Abstract
Ncb2, the β subunit of NC2 complex, a heterodimeric regulator of transcription was earlier shown to be involved in the activated transcription of CDR1 gene in azole resistant isolate (AR) of Candida albicans. This study examines its genome-wide role by profiling Ncb2 occupancy between genetically matched pair of azole sensitive (AS) and AR clinical isolates. A comparison of Ncb2 recruitment between the two isolates displayed that 29 genes had higher promoter occupancy of Ncb2 in the AR isolate. Additionally, a host of genes exhibited exclusive occupancy of Ncb2 at promoters of either AR or AS isolate. The analysis also divulged new actors of multi-drug resistance, whose transcription was activated owing to the differential occupancy of Ncb2. The conditional, sequence-specific positional escape of Ncb2 from the core promoter in AS isolate and its preferential recruitment to the core promoter of certain genes in AR isolates was most noteworthy means of transcription regulation. Together, we show that positional rearrangement of Ncb2 resulting in either activation or repression of gene expression in response to drug-induced stress, represents a novel regulatory mechanism that opens new opportunities for therapeutic intervention to prevent development of drug tolerance in C. albicans cells.
Similar content being viewed by others
Introduction
The commensal Candida albicans turns into a life threatening pathogen in immunocompromised individuals. For this lethality, C. albicans has an array of important traits, including yeast to hyphae transition, secretion of adhesion molecules and phenotypic switching1,2,3. Candidiasis caused by C. albicans is the fourth most common infections in hospitals which on several occasions are systemic and fatal4,5,6. Most commonly used antifungal drugs target unique components of fungal cells, which include either plasma membrane or cell wall (CW). For example, polyenes and azoles target ergosterol, a major unique component of fungal cell membrane while echinocandins target typical CW components. Fluconazole (FLC) is the most common azole which is widely used in clinics due to its low toxicity and easy oral administration7. However, the prolonged use of fungistatic FLC results in the development of tolerance towards the drug.
The Candida cells employ several strategies to acquire resistance not only to the drug in use but also display collateral resistance to other unrelated drugs. Among various mechanisms which contribute in acquiring multi-drug resistance (MDR), enhanced drug export represents an important strategy adopted by the pathogen8,9,10. Thus, most of the clinical MDR isolates of C. albicans are shown to over-express CDR1, CDR2 or MDR1 drug efflux pump protein-encoding genes. Cdr1 and Cdr2 belong to the ATP binding cassette (ABC) super-family of transporter proteins and use energy derived from ATP hydrolysis to expel drugs to the cell exterior. Mdr1 on the other hand is an H+/drug antiporter which belongs to the super-family of major facilitators (MFS)11,12,13. Notably, both ABC and MFS family of transporters contribute to MDR in Candida; the regulatory circuits which control their gene expression are different13,14,15.
CDR1 is a major drug efflux protein-encoding gene which is regulated at the transcriptional as well as at post transcriptional levels9,16,17,18,19,20,21,22. It has been shown that CDR1 promoter harbours various cis-regulatory elements (Sp1, AP-1, and Y-box) as well as specific cis-elements such as Basal Regulatory Element (BRE), Negative Regulatory Element (NRE) and Drug/Steroid Response Element (DRE and SRE) in the 5′ flanking region17,20,23,24. Trans-acting factors regulating CDR1 have also been identified. For example, Non dityrosine 80 (Ndt80), a homolog to a meiosis-specific transcription factor (TF) in Saccharomyces cerevisiae is an activator of CDR118. A regulator was identified to be the Transcriptional Activator of CDR genes (Tac1) binds to the Drug Response Element (DRE) in both the CDR1 and CDR2 promoters19. Interestingly, the zinc cluster family TF Fcr1 (Fluconazole Resistance 1) and the global repressor Tup1 (Thymidine Uptake 1) act as negative regulators of CDR1 expression25,26. A genome-wide profiling (ChIP-on-chip) has shown that another TF of the zinc cluster family, Uptake control 2 (Upc2) regulating transcription of the ERG genes, also targets CDR127.
In addition to the gene-specific transcription factors, there are a variety of accessory factors that act in global manner in regulating MDR genes. For example, chromatin-modifying enzymes and proteins positively or negatively affect the formation of an active transcription initiation complex28,29,30. Negative Cofactor 2 (NC2) is an example of one such global regulator of RNA polymerase II (Pol II) which executes both negative and positive roles in transcription31,32,33,34,35,36. NC2 is a heterodimer comprised of two subunits, NC2α and NC2β (Ncb2) which was originally recognized as a TATA-binding protein (TBP) activity in human nuclear extracts that repressed RNA Pol II transcription37,38. It was later identified in S. cerevisiae as an essential factor and the β and α-subunit-encoding genes are termed as NCB2 (YDR1) and BUR6, respectively38,39. NC2 together with Mot1 [(Modifier of transcription 1 (Mot1) is a conserved and essential Swi2/Snf2 ATPase)] and TBP forms evolutionary conserved complex at the promoter regions40. It occupies opposite binding surfaces to MOT1 on TBP and encircle DNA bound TBP molecule which recognizes the minor groove of the TATA-sequence40. Additionally, NC2 can localize TBP onto the promoters by using its histone fold domain and bind to the underside of the DNA by using C-terminal helix of NC2β which surrounds DNA and binds convex side of TBP40,41. Consequently, NC2 together with TBP can move laterally on DNA. It has been shown that NC2 induces dynamic conformational changes in the TBP-DNA interface that enables TBP to laterally move on the DNA substrate40,42.
