Abstract
Background:
Between 2009 and 2012, there was a worldwide shortage of agalsidase-β for the treatment of Fabry disease. Therefore, alternative treatments were needed, including switching to a different enzyme-replacement therapy.
Purpose:
This is an ongoing observational study assessing the effects of switching from agalsidase-β (1.0 mg/kg every other week) to agalsidase-α (0.2 mg/kg every other week) in 11 patients with Fabry disease.
Methods:
Clinical data were collected for 5 years—2 years before switching and 3 years after switching.
Results:
Measures of renal function such as estimated glomerular filtration rate remained stable during the 3 years after switching to agalsidase-α. Improvements in cardiac mass were recorded in both male and female patients 12 months after switching to agalsidase-α, and the benefit was maintained during 36 months of follow-up. There was no significant difference in the severity of pain experienced by patients before and after switching enzyme-replacement therapy, and no difference in quality-of-life parameters. Agalsidase-α was generally well tolerated, and no patients experienced allergy or developed antibodies to agalsidase-α.
Conclusion:
This observational study supports the safety of switching from agalsidase-β to agalsidase-α at the approved doses, with no loss of efficacy. It also suggests that if an infusion-related allergic reaction occurs in a patient receiving agalsidase-β, switching to agalsidase-α may be a viable option.
Genet Med 16 10, 766–772.
Similar content being viewed by others
Introduction
Fabry disease is a rare metabolic storage disorder that results in progressive effects on multiple organs; clinical manifestations include pain from nerve damage, gastrointestinal symptoms, angiokeratoma, corneal changes, and hearing loss.1,2 Over time, more serious manifestations develop, including renal dysfunction, cardiac disorders such as cardiomyopathy and arrhythmias, and cerebrovascular events such as strokes, which all contribute to a reduced life expectancy.1,2 The treatment options for patients with Fabry disease include long-term enzyme-replacement therapy (ERT) in addition to supportive management.1 ERT using recombinant human α-galactosidase A is intended to reduce disease severity and delay the progression of the disorder.3,4 Two preparations for treatment are available, namely, agalsidase-α (Replagal) and agalsidase-β (Fabrazyme), which are produced using different methods and administered at different doses.3
Both preparations of α-galactosidase A have been shown to be effective in patients with Fabry disease,3,5,6 but there are very few data from direct clinical comparisons of the two preparations. Findings from the ongoing Canadian Fabry Disease Initiative study suggest that when the preparations are administered at their approved dosages, there are no differences between them in terms of clinical outcomes during the first few years of treatment.7 However, the two preparations do differ in several ways. Agalsidase-α is produced using cultured human skin fibroblasts, whereas agalsidase-β is produced by the expression of human α-galactosidase A cDNA in Chinese hamster ovary cells.3 Although they have similar amino acid composition and biochemical properties, different patterns of posttranslational glycosylation of mannose residues have been noted, which alters the uptake of these preparations into tissues and could lead to differences in antigenicity.8,9,10,11 Furthermore, the recommended dosages for the two preparations differ: agalsidase-α is administered every other week at a dose of 0.2 mg/kg intravenously over a 40-min period, whereas the approved dosage for agalsidase-β is 1.0 mg/kg over a period of 1–2 h every other week.
Between 2009 and 2012, viral contamination in the production process of agalsidase-β led to a global shortage of this particular formulation. When it became clear that the shortage of agalsidase-β would not be resolved quickly, some patients who were already receiving agalsidase-β were treated with a reduced dosage so as to preserve the supplies of agalsidase-β. However, in countries where agalsidase-α was available, patients were given the option to switch to the alternative form of ERT at the recommended dosage. It was important to monitor both the “reduced dosage” and the “switch” patients to ensure that the patients were responding adequately to the altered treatment.
