Abstract
Objective:
Mice deficient of the serotonin transporter (5-HTT ko) mice have a reduced brain serotonin content and develop late-onset obesity. To elucidate the pathophysiology of this obesity, we analyzed the expression of the interrelated weight-regulatory molecules: brain-derived neurotrophic factor (BDNF) and leptin receptor (LR) in brain areas associated with nutrition and activity.
Research Design and Methods:
We investigated feeding behavior, physical activity and metabolic parameters of 5-HTT ko and wild-type mice and measured the expression of BDNF and LR in brain areas associated with nutrition and activity using quantitative real-time PCR. The influence of age, gender and fasting was analyzed.
Results:
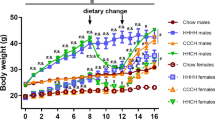
Male 5-HTT ko mice developed obesity without hyperphagia from the age of 5 months. Physical activity was reduced in old male, but not old female, 5-HTT ko mice. The BDNF gene expression in frontal cortex was elevated in young, but reduced in old 5-HTT ko mice. Fasting failed to increase the BDNF gene expression in frontal cortex of young 5 HTT ko mice and in the hypothalamus in old 5-HTT ko mice. The fasting-induced hypothalamic increase of LR was absent in both young and old 5-HTT ko mice.
Conclusions:
We propose that low brain serotonin level due to the 5-HTT ko genotype leads to reduced physical activity and low BDNF, which together with the lack of fasting-induced hypothalamic BDNF and LR production results in late-onset obesity. Although lack of the 5-HTT is a genetic vulnerability factor for obesity, female gender is protective.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Kopelman PG . Obesity as a medical problem. Nature 2000; 404: 635–643.
Kaminski T, Smolinska N, Gajewska A, Siawrys G, Okrasa S, Kochman K et al. Leptin and long form of leptin receptor genes expression in the hypothalamus and pituitary during the luteal phase and early pregnancy in pigs. J Physiol Pharmacol 2006; 57: 95–108.
Konturek SJ, Konturek JW, Pawlik T, Brzozowski T . Brain–gut axis and its role in the control of food intake. J Physiol Pharmacol 2004; 55: 137–154.
Wallenius V, Wallenius K, Ahren B, Rudling M, Carlsten H, Dickson SL et al. Interleukin-6-deficient mice develop mature-onset obesity. Nat Med 2002; 8: 75–79.
Simansky KJ . Serotonergic control of the organization of feeding and satiety. Behav Brain Res 1996; 73: 37–42.
Lebrun B, Bariohay B, Moyse E, Jean A . Brain-derived neurotrophic factor (BDNF) and food intake regulation: a minireview. Auton Neurosci 2006; 126–127: 30–38.
Carroll L, Voisey J, van Daal A . Mouse models of obesity. Clin Dermatol 2004; 22: 345–349.
Coscina DV, Balagura S . Avoidance and escape behavior of rats with aphagia produced by basal diencephalic lesions. Physiol Behav 1970; 5: 651–657.
Pollock JD, Rowland N . Peripherally administered serotonin decreases food intake in rats. Pharmacol Biochem Behav 1981; 15: 179–183.
Kim DK, Tolliver TJ, Huang SJ, Martin BJ, Andrews AM, Wichems C et al. Altered serotonin synthesis, turnover and dynamic regulation in multiple brain regions of mice lacking the serotonin transporter. Neuropharmacology 2005; 49: 798–810.
Bengel D, Murphy DL, Andrews AM, Wichems CH, Feltner D, Heils A et al. Altered brain serotonin homeostasis and locomotor insensitivity to 3, 4-methylenedioxymethamphetamine (‘Ecstasy’) in serotonin transporter-deficient mice. Mol Pharmacol 1998; 53: 649–655.
Xu B, Goulding EH, Zang K, Cepoi D, Cone RD, Jones KR et al. Brain-derived neurotrophic factor regulates energy balance downstream of melanocortin-4 receptor. Nat Neurosci 2003; 6: 736–742.