In an earlier report, we have shown the importance of the β-subunit of NC2 complex in the basal and activated transcription of CDR1 in azole sensitive (AS-Gu4) and azole resistant (AR-Gu5) isolates of C. albicans and also provided evidence that this cofactor may also be involved in the regulation of select genes, particularly those targeted by Tac143. This study evaluates the genome-wide occupancy of Ncb2 between genetically matched, AS and AR isolates of C. albicans. The study shows that Ncb2 occupies the promoters of 25–30% of C. albicans genes of almost every metabolic class in AR and AS isolates, thereby reinforcing its status as a global regulator of gene expression. Notably, there are hosts of novel MDR genes, which are regulated by Ncb2 in response to drug stress, leading to transcriptional activation.
Results
Ncb2 emerged as a global regulator of gene expression in AS and AR isolates of C. albicans
Ncb2, a part of the heterodimeric NC2 complex that controls transcription of genes was earlier shown to be involved in the activated transcription of CDR1 gene in azole resistant (AR) clinical isolate of C. albicans43,44. To understand the global impact of Ncb2 in acquisition of MDR in C. albicans, we performed a genome-wide recruitment analysis of the transcriptional regulator by employing ChIP-on-chip assay and compared it between azole sensitive (AS) and AR isolates. Three biological replicates were processed for each of the isolate to identify DNA regions that were occupied by Ncb2. By using polyclonal anti-Ncb2 antibody, the cross-linked protein-DNA complexes were pulled down43. Signal peaks detected in all the three experiments were chosen. Analysis of the genome-wide data revealed no major difference in terms of percentage occupancy of Ncb2 and varied between 25–30% in AR and AS isolates (Fig. 1a). For further analysis, by employing three different arbitrary cutoffs, all the promoters were ranked according to the P-values and fold enrichments (in comparison with input DNA); I) genes with P-values ≤ 0.055 and fold enrichments ≥1.5 (AS-1873 genes, AR-1550 genes), II) genes with P-values ≤ 0.055 and fold enrichments ≥2.0 (AS-1765 genes, AR-1546), III) genes with P-values ≤ 0.01 and fold enrichments ≥2.0 (AS-1119, AR-960). Furthermore, to compare the genomic binding of Ncb2 between the isolates, the average binding profile for the promoter (fragments: −1/−49, −50/−300, −301/−550, −551/−800, −801/−1250 and ≥1251, relative to the start codon) was computed which showed that Ncb2 mainly recruited at the core promoter regions43,45,46 (Fig. 1b). Genome-wide occupancy data analysis also revealed that Ncb2 binds both inside and downstream of the genes (Supplementary Figs S1 and S2). The most common model for transcriptional activation is that the binding, just upstream of the transcription start site (TSS) helps in placing polymerase II at the beginning of the gene. Presence of TF binding sites inside or downstream of gene may act as regulatory sequence for the same gene or act as enhancer for the control of same gene or downstream gene (acting as a distal enhancer) in orientation and position independent manner47,48,49. Notably, all binding sites may not be functional or represents a non productive binding site or could just be alternative TSS50,51,52.
(a) Charts showing the Ncb2 occupying and non-occupying gene promoters of C. albicans in an Ncb2 ChIP performed in Gu4 (AS) and Gu5 (AR) clinical isolates. (b) Ncb2 binding peaks localizes at the −50/−300 region encompassing the core promoter. Average binding profiles of Ncb2 for the promoter regions were determined relative to ATG start codon for both the isolates. Green line represents localization profile of Ncb2 in the AS isolate; whereas blue line represents localization profile of Ncb2 in AR isolate. Binding distance was calculated for the probe that has maximum normalized log ratio.
Ncb2 occupies genes with diverse metabolic functions
To get an insight into the functional features of the biological processes that are modulated by Ncb2, we have chosen most stringent arbitrary condition for both the isolates (genes with P-values ≤ 0.01 and fold enrichments ≥2.0), and carried out a search at Candida Genome Database (CGD). A closer look at Ncb2 occupancy revealed its binding to the diverse functional classes of genes involved in metabolism, transport, cell wall, filamentous growth, virulence, mitochondrial function, signal transduction, biofilm formation and oxidative stress (Supplementary Tables S1 and S2). Notably, Ncb2 occupancy at the promoter regions of the genes encoding membrane transporter was very conspicuous (Supplementary Tables S1 and S2). For example, Ncb2 not only showed binding at the promoter region of CDR1 but also at the promoters of ABC transporter genes CDR2, CDR4 and MDL2 and MFS transporters including MDR1, FLU1, NGT1, HGT1, HGT2, HGT7, HGT19, HGT20, ITR1, TPO3 and RTA2 (Supplementary Tables S1 and S2). Moreover, promoters of well-known MDR determinants like PDR16, ERG11, NCP1, ERG251 and ERG9 were also found to be occupied by Ncb2 (Supplementary Tables S1 and S2).