The European Medicines Agency raised concerns about an increase in adverse events in patients receiving reduced dosages of agalsidase-β,12,13 and consensus recommendations from a group of experts subsequently indicated that only the approved doses of enzyme-replacement therapies should be used.12,13,14 Switching patients to agalsidase-α remained an option.12,13 However, to date, few data have been reported on the outcome of switching patients between these two treatments.
In Japan, patients with Fabry disease started being switched from agalsidase-β to agalsidase-α in 2009. An ongoing observational study is following 11 patients who were switched, in order to assess the clinical course of patients and the safety of switching from agalsidase-β to agalsidase-α. Preliminary results after 12 months of follow-up suggested that disease stability was maintained after switching.15 We now report long-term data on these patients after ~3 years of follow-up.
Materials and Methods
Full details of the methods used for this ongoing observational longitudinal single-center study have been published previously.15 A brief summary of the design and methodology used for this case series is included here. The study included 11 patients with Fabry disease being treated at Nagoya Central Hospital who had their ERT switched from agalsidase-β (Fabrazyme) to agalsidase-α (Replagal) during November 2009, when constraints on the supply of agalsidase-β started. Earlier, the patients had been receiving ERT with agalsidase-β at the approved dose of 1.0 mg/kg every other week, and the switch was performed without any interval. Patients were switched to agalsidase-α at the approved dose of 0.2 mg/kg every other week, which was administered intravenously over 40–60 min. Before switching, all 11 patients had visited the clinic at biweekly intervals and were fully compliant with agalsidase-β ERT.
The study period covered 5 years—2 years before switching and 3 years after switching. Data were obtained from the patients’ medical records, with the information for the period before enrollment collected retrospectively. Approval was obtained from the institutional review board of Nagoya Central Hospital prior to initiating the study, and the study was conducted in accordance with the Declaration of Helsinki and applicable local laws and regulations. Patients provided written informed consent before inclusion into the study.
Hemizygous males or heterozygous females with a confirmed diagnosis of Fabry disease were eligible for inclusion if they were being treated with agalsidase-β at the recommended dosage for Fabry disease, which had been diagnosed using Japanese diagnostic criteria including reduced α-galactosidase A activity, genotyping, family history, abnormal globotriaosylceramide (Gb3) deposits in biopsy samples, and increased urinary Gb3 excretion. In addition, their medical records were required to include adequate information relevant to their Fabry disease. Patients were excluded if they had received concomitant agalsidase-α before the switch, if the switch took place before November 2009, or if the switch was made for any reason other than the worldwide shortage of agalsidase-β. Patients were required to receive agalsidase-α at the recommended dosage for at least 12 months after the switch from agalsidase-β.
Data collected for the period when patients were receiving agalsidase-β included the following: patient demographics (at the time of the switch), medical history, diagnosis of Fabry disease, vital signs, standard laboratory data (biochemistry, hematology, and urinalysis), plasma and urinary Gb3 levels, renal function (serum and urinary creatinine levels and/or estimated glomerular filtration rate (eGFR)), cardiac mass (left ventricular mass index (LVM index)) obtained by echocardiography, brain natriuretic peptide (BNP) levels, Fabry disease–related pain and quality of life (QoL), and safety/tolerability data (including adverse events and titers for anti–agalsidase-α and β antibodies).
Prospective data collected immediately before switching to agalsidase-α (baseline) and at 6-month intervals thereafter included the following: plasma Gb3 and lyso-Gb3 levels, renal function (eGFR, using the Chronic Kidney Disease Epidemiology Collaboration equation),16 proteinuria (urinary protein-to-creatinine ratio), cardiac mass (LVM adjusted for height using the Devereux formula),17,18 BNP levels, pain scores using the Brief Pain Inventory questionnaire (average and worst scores),19 QoL (using EuroQoL dimensions),20 the severity of Fabry disease signs and symptoms using the Mainz Severity Score Index.21 In addition, the safety and tolerability of agalsidase-α was evaluated by monitoring adverse events, concomitant drug use, laboratory tests, vital signs, antibody production, and the use of premedication for infusion reactions.
