibition. Brain Res. 230, 346–350 (1981)
Zaninetti, M. & Raggenbass, M. Oxytocin receptor agonists enhance inhibitory synaptic transmission in the rat hippocampus by activating interneurons in stratum pyramidale. Eur. J. Neurosci. 12, 3975–3984 (2000)
Földy, C., Lee, S. Y., Szabadics, J., Neu, A. & Soltesz, I. Cell type-specific gating of perisomatic inhibition by cholecystokinin. Nature Neurosci. 10, 1128–1130 (2007)
Wilson, R. I., Kunos, G. & Nicoll, R. A. Presynaptic specificity of endocannabinoid signaling in the hippocampus. Neuron 31, 453–462 (2001)
Yoshida, M. et al. Evidence that oxytocin exerts anxiolytic effects via oxytocin receptor expressed in serotonergic neurons in mice. J. Neurosci. 29, 2259–2271 (2009)
Alberi, S., Dreifuss, J. J. & Raggenbass, M. The oxytocin-induced inward current in vagal neurons of the rat is mediated by G protein activation but not by an increase in the intracellular calcium concentration. Eur. J. Neurosci. 9, 2605–2612 (1997)
Galarreta, M. & Hestrin, S. Frequency-dependent synaptic depression and the balance of excitation and inhibition in the neocortex. Nature Neurosci. 1, 587–594 (1998)
Kraushaar, U. & Jonas, P. Efficacy and stability of quantal GABA release at a hippocampal interneuron-principal neuron synapse. J. Neurosci. 20, 5594–5607 (2000)
Persico, A. M. & Bourgeron, T. Searching for ways out of the autism maze: genetic, epigenetic and environmental clues. Trends Neurosci. 29, 349–358 (2006)
Guastella, A. J. et al. Intranasal oxytocin improves emotion recognition for youth with autism spectrum disorders. Biol. Psychiatry 67, 692–694 (2010)
Anagnostou, E. et al. Intranasal oxytocin versus placebo in the treatment of adults with autism spectrum disorders: a randomized controlled trial. Mol. Autism 3, 16 (2012)
Gogolla, N. et al. Common circuit defect of excitatory-inhibitory balance in mouse models of autism. J. Neurodevelop. Disord. 1, 172–181 (2009)
Dinstein, I. et al. Unreliable evoked responses in autism. Neuron 75, 981–991 (2012)
Kinsley, C. H. et al. Motherhood improves learning and memory. Nature 401, 137–138 (1999)
Verret, L. et al. Inhibitory interneuron deficit links altered network activity and cognitive dysfunction in Alzheimer model. Cell 149, 708–721 (2012)
Lee, S. H. et al. Activation of specific interneurons improves V1 feature selectivity and visual perception. Nature 488, 379–383 (2012)
Glickfeld, L. L. & Scanziani, M. Distinct timing in the activity of cannabinoid-sensitive and cannabinoid-insensitive basket cells. Nature Neurosci. 9, 807–815 (2006)
Madison, D. V. & Nicoll, R. A. Actions of noradrenaline recorded intracellularly in rat hippocampal CA1 pyramidal neurones, in vitro. J. Physiol. (Lond.) 372, 221–244 (1986)
Segal, M. & Bloom, F. E. The action of norepinephrine in the rat hippocampus. IV. The effects of locus coeruleus stimulation on evoked hippocampal unit activity. Brain Res. 107, 513–525 (1976)
Kuo, S. P. & Trussell, L. O. Spontaneous spiking and synaptic depression underlie noradrenergic control of feed-forward inhibition. Neuron 71, 306–318 (2011)
Bergles, D. E., Doze, V. A., Madison, D. V. & Smith, S. J. Excitatory actions of norepinephrine on multiple classes of hippocampal CA1 interneurons. J. Neurosci. 16, 572–585 (1996)
Doze, V. A., Cohen, G. A. & Madison, D. V. Synaptic localization of adrenergic disinhibition in the rat hippocampus. Neuron 6, 889–900 (1991)
Atallah, B. V. & Scanziani, M. Instantaneous modulation of gamma oscillation frequency by balancing excitation with inhibition. Neuron 62, 566–577 (2009)
Freund, T. F. & Buzsaki, G. Interneurons of the hippocampus. Hippocampus 6, 347–470 (1996)
Shu, Y., Hasenstaub, A., Duque, A., Yu, Y. & McCormick, D. A. Modulation of intracortical synaptic potentials by presynaptic somatic membrane potential. Nature 441, 761–765 (2006)
Hippenmeyer, S. et al. A developmental switch in the response of DRG neurons to ETS transcription factor signaling. PLoS Biol. 3, e159 (2005)
Ascoli, G. A., Gasparini, S., Medinilla, V. & Migliore, M. Local control of postinhibitory rebound spiking in CA1 pyramidal neuron dendrites. J. Neurosci. 30, 6434–6442 (2010)
Hao, J., Wang, X. D., Dan, Y., Poo, M. M. & Zhang, X. H. An arithmetic rule for spatial summation of excitatory and inhibitory inputs in pyramidal neurons. Proc. Natl Acad. Sci. USA 106, 21906–21911 (2009)
Acknowledgements
We thank C. Földy for generous assistance with techniques for interneuron recording and classification, and thank Y. Li, M. Tadross, A. Mitra, D. Poburko, L. Prolo, J. Huguenard, S. Hestrin, D. Madison, L. Luo, K. Pelkey and K. Deisseroth for discussion and comments. This work was supported by the Burnett Family Fund, the Mosbacher Fund, and the Mathers Foundation, as well as grants from the National Institute of Mental Health (MH064070, MH071739), the National Institute of Neurological Disorders and Stroke (NS024067) and the Simons Foundation (95395). S.F.O. is supported by an NDSEG fellowship and by an NIMH-NRSA fellowship (F31MH084430). P.L.B. is supported by an SNSF and SFGBM fellowship (PASMP3_140063/1).
Author information
Authors and Affiliations
Contributions
S.F.O. performed all experiments, analysis and modelling. S.N.T. and S.F.O. performed ChR2 experiments. P.L.B. and N.N.T. assisted with interpretation of results and long-term project direction. R.W.T. and G.F. oversaw experiments, analysis, modelling and project direction. S.F.O. and R.W.T. planned and designed experiments and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Figures
This file contains Supplementary Figures 1-13. (PDF 3277 kb)
Rights and permissions
About this article
Cite this article
Owen, S., Tuncdemir, S., Bader, P. et al. Oxytocin enhances hippocampal spike transmission by modulating fast-spiking interneurons. Nature 500, 458–462 (2013). https://doi.org/10.1038/nature12330
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature12330
This article is cited by
-
Connectome dysfunction in patients at clinical high risk for psychosis and modulation by oxytocin
Molecular Psychiatry (2024)
-
Effects of single- and multiple-dose oxytocin treatment on amygdala low-frequency BOLD fluctuations and BOLD spectral dynamics in autism
Translational Psychiatry (2022)
-
Oxytocin-based therapies for treatment of Prader-Willi and Schaaf-Yang syndromes: evidence, disappointments, and future research strategies
Translational Psychiatry (2022)
-
Oxytocin via oxytocin receptor excites neurons in the endopiriform nucleus of juvenile mice
Scientific Reports (2022)
-
Gq neuromodulation of BLA parvalbumin interneurons induces burst firing and mediates fear-associated network and behavioral state transition in mice
Nature Communications (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.