The higher occupancy of Ncb2 in AR isolate has dual effect on transcription
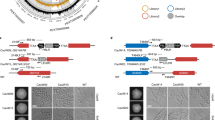
We compared the genome-wide recruitment data of Ncb2 between AS and AR isolates and thereby, identified genes that showed distinct occupancy of Ncb2 at the promoter regions. The analysis revealed that a number of genes have higher enrichment of Ncb2 occupancy in AR isolate (Supplementary Table S3). This included genes implicated in drug response, filamentous growth, various stresses, pathogenesis, transport, cell wall organization, signal transduction, and biofilm formation (Supplementary Table S4). To establish a relation between the higher occupancy of Ncb2 in AR isolate and gene expression, we randomly selected 6 genes (PMT1, orf19.4601, PGA17, RTA4, ERG11 and GSC1) of interest with different normalized log ratios (enrichments) and P-values. Their expression was checked by semi-quantitative end-point RT (reverse transcription) PCR. CDR1 and CDR2 were used as positive controls. Inspite of the higher Ncb2 occupancy, the expression data showed a mixed response of gene expression. For instance, three genes [orf19.4601 (Putative RNA polymerase III transcription initiation factor complex (TFIIIC) subunit), PGA17 (Putative GPI-anchored protein) and GSC1)] were up-regulated in AR isolate whereas PMT1 (Protein mannosyltransferase), RTA4 (Protein similar to S. cerevisiae Rsb1p, involved in fatty acid transport) and ERG11 were down regulated in AR cells (Fig. 2a,b; Supplementary Fig. S3). The validation though limited, points that the higher occupancy of Ncb2 in AR isolate does not always result in transcriptional activation. Nevertheless, Ncb2 probably manifests dual effect on transcriptional control mechanisms. Since genes that showing higher enrichment in ChIP-on-chip data displayed differential gene expression pattern in AS and AR isolates hence we analyzed the relative recruitment dynamics of Ncb2 in both the isolates. For this, we used ChIPed DNA from AS and AR isolates and subjected to ChIP-PCR analysis which revealed that Ncb2 binding correlates well with the ChIP-on-chip data (Fig. 2c,d; Supplementary Fig. S4).
(a) End point semi-quantitative RT-PCR was used to validate some of the genes found to be highly enriched by Ncb2 in the AR isolate and their relation to gene expression. RT-PCR was performed in triplicate and a representative figure (cropped gel pictures) is shown. For full gel see Supplementary Fig. S3. CDR1 and CDR2 genes were used as positive controls for the gene over-expressed in the AR isolate. ACT1 expression level was used as a control for isoexpression between AR and AS isolates. (b) Bar diagram showing the expression profile of genes highly enriched in AR isolate as compared to AS isolate by Ncb2. Bars represent the standard deviations observed for the replicate experiments. (c) Recruitment profiles of Ncb2 in AR isolate as compared to AS isolate determined by ChIP-PCR. For presentation cropped gels are shown. For complete gel see Supplementary Fig. S4. IN represents amplification observed in input sample, control (C) and test (T) are immunoprecipitations carried out on cross-linked chromatin using pre-immune serum and anti-Ncb2 antibody, respectively. Amplification of CDR2 promoter region was used as a positive control. ADH1 and ACT1 promoter regions amplification were used as positive and negative control for Ncb2 binding. (d) Bar diagram representing the Ncb2 enrichments of AR over AS isolate. Bars represent standard deviations for the replicate experiments.
Genes exclusively enriched by Ncb2 in the AR isolate mostly exhibit activated transcription
The analysis of the genome-wide Ncb2 recruitment dynamics revealed 25 gene promoters which displayed an exclusive occupancy of Ncb2 in the AR isolate whereas its binding was absent in AS isolate (Supplementary Table S5). In this category, for validation, we carried out expression analysis for the genes which included orf19.1698, CHK1, orf19.1434, orf19.7063, PDC12, ARO80 and UTP22 with different enrichment ratios and P-values and performed end point semi-quantitative RT-PCR analysis (Fig. 3a,b; Supplementary Fig. S5). Remarkably, with the exception of UTP22, most of the genes showing selective enrichment in the AR isolate were accompanied by an increased expression. This implies that the exclusive enrichment of Ncb2 in AR isolate mainly result in transcriptional activation (Fig. 3a,b; Supplementary Fig. S5). Subsequent ChIP-coupled PCR on ChIPed DNA obtained from AS and AR isolates validated the exclusive recruitment dynamics of Ncb2 (Fig. 3c,d; Supplementary Fig. S6). These results revealed that Ncb2 binding was exclusively present only in the ChIPed DNA isolated from the AR isolate (Fig. 3c,d; Supplementary Fig. S6). Therefore, it can be concluded reasonably that Ncb2 exclusively occupies a subset of genes in the AR isolate and is primarily involved in the positive regulation of gene expression (Fig. 3a,b,c,d; Supplementary Figs S5 and S6).
(a) Semi-quantitative RT-PCR was used to study the effect of exclusive enrichment of Ncb2 and gene expression of selected genes in the AR isolate. Most of the exclusive target genes of Ncb2 in the AR isolate were found activated except for UTP22 which was repressed. CDR1 and CDR2 genes served as controls that over-expressed in the AR isolate. ACT1 expression level was used as control that equally expresses in both the isolates. RT-PCR was performed from c-DNA prepared from three different RNA preparations. For presentation cropped gels are shown. For complete gel see Supplementary Fig. S5. (b) Bar diagram demonstrating expression profile of AR exclusive enriched genes. Bars represent standard deviations observed for the replicate experiments. (c) ChIP-PCR analysis of Ncb2 exclusive occupancy in AR isolate. Chromatin immunoprecipitation was performed on cross linked chromatin isolated from AS and AR isolates, respectively. Control and test designate immunoprecipitations performed on cross-linked chromatin with pre-immune serum and anti-Ncb2 antibody, respectively. Amplification of ADH1 and ACT1 promoter regions were used as a positive and negative control for Ncb2 binding in both the isolates. Cropped gels are used for presentation, for complete gel see Supplementary Fig. S6. (d) Bar diagram showing the Ncb2 exclusive occupancy in AR isolate as compared to AS isolate. Bars represent the standard deviations observed for the replicate experiments.