Only limited statistical analyses were performed because of the small number of patients. Data obtained 6, 12, 18, 24, and 36 months after switching to agalsidase-α were compared with preswitch data (when patients were on agalsidase-β) using one-sample Wilcoxon tests or paired t-tests. P values <0.05 were considered statistically significant.
Results
Thirteen patients with Fabry disease were receiving agalsidase-β prior to the supply problems. Two of the 13 patients on agalsidase-β chose to receive a reduced dosage and were ineligible. Thus, 11 patients were eligible for this observational study and comprised 4 men with classic hemizygous Fabry disease and 7 women with heterozygous-type disease. The mean age of the patients at the time of the switch was 47.3 years. The patients had been receiving agalsidase-β for up to 6 years prior to switching to agalsidase-α. Demographic and preswitch data are summarized in Table 1 . Preliminary data up to 12 months after the switch have been reported in detail previously.15 These preliminary results showed that there were no statistically or clinically significant changes in clinical parameters, with the exception of an improvement in LVM index compared with those in the preswitch period.15 All 11 patients have now completed ~3 years of therapy with agalsidase-α, and updated data covering the 36-month postswitch period are presented here. Using Mainz Severity Score Index scores as a measure of the severity of Fabry disease, we found that all the patients had mild-to-moderate Fabry disease at baseline (Mainz Severity Score Index: ≤40) and that the disease remained stable throughout the course of the 3-year follow-up period after switching to agalsidase-α.
Cardiac status
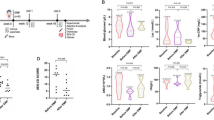
The improvements reported for cardiac parameters 12 months after switching to agalsidase-α15 were maintained throughout the 3-year treatment period. For example, compared with the preswitch value (58.1 g/m2.7), mean LVM index decreased significantly at 6 and 12 months after switching, and the value remained significantly reduced (50.7 g/m2.7; P = 0.0451; paired t-test) after 36 months. Individual values for mean interventricular septal wall diameter, left ventricular posterior wall diameter, and ejection fraction are also shown in Figure 1 . Mean interventricular septal wall diameter decreased significantly from the preswitch value at 6, 12, 24, and 36 months after switching; at 36 months, the mean thickness was 10.8 mm compared with 12.1 mm before switching (P = 0.0426). The mean reduction in left ventricular posterior wall diameter was statistically significant at 12 months after switching. After 36 months, at which point the mean reduction in left ventricular posterior wall diameter was 11.5 mm compared with the preswitch value of 12.3 mm, the difference was significant (P = 0.00236; paired t-test). In addition, the mean ejection fraction before and after the switch was well within the normal limits for our institution (50–88%); the ejection fraction was 72.1% at the time of switching to agalsidase-α and 65.8% after 36 months (P = 0.0340; paired t-test). The benefits recorded for the various cardiac parameters were seen in both male and female patients ( Figure 1 ), although the differences were not always statistically significant in these subgroups because of the small number of patients involved.
There were no notable changes in electrocardiogram parameters after switching from agalsidase-β to agalsidase-α in 10 patients. One patient with classic-type Fabry disease (Case 2) developed atrial fibrillation. The atrial fibrillation resolved spontaneously within ~6 months, and it is not known whether the onset of the arrhythmia was associated with the natural course of Fabry disease or was triggered by the switch in medication. This case of atrial fibrillation was the only notable medical event, and no patient died or developed end-stage renal disease or stroke or required hospitalization for a cardiac event.
The blood level of BNP worsened in one patient (Case 2) after the switch to agalsidase-α; however, the levels tended to improve in six other patients (Cases 1, 5, 6, 9, 10, and 11). Overall, representative BNP levels (determined by calculating an average of values obtained at baseline and at each time point) remained relatively stable after the switch to agalsidase-α ( Figure 2a ). The overall trend was for BNP levels to increase during agalsidase-β therapy and decrease during agalsidase-α therapy. A similar pattern was observed in patients with a baseline BNP level >100 pg/ml ( Figure 2b ).
