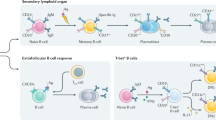
Abstract
Systemic lupus erythematosus (SLE) is a systemic autoimmune disease that is clinically heterogeneous and affects multiple organs. Lupus nephritis is the most frequent severe manifestation of SLE. Conventional immunosuppressive therapy has increased the life expectancy of patients diagnosed with lupus nephritis, but only 70–80% of patients respond to this treatment and its adverse effects are considerable. B cells are central to the pathogenesis of SLE and are, therefore, an attractive therapeutic target. B-cell depletion has been used successfully to treat other autoimmune diseases, such as rheumatoid arthritis and antineutrophil cytoplasmic antibody-associated vasculitis, and many case reports and small nonrandomized trials of B-cell-depleting agents in patients with lupus nephritis have reported positive results. By contrast, two large placebo-controlled trials designed to investigate the efficacy of the B-cell-depleting agents rituximab and ocrelizumab as a treatment for lupus nephritis, failed to meet their primary efficacy end points (LUNAR and BELONG, respectively). This Review discusses the current evidence on the use of B-cell depletion in the treatment of lupus nephritis, which is derived from case studies and clinical trials including a total of over 800 patients.
Key Points
-
Over 20% of patients with lupus nephritis do not respond (or respond poorly) to standard induction therapy using cyclophosphamide or mycophenolate mofetil
-
Approximately two-thirds of patients with refractory or relapsing lupus nephritis respond to rituximab; complete B-cell depletion confers a favourable outcome, whereas relapse is related to B-cell reconstitution
-
Many adult patients (median 20%, range 5–73%) will relapse 1–4 years after B-cell depletion, but patients who relapse respond to further courses of rituximab
-
Rituximab therapy is safe and well tolerated in adult patients with lupus nephritis; the most common adverse events are mild to moderate infusion reactions and infections
-
2–6% of adults with lupus nephritis have disease symptoms that are resistant to multiple therapies, including rituximab, which emphasizes the need for new therapeutic agents
-
Alternative B-cell-depleting monoclonal antibodies, such as anti-CD19 and anti-CD22 antibodies (epratuzumab), and anticytokine therapies (belimumab and atacicept), await evaluation in patients with lupus nephritis
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Rahman, A. & Isenberg, D. A. Systemic lupus erythematosus. N. Engl. J. Med. 358, 929–939 (2008).
Bagavant, H. & Fu, S. M. Pathogenesis of kidney disease in systemic lupus erythematosus. Curr. Opin. Rheumatol. 21, 489–494 (2009).
Manzi, S. et al. Age-specific incidence rates of myocardial infarction and angina in women with systemic lupus erythematosus: comparison with the Framingham study. Am. J. Epidemiol. 145, 408–415 (1997).
Björnådal, L., Yin, L., Granath, F., Klareskog, L. & Ekbom, A. Cardiovascular disease a hazard despite improved prognosis in patients with systemic lupus erythematosus: results from a Swedish population based study 1964–1995. J. Rheumatol. 31, 713–719 (2004).
Gourley, M. F. et al. Methylprednisolone and cyclophosphamide, alone or in combination, in patients with lupus nephritis. A randomized, controlled trial. Ann. Intern. Med. 125, 549–557 (1996).
Flanc, R. S. et al. Treatment of diffuse proliferative lupus nephritis: a meta-analysis of randomized controlled trials. Am. J. Kidney Dis. 43, 197–208 (2004).
Dey, I. D. & Isenberg, D. A. Systematic review and meta-analysis of the association of systemic lupus erythematosus and malignancy [abstract]. Arthritis Rheum. 63, 2283 (2011).
Cameron, J. S. Lupus nephritis: an historical perspective 1968–1998. J. Nephrol. 12, S29–S41 (1999).
Christopher-Stine, L. et al. Renal biopsy in lupus patients with low levels of proteinuria. J. Rheumatol. 34, 332–335 (2007).
Weening, J. J. et al. The classification of glomerulonephritis in systemic lupus erythematosus revisited. J. Am. Soc. Nephrol. 15, 241–250 (2004).
Borchers, A. T., Keen, C. L., Shoenfeld, Y. & Gershwin, M. E. Surviving the butterfly and the wolf: mortality trends in systemic lupus erythematosus. Autoimmun. Rev. 3, 423–453 (2004).