Ncb2 acts both as a positive and negative regulator of gene expression in the AS isolate
The comparative analysis of the genome-wide binding profiles of Ncb2 in the AR and AS isolates also identified genes whose promoters were exclusively enriched in the AS isolate (Supplementary Table S6). GO slim analysis of the genes exclusively enriched in AS isolate showed that these are involved in transport, filamentous growth, response to drug, pathogenesis, and response to stress etc. (Supplementary Table S7). Four genes were chosen with different binding strength of Ncb2 to establish a relation between exclusive occupancy in AS isolate and gene transcription. The validation of the relation between Ncb2 exclusive occupancy in AS isolate and expression of this group of genes confirmed that the Ncb2 has a dual role both as a positive and negative regulator of gene expression (Fig. 4a,b; Supplementary Fig. S7). Interestingly, ChIP-coupled PCR from the ChIPed DNA obtained from AR and AS isolate of these genes confirmed that Ncb2 only occupies the genes of AS isolate (Fig. 4c,d; Supplementary Fig. S8).
(a) To establish a relation between Ncb2 AS-specific enrichment and gene transcription, we employed semi-quantitative RT-PCR analysis for selected genes. Expression data shows that it acts to control gene expression positively and negatively in the AS isolate. CDR1 expression level was used as a control for genes induced in AR strain. ACT1 expression level was used for the normalization of the data. RT-PCR was performed from RNA isolated from three different preparations. Cropped gels were used for presentation. Complete gels are shown in Supplementary Fig. S7. (b) Bar diagram was used to find out the differences in the gene expression of genes exclusively occupied by Ncb2 in the AS isolate. Bars represent the standard deviation observed for the replicate experiments. (c) ChIP analysis of the promoter regions of selected genes of Ncb2 exclusive occupancy in AS isolate. Control and test represents experiment performed on cross-linked chromatin with pre-immune and anti-Ncb2 antibody. Precipitation of ADH1 and ACT1 promoter regions were used as positive and negative control for Ncb2 binding. For presentation purpose cropped gels are shown. For full gel see Supplementary Fig. S8. (d) Bar diagram showing the exclusive occupancy of Ncb2 in AS isolate as compared to AR isolate. Bars represent the standard deviation observed for the replicate experiments.
Ncb2 impacts the transcription of MDR genes
In addition to the global regulatory role of Ncb2, comprehensive analysis of the genome-wide data indicates that it may also have gene-specific functions. For instance, it was observed that Ncb2 differentially occupies promoter regions of the genes that are specifically known to be over-expressed in AR isolates. Among these are the well-known MDR genes CDR1, CHK1, IFU5, RTA3, CDR2 and LCB4, whose over-expression is characteristic of several drug-resistant clinical isolates of C. albicans53. Several other genes that showed Ncb2 differential enrichment in AS and AR isolates which included orf19.2204, orf19.1698, orf19.5614, PDC12, orf19.1676, ADAEC, and ECE1 was also noteworthy. Activated transcription of these genes is reported in one or two azole-resistant clinical isolates, therefore implying their putative roles in MDR53,54.
Previous ChIP-on-chip experiments in C. albicans identified 37 gene promoters that were occupied by Tac1, a Zn(II)2Cys6 family transcription factor involved in the transcriptional activation of CDR1 and CDR29,19,53. The Tac1 regulon includes genes involved in the lipid metabolic process, oxidative stress response regulators and genes of unknown functions53. Notably, earlier we observed that apart from CDR1 and CDR2, Ncb2 could regulate other Tac1 target genes i.e. RTA3, IFU5, LCB4, GPX1 and orf19.188743. Our present analysis extends this observation to show that Ncb2 is involved in the coordinated transcriptional regulation of most of the Tac1 regulon genes (Supplementary Table S8).
Ncb2 escapes upstream of the core promoters/TATA-box regions in AS isolate
Earlier, we had observed that the recruitment of Ncb2 at the core promoter or TATA-box region led to an activated transcription of CDR1 in the AR isolate whereas its shift in binding to upstream of the TATA-box region in AS isolate resulted in its repression43. It is proposed that NC2 binds and stabilizes TBP to retain the initiation factors at the promoter regions wherein it also exerts dynamic changes in the conformation of TBP-DNA complex and mobilizes TBP on the DNA42. Since it is shown that in case of CDR1-activated transcription, Ncb2 together with TBP moves on the DNA, we explored if positional shift of Ncb2 is also associated with the regulation of other genes42,43. We identified 35 genes that display Ncb2 occupancy at the core promoters/TATA-box regions in AR isolate as compared to the AS isolate where it escapes and binds upstream of the core promoters/TATA-box regions (Supplementary Table S9). These genes belong to the biofilm formation (ZCF8, NDH51, orf19.7459, SUN41 PDX1, CRZ2, ECE1), filamentous growth (ZCF8, NDH51, orf19.7459, SUN41, PDX1, SSK1), response to stress (NDH51, CTA8, SSK1, CRZ2), response to drug (CDR1, EFT2), translation (FUN12, EFT2), transport (GIT3, CDR1), cell wall organization (SUN41, SSK1), and pathogenesis (SUN41, SSK1), as identified by GO slim analysis (Supplementary Table S10). We hypothesized that selective occupancy of Ncb2 at the core promoter regions in the AR isolate may be a transcriptional activation mechanism, like we observed for CDR1. However, here the expression data revealed that the positional shift involves both transcriptional activation and repression. Nonetheless, it appears that the positional shift in Ncb2 occupancy for a subset of genes in the presence of drug stress is a novel regulatory mechanism that may either involve positive or negative regulation of gene expression (Fig. 5a; Supplementary Figs S9 and S10). For example, out of 9 genes selected for validation, 4 genes showed over-expression (orf19.4715, SSK1, ADAEC and ECE1) while 5 genes were repressed (FUN12, DFI1, YNK1, orf19.805 and PDX1) in the AR isolate (Fig. 5a; Supplementary Figs S9 and S10).