Madhav TR, Pei Q, Zetterstrom TS . Serotonergic cells of the rat raphe nuclei express mRNA of tyrosine kinase B (trkB), the high-affinity receptor for brain derived neurotrophic factor (BDNF). Brain Res Mol Brain Res 2001; 93: 56–63.
Mattson MP, Maudsley S, Martin B . BDNF and 5-HT: a dynamic duo in age-related neuronal plasticity and neurodegenerative disorders. Trends Neurosci 2004; 27: 589–594.
Ren-Patterson RF, Cochran LW, Holmes A, Lesch KP, Lu B, Murphy DL . Gender-dependent modulation of brain monoamines and anxiety-like behaviors in mice with genetic serotonin transporter and BDNF deficiencies. Cell Mol Neurobiol 2006; 26: 755–780.
Collin M, Hakansson-Ovesjo ML, Misane I, Ogren SO, Meister B . Decreased 5-HT transporter mRNA in neurons of the dorsal raphe nucleus and behavioral depression in the obese leptin-deficient ob/ob mouse. Brain Res Mol Brain Res 2000; 81: 51–61.
Chomczynski P, Sacchi N . Single-step method of RNA isolation by acid guanidinium thiocyanate–phenol–chloroform extraction. Anal Biochem 1987; 162: 156–159.
George A, Buehl A, Sommer C . Wallerian degeneration after crush injury of rat sciatic nerve increases endo- and epineurial tumor necrosis factor-alpha protein. Neurosci Lett 2004; 372: 215–219.
Lin S, Huang XF . Fasting increases leptin receptor mRNA expression in lean but not obese (ob/ob) mouse brain. Neuroreport 1997; 8: 3625–3629.
Duan W, Lee J, Guo Z, Mattson MP . Dietary restriction stimulates BDNF production in the brain and thereby protects neurons against excitotoxic injury. J Mol Neurosci 2001; 16: 1–12.
Trotter NL . Electron-opaque bodies and fat droplets in mouse liver after fasting or glucose injection. J Cell Biol 1967; 34: 703–711.
Kernie SG, Liebl DJ, Parada LF . BDNF regulates eating behavior and locomotor activity in mice. EMBO J 2000; 19: 1290–1300.
Lyons WE, Mamounas LA, Ricaurte GA, Coppola V, Reid SW, Bora SH et al. Brain-derived neurotrophic factor-deficient mice develop aggressiveness and hyperphagia in conjunction with brain serotonergic abnormalities. Proc Natl Acad Sci USA 1999; 96: 15239–15244.
Nonogaki K, Strack AM, Dallman MF, Tecott LH . Leptin-independent hyperphagia and type 2 diabetes in mice with a mutated serotonin 5-HT2C receptor gene. Nat Med 1998; 4: 1152–1156.
Tecott LH, Sun LM, Akana SF, Strack AM, Lowenstein DH, Dallman MF et al. Eating disorder and epilepsy in mice lacking 5-HT2c serotonin receptors. Nature 1995; 374: 542–546.
Yamauchi T, Kamon J, Waki H, Terauchi Y, Kubota N, Hara K et al. The fat-derived hormone adiponectin reverses insulin resistance associated with both lipoatrophy and obesity. Nat Med 2001; 7: 941–946.
Qu Y, Villacreses N, Murphy DL, Rapoport SI . 5-HT2A/2C receptor signaling via phospholipase A2 and arachidonic acid is attenuated in mice lacking the serotonin reuptake transporter. Psychopharmacology (Berl) 2005; 180: 12–20.
Rioux A, Fabre V, Lesch KP, Moessner R, Murphy DL, Lanfumey L et al. Adaptive changes of serotonin 5-HT2A receptors in mice lacking the serotonin transporter. Neurosci Lett 1999; 262: 113–116.