Renal function
All indexes of renal function remained stable after switching from agalsidase-β to agalsidase-α. In particular, eGFR, which had been stable during the 24 months prior to the switch, did not change significantly during the 36 months after switching ( Figure 3 ). There were also no significant differences in the number of patients with stages 1/2/3 renal dysfunction: 5/5/1 (−24m), 4/7/0 (−12m), 6/4/1 (at switch), 5/5/1 (+12m), 4/6/1 (+24m), and 8/3/0 (+36m), respectively. In addition, there were no significant changes in urine levels of protein, creatinine, protein/creatinine ratio (Supplementary Figure S1 online), or N-acetyl-β-d-glucosaminidase, or in blood and urine levels of β2-microglobulin during the postswitch period.
Pain and QoL
There was no significant difference in the severity of pain experienced by patients before and after switching therapy and no difference in QoL parameters.
Globotriaosylceramide level
Plasma Gb3 level gradually decreased during treatment with agalsidase-β, and no significant increase was observed after switching to agalsidase-α ( Figure 4a ). There was also no significant change in the level of globotriaosylsphingosine (lyso-Gb3) after switching ( Figure 4b ).
Individual Gb3 and lyso-Gb3 levels. Gb3 (a) and lyso-Gb3 (b) levels before and after switching from agalsidase-β to agalsidase-α (key for individual case colors as in Figure 1 ).
Safety and tolerability
There were no indications of drug-related liver injury; no clinically significant abnormalities in bilirubin or in hepatic enzymes, including aspartate transaminase, alanine aminotransferase, and γ-glutamyl transferase, were observed during treatment with either agalsidase-β or agalsidase-α.
One male patient (Case 1) had experienced a treatment-related allergic reaction to agalsidase-β, which manifested as an aggravation of allergic rhinitis with nasal discharge during administration of the infusion. This reaction disappeared after the patient was switched to agalsidase-α. No allergic reactions were seen in the other 10 patients for either preparation.
In addition to an allergic reaction, the patient designated as Case 1 had also developed a positive test for antibodies against agalsidase-β. Tests for antibodies against agalsidase-α were negative before switching in all patients, including Case 1, and remained negative when retested up to 36 months after switching.
Discussion
The global shortage of agalsidase-β had led to many patients with Fabry disease being switched to agalsidase-α, the alternative α-galactosidase A preparation. Little information is available on the effect of switching between these products, in terms of efficacy and safety.22 In our center, we have been following 11 patients who were switched to agalsidase-α in November 2009 in order to assess any clinical changes that occurred after the switch. We have previously reported preliminary data after 1 year of treatment with agalsidase-α, at which time no detrimental effects related to the switch were noted. Indeed, the main change observed was an improvement in LVM index.15
Once ERT has commenced, life-long treatment is usual, and it was considered important to continue monitoring our group of patients over a long term to confirm the efficacy and safety of switching therapy. These 11 patients have now received ~3 years of treatment with agalsidase-α, and the results suggest that patients with Fabry disease receiving agalsidase-β can be safely switched to agalsidase-α with no loss of efficacy.
A retrospective observational study in the Netherlands looked at outcomes in 35 patients with Fabry disease who were receiving treatment with agalsidase-β at the time the shortage started.23 Two patients switched immediately to agalsidase-α, 18 initially received a reduced dose of agalsidase-β and later switched to agalsidase-α, and 15 continued on a reduced dose of agalsidase-β. The median duration of agalsidase-α treatment after switching was 0.9 years (range: 0.5–1.4 years) in the first two groups. There were no significant differences in the incidence of clinical events before and during the shortage. An increase in lyso-Gb3 was seen during the shortage, which the authors considered more likely to be related to the dose rather than the actual preparation being received.23 More recently, Pisani and colleagues24 in Italy reported results very similar to ours in a group of 10 patients who switched from agalsidase-β to agalsidase-α and who were followed for at least 20 months. eGFR remained stable during ERT and cardiac mass decreased significantly during treatment with agalsidase-β, and this benefit was maintained during ERT with agalsidase-α. Symptoms of pain and health-status scores did not deteriorate during agalsidase-α treatment; again, this is similar to the findings in the present study.