(a) The expression analysis of the selected genes by end point semi-quantitative RT-PCR revealed that Ncb2 acts as activator and repressor of gene transcription in AR isolate. CDR1 expression level was used as positive control for genes over-expressed in AR isolate. ACT1 expression level was used as endogenous control for both the isolates. Cropped gel bands are shown, for complete gel see Supplementary Fig. S10. (b) Schematic representation of the positions of primer sets used in chromatin restriction digestion couple immunoprecipiation (CRIP) experiments. (c) CRIP assay depicting the recruitment of Ncb2 at the core promoter upstream region of PDX1 gene in AS isolate whereas recruitment was found at core promoter region near ATG codon in AR isolate. IN, C, and T denote input DNA and immunoprecipitation found with control pre-immune serum and anti-Ncb2 antibody, respectively. A bar diagram displaying the difference in occupancy of Ncb2 at core promoter upstream and core promoter near ATG codon containing fragments of the PDX1 promoter between AR and AS isolates is shown on the right. (d) CRIP assay showing the Ncb2 differential position occupancy at DFI1 promoter upstream region and at core promoter region in AS and AR isolates. Cropped gels are shown, see Supplementary Fig. S11 for full gel. On the right side of the figure a bar diagram representing the difference in occupancy of Ncb2 in AR and AS isolates is shown.
Further, shift in the positional occupancy of Ncb2 was confirmed by using chromatin restriction digestion coupled immunoprecipitation (CRIP) assay. For this, genes that displayed shift in occupancy of Ncb2 and showed regulated gene expression in AS and AR isolates were chosen. CRIP assay was performed with genes such as PDX1, DFI1, ADAEC and ECE1 in AS and AR isolates. Their promoter DNA was separated into ATG codon containing core promoter and upstream of the core promoter fragments by using HinFI, EcoRV, XhoI and DpnI site as depicted in Fig. 5b. The fragmented chromatin was precipitated by using anti-Ncb2 antibody followed by PCR using two primer pairs for each gene (Fig. 5b). Interestingly, it was observed that in the AS isolate Ncb2 occupancy was detected at upstream of the core promoter regions while there was no occupancy at the core promoter near ATG codon (Figs 5c,d and 6a,b; Supplementary Figs S11 and S12). Of note, a totally reverse scenario was observed in the AR isolate where Ncb2 was exclusively recruited at the core promoter regions (Figs 5c,d and 6a,b; Supplementary Figs S11 and S12).
(a) CRIP assay showing the differential recruitment of Ncb2 at ADAEC promoter region in AS and AR isolates. The result demonstrate that in AS isolate Ncb2 recruited at the upstream of core promoter region whereas its recruitment was found at the core promoter near ATG codon in AR isolate. On the right side of the figure a bar diagram depicting the difference in enrichment profiles is shown. (b) Ncb2 occupy different positions at promoter regions of ECE1 in AS and AR isolates. CRIP assay showing the differential recruitment profiles of Ncb2 at gene promoter of ECE1. Its occupancy was found at upstream region of core promoter in AS isolates whereas it occupies at the core promoter region in AR isolates. On the right side of the figure a bar diagram representing the observed differences of Ncb2 occupancy in AS and AR isolates is shown. For demonstration cropped gel bands are shown. Complete gels are shown in Supplementary Fig. S12. (c) Ncb2 preferentially recruited at A-rich sequence motif of DNA. Logo of the Ncb2 DNA binding motif discovered by Rsat using 500 promoter regions that have highest enrichment profiles in ChIP-on-chip data.
A hypothetical model depicting differential positional occupancy at the promoter regions is shown in Fig. 7a. Analysis of the promoter regions of upstream positional shift in AS isolate revealed that Ncb2 prefers different positions at the promoter regions of the genes. For instance, the distance of Ncb2 occupancy at A-rich Ncb2 binding motif of DNA varied between −135 bp and −3659 bp (Fig. 7b; Supplementary Table S9). In contrast, in AR isolate Ncb2 occupies the core promoter region at a slightly dissimilar sequence in comparison to AS isolate (Fig. 7c).
(a) Hypothetical depiction of the preferred positional occupancy of Ncb2 at core promoter or TATA-box regions in a number of genes in AR isolate. TBP and TSS stand for TATA-binding protein and transcription start site, respectively. Bur6 and Ncb2 denote α and β-subunits of NC2 complex. (b) Logo demonstrating A-rich sequence motif of Ncb2 binding in the DNA sequence of TATA-upstream regions/upstream of core promoter regions in the AS isolate. (c) Illustration of sequence logo of Ncb2 binding motif in the DNA sequences of core promoter/TATA-box regions in the AR isolate.
Tac1 plays a role in regulation of AR specific target genes of Ncb2
Earlier, we had evaluated the possible role of Ncb2 in regulating MDR genes, specifically those co-regulated with CDR1 by the transcription regulator, Tac1. We observed that Ncb2 occupied the promoter regions of a few of the Tac1-regulon genes43. In this study, we evaluated whether Ncb2 recruitment is Tac1 dependent. For this, its recruitment dynamics at promoter regions of MDR-genes in the Δtac1 strain was determined. We observed that Ncb2 was associated at the promoter regions of selected MDR genes from all categories (AR highly enriched genes, AR exclusive genes, AS exclusive genes, positional shift category) which included RTA4, ERG11, CDR2, HEM15, orf19.6898.1, NOP14, CDR1, DFI1, PDX1, RTA3 and MDR1, however, orf19.1698, CHK1 and orf19.7063 showed no occupancy of Ncb2. Notably, all the three genes which appeared to be Tac1 dependent belongs to the AR exclusive Ncb2 enriched category. Among control genes included in the analysis, Ncb2 was seen to be bound to the ADH1 promoter in Δtac1 strain, while it was not seen on the ACT1 promoter (Fig. 8a,b; Supplementary Fig. S13). Whether Ncb2 occupancy also impacts performance of other major TFs such as Ndt80, Mrr1, Upc2 and Ada2 contributing to MDR in C. albicans, remains to be explored27,30,55,56. In addition, we also analyzed the promoter regions (250 bp around the detected peaks for the genes that exhibited differential positional occupancy in AS and AR isolates) for the presence of other TFs motif, comprising transcriptional regulators of MDR such as Tac1, Ndt80, Mrr1, Ada2 and Upc2. In this context our preliminary analysis revealed no cis-elements in the promoter regions analyzed.