Uchida-Kitajima S, Yamauchi T, Takashina Y, Okada-Iwabu M, Iwabu M, Ueki K et al. 5-Hydroxytryptamine 2A receptor signaling cascade modulates adiponectin and plasminogen activator inhibitor 1 expression in adipose tissue. FEBS Lett 2008; 582: 3037–3044.
Nomura S, Shouzu A, Omoto S, Nishikawa M, Iwasaka T . 5-HT2A receptor antagonist increases circulating adiponectin in patients with type 2 diabetes. Blood Coagul Fibrinolysis 2005; 16: 423–428.
Dourish CT, Hutson PH, Curzon G . Low doses of the putative serotonin agonist 8-hydroxy-2-(di-n-propylamino) tetralin (8-OH-DPAT) elicit feeding in the rat. Psychopharmacology (Berl) 1985; 86: 197–204.
Kennett GA, Ainsworth K, Trail B, Blackburn TP . BW 723C86, a 5-HT2B receptor agonist, causes hyperphagia and reduced grooming in rats. Neuropharmacology 1997; 36: 233–239.
Simansky KJ, Nicklous DM . Parabrachial infusion of D-fenfluramine reduces food intake. Blockade by the 5-HT(1B) antagonist SB-216641. Pharmacol Biochem Behav 2002; 71: 681–690.
Mathews TA, Fedele DE, Coppelli FM, Avila AM, Murphy DL, Andrews AM . Gene dose-dependent alterations in extraneuronal serotonin but not dopamine in mice with reduced serotonin transporter expression. J Neurosci Methods 2004; 140: 169–181.
Fabre V, Beaufour C, Evrard A, Rioux A, Hanoun N, Lesch KP et al. Altered expression and functions of serotonin 5-HT1A and 5-HT1B receptors in knock-out mice lacking the 5-HT transporter. Eur J Neurosci 2000; 12: 2299–2310.
Dringenberg HC, Hargreaves EL, Baker GB, Cooley RK, Vanderwolf CH . p-chlorophenylalanine-induced serotonin depletion: reduction in exploratory locomotion but no obvious sensory-motor deficits. Behav Brain Res 1995; 68: 229–237.
Muldoon MF, Mackey RH, Williams KV, Korytkowski MT, Flory JD, Manuck SB . Low central nervous system serotonergic responsivity is associated with the metabolic syndrome and physical inactivity. J Clin Endocrinol Metab 2004; 89: 266–271.
Kotz CM, Billington CJ, Levine AS . Obesity and aging. Clin Geriatr Med 1999; 15: 391–412.
Olivier JD, Cools AR, Olivier B, Homberg JR, Cuppen E, Ellenbroek BA . Stress-induced hyperthermia and basal body temperature are mediated by different 5-HT(1A) receptor populations: a study in SERT knockout rats. Eur J Pharmacol 2008; 590: 190–197.
Ono M, Ichihara J, Nonomura T, Itakura Y, Taiji M, Nakayama C et al. Brain-derived neurotrophic factor reduces blood glucose level in obese diabetic mice but not in normal mice. Biochem Biophys Res Commun 1997; 238: 633–637.
Neeper SA, Gomez-Pinilla F, Choi J, Cotman CW . Physical activity increases mRNA for brain-derived neurotrophic factor and nerve growth factor in rat brain. Brain Res 1996; 726: 49–56.
Yamamoto T, Shibata Y . Direct fiber connections between the frontal cortex and the hypothalamus in the rat: an electron microscope study. Pharmacol Biochem Behav 1975; 3: 15–22.
Molteni R, Wu A, Vaynman S, Ying Z, Barnard RJ, Gomez-Pinilla F . Exercise reverses the harmful effects of consumption of a high-fat diet on synaptic and behavioral plasticity associated to the action of brain-derived neurotrophic factor. Neuroscience 2004; 123: 429–440.