Cervera, R. et al. Morbidity and mortality in systemic lupus erythematosus during a 10-year period: a comparison of early and late manifestations in a cohort of 1,000 patients. Medicine (Baltimore) 82, 299–308 (2003).
Tesar, V. & Hruskova, Z. Treatment of proliferative lupus nephritis: a slowly changing landscape. Nat. Rev. Nephrol. 7, 96–109 (2011).
Houssiau, F. A. et al. Immunosuppressive therapy in lupus nephritis: the Euro-Lupus nephritis trial, a randomized trial of low-dose versus high-dose intravenous cyclophosphamide. Arthritis Rheum. 46, 2121–2131 (2002).
Ginzler, E. M. et al. Mycophenolate mofetil or intravenous cyclophosphamide for lupus nephritis. N. Engl. J. Med. 353, 2219–2228 (2005).
Chan, T. M. et al. Efficacy of mycophenolate mofetil in patients with diffuse proliferative lupus nephritis. Hong Kong-Guangzhou nephrology study group. N. Engl. J. Med. 343, 1156–1162 (2000).
Appel, G. B. et al. Mycophenolate mofetil versus cyclophosphamide for induction treatment of lupus nephritis. J. Am. Soc. Nephrol. 20, 1103–1112 (2009).
Chan, T. M., Tse, K. C., Tang, C. S., Mok, M. Y. & Li, F. K. Long-term study of mycophenolate mofetil as continuous induction and maintenance treatment for diffuse proliferative lupus nephritis. J. Am. Soc. Nephrol. 16, 1076–1084 (2005).
Ciruelo, E., de la Cruz, J., López, I. & Gómez Reino, J. J. Cumulative rate of relapse of lupus nephritis after successful treatment with cyclophosphamide. Arthritis Rheum. 39, 2028–2034 (1996).
Moroni, G. et al. The long-term outcome of 93 patients with proliferative lupus nephritis. Nephrol. Dial. Transplant. 22, 2531–2539 (2007).
Mosca, M. et al. Renal flares in 91 SLE patients with diffuse proliferative glomerulonephritis. Kidney Int. 61, 1502–1509 (2002).
Illei, G. G. et al. Renal flares are common in patients with severe proliferative lupus nephritis treated with pulse immunosuppressive therapy: long-term follow-up of a cohort of 145 patients participating in randomized controlled studies. Arthritis Rheum. 46, 995–1002 (2002).
Houssiau, F. A. et al. The 10-year follow-up data of the Euro-Lupus nephritis trial comparing low-dose and high-dose intravenous cyclophosphamide. Ann. Rheum. Dis. 69, 61–64 (2010).
Clatworthy, M. R. & Smith, K. G. B cells in glomerulonephritis: focus on lupus nephritis. Semin. Immunopathol. 29, 337–353 (2007).
Arce-Salinas, C. A., Rodríguez-García, F. & Gómez-Vargas, J. I. Long-term efficacy of anti-CD20 antibodies in refractory lupus nephritis. Rheumatol. Int. 32, 1245–1249 (2012).
Jónsdóttir, T., Sundelin, B., Welin Henriksson, E., van Vollenhoven, R. F. & Gunnarsson, I. Rituximab-treated membranous lupus nephritis: clinical outcome and effects on electron dense deposits. Ann. Rheum. Dis. 70, 1172–1173 (2011).
Pepper, R. et al. Rituximab is an effective treatment for lupus nephritis and allows a reduction in maintenance steroids. Nephrol. Dial. Transplant. 24, 3717–3723 (2009).
Boletis, J. N. et al. Rituximab and mycophenolate mofetil for relapsing proliferative lupus nephritis: a long-term prospective study. Nephrol. Dial. Transplant. 24, 2157–2160 (2009).
Melander, C. et al. Rituximab in severe lupus nephritis: early B-cell depletion affects long-term renal outcome. Clin. J. Am. Soc. Nephrol. 4, 579–587 (2009).
Li, E. K. et al. Is combination rituximab with cyclophosphamide better than rituximab alone in the treatment of lupus nephritis? Rheumatology (Oxford) 48, 892–898 (2009).
Gunnarsson, I. et al. Histopathologic and clinical outcome of rituximab treatment in patients with cyclophosphamide-resistant proliferative lupus nephritis. Arthritis Rheum. 56, 1263–1272 (2007).