(a) ChIP assay of Ncb2 recruitment with the promoters of a selected set of MDR genes of C. albicans in Δtac1 strain. Cropped gel pictures are shown for presentation. For complete gel pictures see Supplementary Fig. S13. (b) The enrichment of Ncb2 recruitment at the promoters of selected MDR genes in Δtac1 strain is represented as a bar diagram. Error bars denote standard deviations for the replicate experiments. IN, C, and T are the same as those described for earlier figures.
Discussion
Continuous exposure to the antifungal drugs are recognized as environmental cues that triggers different mechanisms to develop drug tolerance and in the selection of the drug-resistant strains with the capacity to permanently withstand high thresholds of drug concentrations57. Therefore, understanding the molecular basis of the mechanisms that control the development of drug tolerance is of utmost importance55. In this context, we have earlier identified a novel mechanism of transcriptional regulation by a heterodimeric regulator Ncb2 which activate the transcription of CDR1 in AR clinical isolate of C. albicans43. Ncb2 is a part of heterodimeric NC2 complex which also comprises NC2α. NC2 directly interacts with TBP and prevent its association to TFIIB and TFIIA to inhibit transcription37,58. Genome-wide expression analysis has revealed that NC2 is involved in transcriptional repression and to a lesser extent in transcriptional activation36,59. Mechanistic details how NC2 affects positive regulation of gene expression is largely unknown. In our previous report it was observed that Ncb2 occupied core promoter upstream region in AS isolate whereas it positions at the core promoter region in AR isolate of CDR1 thereby leading to the formation of alternative pre-initiation complex assembly for transcription initiation. This study has further examined the genome-wide role of Ncb2 in C. albicans, particularly in the context of MDR. Our genome-wide ChIP-on-chip occupancy of Ncb2 between genetically matched pair of AS and AR isolates provides interesting insights into the novel regulation of transcription in response to drug stress in C. albicans.
Extensive analysis revealed that the transcription regulation by Ncb2 is not only restricted to CDR1 but also several genes across the entire genome show moderate to high occupancy of the regulator. It is also apparent that most of the MDR genes, which are co-regulated along with CDR1 by Tac1, are also enriched and regulated by Ncb2. The select validation of the enrichment of Ncb2 data points that the higher occupancy of the regulator does not necessarily result in the transcriptional activation as the expression of several genes was also repressed. Notwithstanding, the dual role of Ncb2, the genome-wide occupancy profiling reaffirmed its role as a global regulator of gene expression46,60.
A comparative analysis of the genome-wide binding profile of Ncb2 between AS and AR isolates disclosed that in response to drug stress, Ncb2 selectively escapes or binds to the core promoters/TATA-box regions. There were host of genes whose promoters showed positional shift of bound Ncb2 at different positions between AS and AR isolates. For example, while Ncb2 recruited at the core promoters/TATA-box regions in AR isolate, it exclusively positioned upstream of the core promoters/TATA-box regions in a set of genes of AS isolate. Interestingly, it has been observed earlier that the binding of NC2 (NC2α + Ncb2) complex to the DNA bound TBP results in the mobilization of TBP away from the TATA box or allows it to escape and return to the TATA binding mode probably by inducing a conformational change in the TBP-DNA complex42. Our earlier data where a close interaction between Ncb2, NC2α and TBP has been demonstrated supports such a mechanism43. It appears that Ncb2 not only binds and stabilizes TBP to maintain the initiation factor at the promoter regions, but it also mobilizes the TBP on the DNA, possibly together with NC2α. This mode of action of Ncb2 may explain its dual functionality42.
There were also a set of genes, which showed exclusive occupancy of Ncb2 on their promoters in AR isolate. Interestingly, most of such genes showed positive regulation of transcription. These results imply that an exclusive occupancy of Ncb2 in the AR isolate may exert a positive impact on gene expression by recruiting active pre-initiation complex at the promoters. In comparison, the genes whose promoter regions showed comparatively higher enrichment of Ncb2 occupancy in the AR isolate resulting in an activation or repression, points that the higher occupancy may not always lead to positive regulation of the transcription since it also depends on the promoter occupancies of other transcriptional activators46,60. The exclusive positional shift of Ncb2 at the core promoters/TATA-box regions in a set of genes in the AR isolate represent fine tuning of the regulation of gene expression to adapt the pathogen to drug stress. The members of such group comprised not only well-known MDR genes but also include several uncharacterized genes. The higher expression observed following validation of select genes suggest that these could represent novel genes who have important role to play in MDR of C. albicans.