Celada P, Siuciak JA, Tran TM, Altar CA, Tepper JM . Local infusion of brain-derived neurotrophic factor modifies the firing pattern of dorsal raphe serotonergic neurons. Brain Res 1996; 712: 293–298.
Ahima RS, Prabakaran D, Mantzoros C, Qu D, Lowell B, Maratos-Flier E et al. Role of leptin in the neuroendocrine response to fasting. Nature 1996; 382: 250–252.
Ellacott KL, Cone RD . The central melanocortin system and the integration of short- and long-term regulators of energy homeostasis. Recent Prog Hormone Res 2004; 59: 395–408.
King BM . The rise, fall, and resurrection of the ventromedial hypothalamus in the regulation of feeding behavior and body weight. Physiol Behav 2006; 87: 221–244.
Hisadome K, Smith MA, Choudhury AI, Claret M, Withers DJ, Ashford ML . 5-HT inhibition of rat insulin 2 promoter Cre recombinase transgene and proopiomelanocortin neuron excitability in the mouse arcuate nucleus. Neuroscience 2009; 159: 83–93.
Nonogaki K, Nozue K, Oka Y . Hyperphagia alters expression of hypothalamic 5-HT2C and 5-HT1B receptor genes and plasma des-acyl ghrelin levels in Ay mice. Endocrinology 2006; 147: 5893–5900.
Kimm SY, Glynn NW, Obarzanek E, Kriska AM, Daniels SR, Barton BA et al. Relation between the changes in physical activity and body-mass index during adolescence: a multicentre longitudinal study. Lancet 2005; 366: 301–307.
Frayling TM, Timpson NJ, Weedon MN, Zeggini E, Freathy RM, Lindgren CM et al. A common variant in the FTO gene is associated with body mass index and predisposes to childhood and adult obesity, 2007; 316: 889–894.
Herbeth B, Aubry E, Fumeron F, Aubert R, Cailotto F, Siest G et al. Polymorphism of the 5-HT2A receptor gene and food intakes in children and adolescents: the Stanislas Family Study. Am J Clin Nutr 2005; 82: 467–470.
Sookoian S, Gemma C, Garcia SI, Gianotti TF, Dieuzeide G, Roussos A et al. Short allele of serotonin transporter gene promoter is a risk factor for obesity in adolescents. Obesity 2007; 15: 271–276.
Beckers S, Peeters A, Zegers D, Mertens I, Van Gaal L, Van Hul W . Association of the BDNF Val66Met variation with obesity in women. Mol Genet Metab 2008; 95: 110–112.
Acknowledgements
We thank L Biko, H Brünner, B Dekant, B Gado and S Mildner for technical assistance and A Spahn for help with the statistical assessments. The study was supported by Deutsche Forschungsgemeinschaft (Le629/4–2 and SFB 581/B9 to K.P.L.) and by intramural funds of the Universities of Würzburg and Lübeck.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Üçeyler, N., Schütt, M., Palm, F. et al. Lack of the serotonin transporter in mice reduces locomotor activity and leads to gender-dependent late onset obesity. Int J Obes 34, 701–711 (2010). https://doi.org/10.1038/ijo.2009.289
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2009.289
Keywords
This article is cited by
-
Novel Antidepressant-Like Properties of the Iron Chelator Deferiprone in a Mouse Model of Depression
Neurotherapeutics (2022)
-
Serotonin transporter gene promoter methylation status correlates with in vivo prefrontal 5-HTT availability and reward function in human obesity
Translational Psychiatry (2017)
-
Central serotonin transporter availability in highly obese individuals compared with non-obese controls: A [11C] DASB positron emission tomography study
European Journal of Nuclear Medicine and Molecular Imaging (2016)
-
Long-Term Consumption of Fish Oil-Enriched Diet Impairs Serotonin Hypophagia in Rats
Cellular and Molecular Neurobiology (2010)