Vigna-Perez, M. et al. Clinical and immunological effects of rituximab in patients with lupus nephritis refractory to conventional therapy: a pilot study. Arthritis Res. Ther. 8, R83 (2006).
Sfikakis, P. P. et al. Remission of proliferative lupus nephritis following B cell depletion therapy is preceded by down-regulation of the T cell costimulatory molecule CD40 ligand: an open-label trial. Arthritis Rheum. 52, 501–513 (2005).
Vital, E. M. et al. B cell biomarkers of rituximab responses in systemic lupus erythematosus. Arthritis Rheum. 63, 3038–3047 (2011).
Roccatello, D. et al. Intensive short-term treatment with rituximab, cyclophosphamide and methylprednisolone pulses induces remission in severe cases of SLE with nephritis and avoids further immunosuppressive maintenance therapy. Nephrol. Dial. Transplant. 26, 3987–3992 (2011).
Terrier, B. et al. Safety and efficacy of rituximab in systemic lupus erythematosus: results from 136 patients from the French autoimmunity and rituximab registry. Arthritis Rheum. 62, 2458–2466 (2010).
Catapano, F., Chaudhry, A. N., Jones, R. B., Smith, K. G. & Jayne, D. W. Long-term efficacy and safety of rituximab in refractory and relapsing systemic lupus erythematosus. Nephrol. Dial. Transplant. 25, 3586–3592 (2010).
Garcia-Carrasco, M. et al. Anti-CD20 therapy in patients with refractory systemic lupus erythematosus: a longitudinal analysis of 52 Hispanic patients. Lupus 19, 213–219 (2010).
Lateef, A., Lahiri, M., Teng, G. G. & Vasoo, S. Use of rituximab in the treatment of refractory systemic lupus erythematosus: Singapore experience. Lupus 19, 765–770 (2010).
Lu, T. Y. et al. A retrospective seven-year analysis of the use of B cell depletion therapy in systemic lupus erythematosus at University College London Hospital: the first fifty patients. Arthritis Rheum. 61, 482–487 (2009).
Lindholm, C. et al. Longterm clinical and immunological effects of anti-CD20 treatment in patients with refractory systemic lupus erythematosus. J. Rheumatol. 35, 826–833 (2008).
Jónsdóttir, T. et al. Treatment of refractory SLE with rituximab plus cyclophosphamide: clinical effects, serological changes, and predictors of response. Ann. Rheum. Dis. 67, 330–334 (2008).
Tanaka, Y. et al. A multicenter phase I/II trial of rituximab for refractory systemic lupus erythematosus. Mod. Rheumatol. 17, 191–197 (2007).
Leandro, M. J., Cambridge, G., Edwards, J. C., Ehrenstein, M. R. & Isenberg, D. A. B-cell depletion in the treatment of patients with systemic lupus erythematosus: a longitudinal analysis of 24 patients. Rheumatology (Oxford) 44, 1542–1545 (2005).
Looney, R. J. et al. B cell depletion as a novel treatment for systemic lupus erythematosus: a phase I/II dose-escalation trial of rituximab. Arthritis Rheum. 50, 2580–2589 (2004).
Rovin, B. H. et al. Efficacy and safety of rituximab in patients with active proliferative lupus nephritis: the lupus nephritis assessment with rituximab (LUNAR) study. Arthritis Rheum. 64, 1215–1226 (2012).
Mysler, E. F. et al. Efficacy and safety of ocrelizumab, a humanized antiCD20 antibody, in patients with active proliferative lupus nephritis (LN): results from the randomized, double-blind phase III BELONG study [abstract]. Arthritis Rheum. 62, 1455 (2010).
Bomback, A. S. & Appel, G. B. Updates on the treatment of lupus nephritis. J. Am. Soc. Nephrol. 21, 2028–2035 (2010).
Rezvani, A. R. & Maloney, D. G. Rituximab resistance. Best Pract. Res. Clin. Haematol. 24, 203–216 (2011).
Genovese, M. C. et al. Ocrelizumab, a humanized anti-CD20 monoclonal antibody, in the treatment of patients with rheumatoid arthritis: a phase I/II randomized, blinded, placebo-controlled, dose-ranging study. Arthritis Rheum. 58, 2652–2661 (2008).