The analysis of Ncb2 bound DNA regions showed that the frequency of Ncb2 motif is significantly higher as compared to the random sequences. Importantly, Ncb2 motif was found to be distributed around the detected peaks and constitute A-rich sequence (Fig. 6c). This is similar to the sequences that are found for the promoter regions of upstream positional shift in AS isolate (Fig. 7b). Interestingly, the presence of approximately repeated patterns or motifs signifies that these are regulated by the same gene regulatory mechanisms. Additionally, conservation of Ncb2 binding sequences in AS isolate for the genes that showed the core promoter upstream binding indicated that certain changes occurred in the genome of AR isolate that ultimately lead to the re-positioning of Ncb2 at the core promoters/TATA box regions. This possibly results in the regulated gene expression to adapt the pathogen to combat the effect of the drug.
In conclusion, we demonstrate a new mode of action of Ncb2 in C. albicans in response to drug stress by alternating its binding to the core promoters/TATA regions to either activate or repress transcription. Since, negative or positive regulation by Ncb2 does not correspond to the strength of the promoter occupancy; it may probably depend on the downstream promoter occupancies of other co-activators. Considering that MDR is a multifactorial manifestation of many regulatory mechanisms, this study provides an example that a global regulator may actively be involved in MDR acquisition in C. albicans.
Materials and Methods
Strains and growth
C. albicans azole resistant (AR) and azole sensitive (AS) isolates were grown in YEPD medium (1% yeast extract, 2% peptone, and 2% dextrose) in 30 °C incubator at 200 rpm for routine experimental purposes. Strains details are given in Supplementary Table S11.
Primers
All the primers used in this study are listed in Supplementary Table S12.
ChIP-chip amplification, labeling, hybridization and data analysis
ChIP experiments were carried out as described earlier43. In brief, 15 ml of cultures in YEPD medium (AS and AR cells) were cross-linked with 1% formaldehyde for 20 min at RT. The cross-linking reaction was quenched with 125 mM glycine. Cells were washed twice with ice cold TBS (Tris Buffered Saline: 20 mM Tris-Cl, pH 7.5, containing 150 mM NaCl) and resuspended in Zymolyase buffer (50 mM Tris, pH 7.4, 10 mM MgCl2, 1 M sorbitol, and 30 mM DTT) for the formation of spheroplast. Spheroplasts were washed twice with cold TBS containing 1 M sorbitol, resuspended in ChIP lysis buffer (50 mM HEPES-KOH, pH 7.5, 140 mM NaCl, 1 mM EDTA pH 8.0, 1% Triton X-100, 0.1% sodium deoxycholate, and 1 mM PMSF). SDS was added at a final concentration of 0.5%. Cells were sonicated for 25 sec at amplitude of 6 for seven cycles. After sonication cell suspensions were centrifuged and termed soluble total chromatin (STC). Fifteen μl samples were run on 1% agarose gel to check the size of the fragmented chromatin (approximate size ~250 bp to ~500 bp). STC was diluted 5 times with ChIP lysis buffer and pre-cleared. After pre-clearing the samples pre-immune serum (negative control) and anti-Ncb2 antibody was added at a final concentration of 2–5 μg and incubated O/N43. Next morning 20 μl packed volume of protein-A agarose beads were added, bound immune complexes were precipitated and washed twice with ChIP lysis buffer, TSE 150 (50 mM Tris-Cl, pH 8.0, 150 mM NaCl, 1% Triton X-100, 0.1% SDS, 1 mM EDTA, pH 8.0, and 1 mM PMSF), TSE 500 (50 mM Tris-Cl, pH 8.0, 500 mM NaCl, 1% Triton X-100, 0.1% SDS, 1 mM EDTA, pH 8.0, and 1 mM PMSF), buffer III (10 mM Tris-Cl, pH 8.0, 1 mM EDTA, pH 8.0, 250 mM LiCl, 1% NP-40, and 1% sodium deoxycholate), and finally with TE (10 mM Tris-Cl, pH 8, and 1 mM EDTA, pH 8.0). After washing, immunoprecipitated complexes were eluted in buffer containing 50 mM Tris-Cl, pH 7.5, 10 mM EDTA, pH 8.0, and 1% SDS at 65 °C O/N. The immunoprecipitated complexes and input DNA were treated with proteinase-K. The precipitated DNA were either extracted by PCI (phenol:chloform:isoamylalcohol) followed by ethanol precipitation using glycogen (30 μg) as a DNA carrier or purified using Qiagen PCR purification kit. The input and immunoprecipitated DNA were dissolved in 100 μl and 20 μl TE for the validation of ChIP-on-chip data or eluted in 70 μl of TE buffer for samples that were used for ChIP-on-chip experiments. In PCR analysis, amplifications were carried out for 25 to 28 cycles.
A high-density oligonucleotide microarray with 5000 bp upstream (promoters) and 500 bp downstream regions (genes) of C. albicans (SC5314) was used. It covered 6204 orfs, representing 99.7% of the total genome comprising 180000 unique 60 mer probes in situ synthesized using “SurePrint” technology (Agilent) that was optimized for DNA-protein binding studies. To study the Ncb2 genome-wide association, ChIPed DNA purified by Qiagen PCR purification kit send to Genotypic technology Bengaluru for linear amplification by LMPCR (ligation mediated polymerase chain reaction) and size distribution of amplified fragment (~250–500 bp) as well as yields were determined. LMPCR was performed by using 200 ng grams of input and immunoprecipitated DNA was blunted at 12 °C for 20 min using T4 DNA polymerase as per the Mammalian ChIP-on-chip Protocol Version 10.1 of Agilent Technologies. The blunted DNA was cleaned by phenol:chloroform extraction and the precipitated DNA were re-suspended in 25 μl of water and the entire DNA was ligated to linkers overnight. The linker-ligated DNA was subjected to two rounds of LMPCR as per protocol and the product was precipitated and re-suspended in 25 μl of water.