Our observational study involving 11 patients in Japan who were all switched to agalsidase-α has now been under way for ~3 years. An initial review of the data for the first year after switching was encouraging. Compared with the preswitch period, patients remained stable, based on eGFR, pain, and QoL assessments, while improvements in LV parameters were recorded, albeit in a relatively small number of patients.15 In addition, plasma Gb3 and lyso-Gb3 levels remained stable after switching to agalsidase-α and showed no evidence of increasing up to 36 months after the switch. Plasma lyso-Gb3 is a hallmark of Fabry disease, and an increase in its level could indicate a reduction in the efficacy of ERT.25
The current analysis confirms and extends these findings. Of note, the improvement in LVMI seen at 1 year appears to have largely been maintained after the additional 2 years of follow-up. In addition, mean ejection fraction, which was slightly elevated during the preswitch period, in conjunction with the left ventricular hypertrophy, decreased to within the normal range after the switch and remained stable up to 3 years. These improvements in LV function were also supported by blood levels of BNP, which tended to decrease after switching to agalsidase-α. BNP levels have been shown to be powerful markers for the prognosis and risk stratification in the setting of heart failure,26 and they are also predictive of prognosis in patients with predialysis chronic kidney disease.27 Cardiac involvement is common in Fabry disease and contributes to a reduced QoL and an increased risk of premature mortality.4 These data confirm that there was no deterioration in cardiac function after switching therapy because of the shortage of agalsidase-β and suggest that if for any reason this form of the enzyme cannot be used (lack of availability, poor tolerability, allergy, etc.), then switching to agalsidase-α is a viable therapeutic option. Renal data over the 3-year postswitch period showed that there was no deterioration in renal function compared with the preswitch period, a finding consistent with the 1-year results.15 Progressive renal dysfunction is a key factor associated with premature mortality in patients with Fabry disease, particularly men, and it is important that treatment should stabilize renal function.4 There were also no differences in pain severity or in QoL at 3 years after switching, compared with the period before switching.
Agalsidase-α was generally well tolerated during the 3-year period of assessment. None of the 11 patients developed a positive test for antibodies to agalsidase-α, including one patient who had previously developed antibodies to agalsidase-β prior to the switch. This patient had experienced an allergic reaction to agalsidase-β, which took the form of allergic rhinitis during administration of the infusion, but he did not develop an infusion-related reaction to agalsidase-α. Crossreactivity against agalsidase-α and -β has been observed for IgG antibodies28; however, an IgE antibody response has only been reported for agalsidase-β, and so IgE crossreactivity has not been reported to date.29 A switch to agalsidase-α may be appropriate for some patients who experience an infusion-related allergic reaction to agalsidase-β.
Our study has a number of limitations that need to be considered. Perhaps most importantly, Fabry disease is rare and we only had a small number of patients who switched from agalsidase-β to agalsidase-α, and therefore the results of the statistical analyses should be interpreted with caution. Nonetheless, the data obtained at 3 years after the switch support the preliminary results after 1 year. Given the limited amount of data available in the literature about the effect of switching from one formulation to another, this single-center study provides some useful longitudinal information for physicians managing patients who have had their therapy switched from agalsidase-β. In addition, our findings are consistent with results reported by two European groups.23,24 This should provide some reassurance to physicians and their patients who were switched to agalsidase-α.