Morschhauser, F. et al. Results of a phase I/II study of ocrelizumab, a fully humanized anti-CD20 mAb, in patients with relapsed/refractory follicular lymphoma. Ann. Oncol. 21, 1870–1876 (2010).
Rigby, W. et al. Safety and efficacy of ocrelizumab in patients with rheumatoid arthritis and an inadequate response to methotrexate: results of a forty-eight-week randomized, double-blind, placebo-controlled, parallel-group phase III trial. Arthritis Rheum. 64, 350–359 (2012).
US National library of Medicine. Study of the safety of MDX-1342 in combination with methotrexate in patients with rheumatoid arthritis (MDX1342–1301). Clinicaltrials.gov [online].
Traczewski, P. & Rudnicka, L. Treatment of systemic lupus erythematosus with epratuzumab. Br. J. Clin. Pharmacol. 71, 175–182 (2011).
Dörner, T. et al. Initial clinical trial of epratuzumab (humanized anti-CD22 antibody) for immunotherapy of systemic lupus erythematosus. Arthritis Res. Ther. 8, R74 (2006).
Jacobi, A. M. et al. Differential effects of epratuzumab on peripheral blood B cells of patients with systemic lupus erythematosus versus normal controls. Ann. Rheum. Dis. 67, 450–457 (2008).
Leonard, J. P. et al. Durable complete responses from therapy with combined epratuzumab and rituximab: final results from an international multicenter, phase 2 study in recurrent, indolent, non-Hodgkin lymphoma. Cancer 113, 2714–2723 (2008).
Moisini, I. & Davidson, A. BAFF: a local and systemic target in autoimmune diseases. Clin. Exp. Immunol. 158, 155–163 (2009).
Navarra, S. V. et al. Efficacy and safety of belimumab in patients with active systemic lupus erythematosus: a randomised, placebo-controlled, phase 3 trial. Lancet 377, 721–731 (2011).
Dooley, M. A. et al. Effect of belimumab treatment on renal outcomes: results from phase 3 belimumab clinical trials in patients with systemic lupus erythematosus [abstract]. Arthritis Rheum. 63, 2472 (2011)
Pena-Rossi, C. et al. An exploratory dose-escalating study investigating the safety, tolerability, pharmacokinetics and pharmacodynamics of intravenous atacicept in patients with systemic lupus erythematosus. Lupus 18, 547–555 (2009).
US National library of Medicine. Atacicept phase II/III in generalized systemic lupus erythematosus (April SLE). Clinicaltrials.gov [online].
Jónsdóttir, T. et al. Clinical improvements in proliferative vs membranous lupus nephritis following B-cell depletion: pooled data from two cohorts. Rheumatology (Oxford) 49, 1502–1504 (2010).
Díaz-Lagares, C. et al. The UK-BIOGEAS Registry. Efficacy of rituximab in 164 patients with biopsy-proven lupus nephritis: pooled data from European cohorts. Autoimmun. Rev. 11, 357–364 (2012).
Dooley, M. A. et al. Mycophenolate versus azathioprine as maintenance therapy for lupus nephritis. N. Engl. J. Med. 365, 1886–1895 (2011).
Houssiau, F. A. et al. Azathioprine versus mycophenolate mofetil for long-term immunosuppression in lupus nephritis: results from the MAINTAIN nephritis trial. Ann. Rheum. Dis. 69, 2083–2089 (2010).
Contreras, G. et al. Sequential therapies for proliferative lupus nephritis. N. Engl. J. Med. 350, 971–980 (2004).
Laskari, K., Mavragani, C. P., Tzioufas, A. G. & Moutsopoulos, H. M. Mycophenolate mofetil as maintenance therapy for proliferative lupus nephritis: a long-term observational prospective study. Arthritis Res. Ther. 12, R208 (2010).
Smith, K. G. & Clatworthy, M. R. FcgammaRIIB in autoimmunity and infection: evolutionary and therapeutic implications. Nat. Rev. Immunol. 10, 328–343 (2010).
Niederer, H. A., Clatworthy, M. R., Willcocks, L. C. & Smith, K. G. FcgammaRIIB, FcgammaRIIIB, and systemic lupus erythematosus. Ann. NY Acad. Sci. 1183, 69–88 (2010).
Lim, S. H. et al. Fc gamma receptor IIb on target B cells promotes rituximab internalization and reduces clinical efficacy. Blood 118, 2530–2540 (2011).
