Abstract
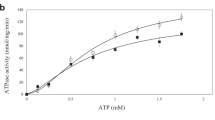
The energy-coupling factor (ECF) transporters constitute a novel family of conserved membrane transporters in prokaryotes that have a similar domain organization to the ATP-binding cassette transporters1,2,3. Each ECF transporter comprises a pair of cytosolic ATPases (the A and A′ components, or EcfA and EcfA′), a membrane-embedded substrate-binding protein (the S component, or EcfS) and a transmembrane energy-coupling component (the T component, or EcfT) that links the EcfA–EcfA′ subcomplex to EcfS. The structure and transport mechanism of the quaternary ECF transporter remain largely unknown. Here we report the crystal structure of a nucleotide-free ECF transporter from Lactobacillus brevis at a resolution of 3.5 Å. The T component has a horseshoe-shaped open architecture, with five α-helices as transmembrane segments and two cytoplasmic α-helices as coupling modules connecting to the A and A′ components. Strikingly, the S component, thought to be specific for hydroxymethyl pyrimidine, lies horizontally along the lipid membrane and is bound exclusively by the five transmembrane segments and the two cytoplasmic helices of the T component. These structural features suggest a plausible working model for the transport cycle of the ECF transporters.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Rodionov, D. A. et al. A novel class of modular transporters for vitamins in prokaryotes. J. Bacteriol. 191, 42–51 (2009)
Eitinger, T., Rodionov, D. A., Grote, M. & Schneider, E. Canonical and ECF-type ATP-binding cassette importers in prokaryotes: diversity in modular organization and cellular functions. FEMS Microbiol. Rev. 35, 3–67 (2011)
Erkens, G. B., Majsnerowska, M., Ter Beek, J. & Slotboom, D. J. Energy coupling factor-type ABC transporters for vitamin uptake in prokaryotes. Biochemistry 51, 4390–4396 (2012)
Rees, D. C., Johnson, E. & Lewinson, O. ABC transporters: the power to change. Nature Rev. Mol. Cell Biol. 10, 218–227 (2009)
Oldham, M. L., Khare, D., Quiocho, F. A., Davidson, A. L. & Chen, J. Crystal structure of a catalytic intermediate of the maltose transporter. Nature 450, 515–521 (2007)
Oldham, M. L. & Chen, J. Crystal structure of the maltose transporter in a pretranslocation intermediate state. Science 332, 1202–1205 (2011)
Oldham, M. L. & Chen, J. Snapshots of the maltose transporter during ATP hydrolysis. Proc. Natl Acad. Sci. USA 108, 15152–15156 (2011)
Locher, K. P., Lee, A. T. & Rees, D. C. The E. coli BtuCD structure: a framework for ABC transporter architecture and mechanism. Science 296, 1091–1098 (2002)
Pinkett, H. W., Lee, A. T., Lum, P., Locher, K. P. & Rees, D. C. An inward-facing conformation of a putative metal-chelate-type ABC transporter. Science 315, 373–377 (2007)
Hvorup, R. N. et al. Asymmetry in the structure of the ABC transporter-binding protein complex BtuCD-BtuF. Science 317, 1387–1390 (2007)
Dawson, R. J. & Locher, K. P. Structure of a bacterial multidrug ABC transporter. Nature 443, 180–185 (2006)
Dawson, R. J. & Locher, K. P. Structure of the multidrug ABC transporter Sav1866 from Staphylococcus aureus in complex with AMP-PNP. FEBS Lett. 581, 935–938 (2007)
Duurkens, R. H., Tol, M. B., Geertsma, E. R., Permentier, H. P. & Slotboom, D. J. Flavin binding to the high affinity riboflavin transporter RibU. J. Biol. Chem. 282, 10380–10386 (2007)
Hebbeln, P., Rodionov, D. A., Alfandega, A. & Eitinger, T. Biotin uptake in prokaryotes by solute transporters with an optional ATP-binding cassette-containing module. Proc. Natl Acad. Sci. USA 104, 2909–2914 (2007)
Erkens, G. B. & Slotboom, D. J. Biochemical characterization of ThiT from Lactococcus lactis: a thiamin transporter with picomolar substrate binding affinity. Biochemistry 49, 3203–3212 (2010)
Zhang, P., Wang, J. & Shi, Y. Structure and mechanism of the S component of a bacterial ECF transporter. Nature 468, 717–720 (2010)