In conclusion, after 3 years of follow-up, this study suggests that patients with Fabry disease receiving agalsidase-β, including those who experience infusion-related allergic reactions, can be safely switched to agalsidase-α with no loss of efficacy.
Disclosure
The authors declare no conflict of interest.
References
Germain DP . Fabry disease. Orphanet J Rare Dis 2010;5:30.
Mehta A, Beck M, Eyskens F, et al. Fabry disease: a review of current management strategies. QJM 2010;103:641–659.
Ramaswami U . Update on role of agalsidase alfa in management of Fabry disease. Drug Des Devel Ther 2011;5:155–173.
Mehta A, West ML, Pintos-Morell G, et al. Therapeutic goals in the treatment of Fabry disease. Genet Med 2010;12:713–720.
Lidove O, West ML, Pintos-Morell G, et al. Effects of enzyme replacement therapy in Fabry disease–a comprehensive review of the medical literature. Genet Med 2010;12:668–679.
Keating GM . Agalsidase alfa: a review of its use in the management of Fabry disease. BioDrugs 2012;26:335–354.
West ML, LeMoine KM, Bichet DG, et al. A randomized controlled trial of enzyme replacement therapy in Fabry nephropathy: the Canadian Fabry Disease Initiative at year two [Abstract SA-PO2780]. J Am Soc Neph 2009;20:747A.
Lee K, Jin X, Zhang K, et al. A biochemical and pharmacological comparison of enzyme replacement therapies for the glycolipid storage disorder Fabry disease. Glycobiology 2003;13:305–313.
Sakuraba H, Murata-Ohsawa M, Kawashima I, et al. Comparison of the effects of agalsidase alfa and agalsidase beta on cultured human Fabry fibroblasts and Fabry mice. J Hum Genet 2006;51:180–188.
Huby RD, Dearman RJ, Kimber I . Why are some proteins allergens? Toxicol Sci 2000;55:235–246.
Bekri S . Importance of glycosylation in enzyme replacement therapy. In: Mehta A, Beck M, Sunder-Plassmann G (eds). Fabry Disease: Perspectives from 5 Years of FOS. Oxford PharmaGenesis: Oxford, UK (2006).
European Medicines Agency. Assessment report on the shortage of Fabrazyme. Overview of shortage period: spontaneous reports from June 2009 through 15 September 2010 and registry data from June 2009 through 05 August 2010. EMEA/H/C/000370. http://www.ema.europa.eu/docs/en_GB/document_library/Other/2010/11/WC500099241.pdf Accessed December 2012.
European Medicines Agency. European Medicines Agency reviews treatment recommendations for Fabrazyme. http://www.ema.europa.eu/docs/en_GB/document_library/Press_release/2010/10/WC500098370.pdf Accessed December 2012.
Linthorst GE, Germain DP, Hollak CE, et al.; European Medicines Agency. Expert opinion on temporary treatment recommendations for Fabry disease during the shortage of enzyme replacement therapy (ERT). Mol Genet Metab 2011;102:99–102.
Tsuboi K, Yamamoto H . Clinical observation of patients with Fabry disease after switching from agalsidase beta (Fabrazyme) to agalsidase alfa (Replagal). Genet Med 2012;14:779–786.
Levey AS, Stevens LA, Schmid CH, et al.; CKD-EPI (Chronic Kidney Disease Epidemiology Collaboration). A new equation to estimate glomerular filtration rate. Ann Intern Med 2009;150:604–612.
Devereux RB, Alonso DR, Lutas EM, et al. Echocardiographic assessment of left ventricular hypertrophy: comparison to necropsy findings. Am J Cardiol 1986;57:450–458.
Lang RM, Bierig M, Devereux RB, et al.; Chamber Quantification Writing Group; American Society of Echocardiography’s Guidelines and Standards Committee; European Association of Echocardiography. Recommendations for chamber quantification: a report from the American Society of Echocardiography’s Guidelines and Standards Committee and the Chamber Quantification Writing Group, developed in conjunction with the European Association of Echocardiography, a branch of the European Society of Cardiology. J Am Soc Echocardiogr 2005;18:1440–1463.