Turner-Stokes, T. et al. The efficacy of repeated treatment with B-cell depletion therapy in systemic lupus erythematosus: an evaluation. Rheumatology (Oxford) 50, 1401–1408 (2011).
Calabrese, L. H., Molloy, E. S., Huang, D. & Ransohoff, R. M. Progressive multifocal leukoencephalopathy in rheumatic diseases: evolving clinical and pathologic patterns of disease. Arthritis Rheum. 56, 2116–2128 (2007).
Tak, P. P. et al. Safety and efficacy of ocrelizumab in patients with rheumatoid arthritis and an inadequate response to at least one tumor necrosis factor inhibitor: results of a forty-eight-week randomized, double-blind, placebo-controlled, parallel-group phase III trial. Arthritis Rheum. 64, 360–370 (2012).
Stohl, W. et al. Safety and efficacy of ocrelizumab in combination with methotrexate in MTX-naive subjects with rheumatoid arthritis: the phase III FILM trial. Ann. Rheum. Dis. doi:10.1136/annrheumdis-2011-200706.
Petri, M. et al. Randomized controlled trials of epratuzumab (anti-CD22 mAb targeting B cells) reveal clinically meaningful improvements in patients with moderate/severe SLE flares [abstract]. Ann. Rheum. Dis. 67, 53 (2008).
Wallace, D. et al. Randomized controlled trials of epratuzumab (anti-CD22 mAb targeting B cells) reveal clinically meaningful reductions in corticosteroid use with favorable safety profile in moderate and severe flaring SLE patients [abstract]. Ann. Rheum. Dis. 67, 212 (2008).
Strand, V. et al. Randomized controlled trials of epratuzumab (anti-CD22 mAb targeting B cells) reveal clinically meaningful improvements in health related quality of life in SLE patients with high disease activity and low baseline self-report measures [abstract]. Ann. Rheum. Dis. 67, 212 (2008).
Wallace, D. J. et al. Epratuzumab demonstrates clinically meaningful improvements in patients with moderate to severe systemic lupus erythematosus (SLE): results from EMBLEM, a phase IIb study [abstract]. Arthritis Rheum. 62, 1452 (2010).
Kalunian, K. C. et al. BILAG-measured improvement in moderately and severely affected body systems in patients with systemic lupus erythematosus (SLE) by epratuzumab: results from EMBLEM, a phase IIb study [abstract]. Arthritis Rheum. 62, 453 (2010).
US National library of Medicine. Study of epratuzumab in systemic lupus erythematosus. Clinicaltrials.gov [online].
Rhee, E. P., Laliberte, K. A. & Niles, J. L. Rituximab as maintenance therapy for anti-neutrophil cytoplasmic antibody-associated vasculitis. Clin. J. Am. Soc. Nephrol. 5, 1394–1400 (2010).
Author information
Authors and Affiliations
Contributions
J. W. Gregersen researched the data for the article. D. R. W. Jayne contributed to discussions of the content, writing the article and to review and/or editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
D. R. W. Jayne has received research grants and consultancy fees from Baxter, Biogen, GlaxoSmithKline, Genzyme, Merck Serono and Roche/Genentech, and research grants from Vifor Pharma. J. W. Gregersen declares no competing interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Gregersen, J., Jayne, D. B-cell depletion in the treatment of lupus nephritis. Nat Rev Nephrol 8, 505–514 (2012). https://doi.org/10.1038/nrneph.2012.141
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneph.2012.141
This article is cited by
-
Muskelschwäche behandeln, Krisen vorbeugen
NeuroTransmitter (2024)
-
Therapeutic efficacy of anti-CD19 CAR-T cells in a mouse model of systemic lupus erythematosus
Cellular & Molecular Immunology (2021)
-
MicroRNA-146a-deficient mice develop immune complex glomerulonephritis
Scientific Reports (2019)
-
Hormonal Modulation of Dendritic Cells Differentiation, Maturation and Function: Implications for the Initiation and Progress of Systemic Autoimmunity
Archivum Immunologiae et Therapiae Experimentalis (2017)
-
Sex-specific effects of LiCl treatment on preservation of renal function and extended life-span in murine models of SLE: perspective on insights into the potential basis for survivorship in NZB/W female mice
Biology of Sex Differences (2016)