Erkens, G. B. et al. The structural basis of modularity in ECF-type ABC transporters. Nature Struct. Mol. Biol. 18, 755–760 (2011)
Berntsson, R. P. et al. Structural divergence of paralogous S components from ECF-type ABC transporters. Proc. Natl Acad. Sci. USA 109, 13990–13995 (2012)
Holm, L. & Sander, C. Protein structure comparison by alignment of distance matrices. J. Mol. Biol. 233, 123–138 (1993)
Seyedarabi, A., Sullivan, J. A., Sasakawa, C. & Pickersgill, R. W. A disulfide driven domain swap switches off the activity of Shigella IpaH9.8 E3 ligase. FEBS Lett. 584, 4163–4168 (2010)
Neubauer, O. et al. Two essential arginine residues in the T components of energy-coupling factor transporters. J. Bacteriol. 191, 6482–6488 (2009)
Lezon, T. R. & Bahar, I. Constraints imposed by the membrane selectively guide the alternating access dynamics of the glutamate transporter GltPh. Biophys. J. 102, 1331–1340 (2012)
Otwinowski, Z. & Minor, W. Processing of X-ray diffraction data collected in oscillation mode. Methods Enzymol. 276, 307–326 (1997)
Schneider, T. R. & Sheldrick, G. M. Substructure solution with SHELXD. Acta Crystallogr. D 58, 1772–1779 (2002)
McCoy, A. et al. Phaser crystallographic software. J. Appl. Crystallogr. 40, 658–674 (2007)
Cowtan, K. & DM: an automated procedure for phase improvement by density modification. CCP4 Newslett. 31, 34–38 (1994)
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D 60, 2126–2132 (2004)
DeLaBarre, B. & Brunger, A. T. Considerations for the refinement of low-resolution crystal structures. Acta Crystallogr. D 62, 923–932 (2006)
Adams, P. D. et al. PHENIX: building new software for automated crystallographic structure determination. Acta Crystallogr. D 58, 1948–1954 (2002)
DeLano, W. L. The PyMOL Molecular Graphics System. http://www.pymol.org (2002)
Collaborative Computational Project, 4. The CCP4 suite: programs for protein crystallography. Acta Crystallogr. D 50, 760–763 (1994)
Humphrey, W., Dalke, A. & Schulten, K. VMD: visual molecular dynamics. J. Mol. Graph. 14, 33–38 (1996)
Acknowledgements
We thank S. Huang and J. He at SSRF beamline BL17U for on-site assistance. This work was supported by funds from the Ministry of Science and Technology (973 Programs 2009CB918801 and 2013CB910602) and the National Natural Science Foundation of China (31021002 and 31130002).
Author information
Authors and Affiliations
Contributions
T.W., G.F., X.G. and Y.S. designed all experiments. T.W., G.F., X.P., J. Wu, X.G. and J. Wa. performed the experiments. All authors contributed to data analysis. T.W., G.F., X.G., J. Wa. and Y.S. contributed to manuscript preparation. Y.S. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Table 1, Supplementary Figures 1-12 Supplementary References. (PDF 4106 kb)
Suggested directionality of ECF motion
This animation shows the simulated motion of ECF and gives a sense of the direction of movement by the ECF transporter. (MPG 3969 kb)
Rights and permissions
About this article
Cite this article
Wang, T., Fu, G., Pan, X. et al. Structure of a bacterial energy-coupling factor transporter. Nature 497, 272–276 (2013). https://doi.org/10.1038/nature12045
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature12045
This article is cited by
-
Identification of inhibitors targeting the energy-coupling factor (ECF) transporters
Communications Biology (2023)
-
Membrane mediated toppling mechanism of the folate energy coupling factor transporter
Nature Communications (2020)
-
Structure and mechanism of a group-I cobalt energy coupling factor transporter
Cell Research (2017)
-
Structural insight in the toppling mechanism of an energy-coupling factor transporter
Nature Communications (2016)
-
Structures of FolT in substrate-bound and substrate-released conformations reveal a gating mechanism for ECF transporters
Nature Communications (2015)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.