Tan G, Jensen MP, Thornby JI, Shanti BF . Validation of the Brief Pain Inventory for chronic nonmalignant pain. J Pain 2004;5:133–137.
The EuroQoL Group. EuroQoL-a new facility for the measurement of health-related quality of life. Health Policy 1990;16:199–208.
Whybra C, Kampmann C, Krummenauer F, et al. The Mainz Severity Score Index: a new instrument for quantifying the Anderson-Fabry disease phenotype, and the response of patients to enzyme replacement therapy. Clin Genet 2004;65:299–307.
Linthorst GE, Burlina AP, Cecchi F, et al. Recommendations on reintroduction of agalsidase Beta for patients with Fabry disease in Europe, following a period of shortage. JIMD Rep 2013;8:51–56.
Smid BE, Rombach SM, Aerts JM, et al. Consequences of a global enzyme shortage of agalsidase beta in adult Dutch Fabry patients. Orphanet J Rare Dis 2011; 6:69.
Pisani A, Spinelli L, Visciano B, et al. Effects of switching from agalsidase Beta to agalsidase alfa in 10 patients with Anderson-Fabry disease. JIMD Rep 2013;9:41–48.
van Breemen MJ, Rombach SM, Dekker N, et al. Reduction of elevated plasma globotriaosylsphingosine in patients with classic Fabry disease following enzyme replacement therapy. Biochim Biophys Acta 2011;1812:70–76.
Maisel A . B-type natriuretic peptide levels: diagnostic and prognostic in congestive heart failure: what’s next? Circulation 2002;105:2328–2331.
Yasuda K, Kimura T, Sasaki K, et al. Plasma B-type natriuretic peptide level predicts kidney prognosis in patients with predialysis chronic kidney disease. Nephrol Dial Transplant 2012;27:3885–3891.
Linthorst GE, Hollak CE, Donker-Koopman WE, Strijland A, Aerts JM . Enzyme therapy for Fabry disease: neutralizing antibodies toward agalsidase alpha and beta. Kidney Int 2004;66:1589–1595.
Tanaka A, Takeda T, Hoshina T, et al. Enzyme replacement therapy in a patient with Fabry disease and the development of IgE antibodies against agalsidase beta but not agalsidase alpha. J Inherit Metab Dis 2010;33(suppl 3):S245–252.
Acknowledgements
The authors acknowledge Professor Yoshikatsu Eto, Professor Toya Ohashi, and Dr Masahisa Kobayashi (The Jikei University School of Medicine) for performing gene mutation measurements, and Professor Fumio Endo and Dr Kimitoshi Nakamura (Kumamoto University Hospital) for completing measurements of enzyme activity. The authors thank all patients who participated in this study, and Steve Clissold and Kathy Croom of Content Ed Net for editorial assistance, which was funded by Shire. They also thank Genzyme Corporation, a Sanofi company, for measurement of antibody to agalsidase-β, Shire for measurement of Gb3 and lyso-Gb3, and Dainippon Sumitomo Pharma Co, Ltd for measurement of antibody to agalsidase-α. Shire funded medical writing assistance by Content Ed Net.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Figure S1
(DOC 120 kb)
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-No Derivative Works 3.0 License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/3.0/
About this article
Cite this article
Tsuboi, K., Yamamoto, H. Clinical course of patients with Fabry disease who were switched from agalsidase-β to agalsidase-α. Genet Med 16, 766–772 (2014). https://doi.org/10.1038/gim.2014.28
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gim.2014.28
Keywords
This article is cited by
-
A prospective 10‐year study of individualized, intensified enzyme replacement therapy in advanced Fabry disease
Journal of Inherited Metabolic Disease (2015)