Two μg of input, ChIPed or immunoprecipitated DNA were labeled using Agilent Genomic DNA labeling kit PLUS and Cy3 (for ChIPed or immunoprecipitated DNA) or Cy5 (for input DNA) dUTP from Agilent. DNA yield and incorporation of label (specific activity) was measured using Nano Drop spectrophotometer. Labeled DNA were hybridized at 65 °C for 24 hr to Agilent 60 mer oligonucleotides. Hybridized arrays were scanned using high throughput Agilent scanner with “SureScan” technology, and process through automated feature extraction using Agilent feature extraction software. After quantification and normalization of the signals (ChIPed-DNA or immunoprecipitated DNA/input DNA), the binding profiles were represented as normalized log ratios. Normalization of data and statistical analysis were performed using Agilent’s DNA analytics software (Agilent genomic workbench lite edition 6.5; ChIP-on-chip application, and Microsoft excel). Normalization methods used were median blanks subtraction and intra-array (intensity-dependent) LOWESS normalization. In blanks subtraction normalization for each channel (IP or input) of each replicate of each array, the median of the intensities of the blank spots is subtracted from each intensity on that replicate. As in all normalization steps, any negative signal intensities that result will cause the probe to be flagged as ‘excluded.’ Intra-array normalization attempts to correct for artifacts caused by nonlinear rates of dye incorporation, as well as inconsistencies in the relative fluorescence intensity between some red and green dyes. The LOWESS (locally weighted scatter plot smoothing) algorithm normalizes the channels within each array using a nonlinear polynomial fit to the data, and effectively normalizes by probes and by arrays. Whitehead per-array neighborhood model was used to detect the protein-DNA binding event in DNA Analytics software (DNA analytics detects robust peaks of probe signal corresponding to binding events). This method uses the distribution of all probes on each array to compute robust regions of increased probe signal (termed ‘peaks’). The algorithm does this by examining groups of probe triplets which are significantly enriched, yielding robust binding event detection. The Whitehead model samples every probe and its immediate upstream or downstream neighboring probe to identify a robust estimate of the location of bound protein. Three ChIPed DNA samples were processed for each strain. The data sets that support the results of this article are available in the GEO repository [ http://www.ncbi.nlm.nih.gov/geo/] with accession number GSE768558 for the tiling array data.
Isolation of RNA and RT PCR
Total RNA from AS and AR isolates were extracted by using Trizol reagent (Sigma). Briefly, cells from 15 ml of log phase cultures were centrifuged and washed twice with DEPC treated water. Three hundred mg of acid washed sterilized beads were added with 1 ml of Trizol reagent. Bead beating was done for 1 min, 3 times at 1 min interval on ice. After bead beating, 200 μl of chloroform was added, cell suspensions were mixed and centrifuged at 13 K for 10 min. Clear supernatants were taken out and 600 μl of isopropanol was added in each sample and stored at −20 °C O/N. Next day, samples were centrifuged at 13 K for 10 min at 4 °C and RNA pellet was washed with 75% ice cold ethanol. Samples were air dried and dissolved in 30 μl of DEPC treated water. Reverse transcription-PCR (RT-PCR) was performed as mentioned in the RevertAidTMH Minus kit (MBI, Fermentas). Briefly, a total of 10 μg RNA was treated with DNase for 30 min at 37 °C. Reactions were terminated by adding 1 μl of 25 mM EDTA, pH 8.0, and incubated further at 65 °C for 10 min. One μl of DNA free total RNA (1 μg) was primed with oligo(dT)18 and random hexamers for first strand c-DNA synthesis at 42 °C for 60 min. Reverse transcription reaction was terminated by heating at 70 °C for 5 min. Half a microlitre of c-DNA was used in PCR (25 μl reaction volume) primed with specific forward and reverse primers. Twenty-five to thirty cycles of amplifications were carried out to synthesize second strand unless otherwise stated. ACT1 was used as an endogenous control. For every RT-PCR analysis, c-DNA was synthesized from RNA isolated from three different preparations. The PCR-amplified products were analyzed by agarose gel electrophoresis and quantified by densitometry.
Ncb2 consensus motif discovery
To find out the consensus binding site of Ncb2, the promoter sequences were analyzed with Regulatory sequence analysis tool (RSAT, http://rsat.sb-roscoff.fr) and Multiple EM for Motif Elucidation (MEME) program (http://meme-suite.org/tools/meme). For the analysis of Ncb2 consensus motif, 500 promoter sequences of the genes that showed highest Ncb2 binding normalized log ratio were selected. Three hundred bp sequence of the promoter regions were chosen around the detected peaks. The program was allowed to discover motif with oligo and position analysis with oligomer length above 6–7 bp and five motifs per algorithm searched on both strands. To find out the Ncb2 consensus sequence for the genes that showed core promoters/TATA-box upstream binding in AS isolate, 250 bp sequences were used around the detected binding peak. Furthermore, for the analysis of conserved sequences in the AR isolate in which Ncb2 binds at core promoters/TATA-box regions, 125 bp upstream and downstream sequences were taken from the ATG codon.
CRIP assay
The chromatin restriction digestion-coupled immunoprecipitation (CRIP) assay was performed as described earlier for the ChIP assay, with minor changes as described43. STC was fragmented by sonication to have a size of ~250 to ~500 bp. The Fragmented STC was subjected to restriction digestion (PDX1 promoter with HinFI, DFI1 with EcoRV, ADAEC with XhoI and ECE1 with DpnI). Before digestion, chromatin was precipitated at room temperature by using 1 volume of isopropanol, and the pellet was recovered after centrifugation at 8,000 rpm for 10 min at room temperature and was washed twice with 70% alcohol at room temperature. The air dried pellet was solubilized in 1X universal Fast digest buffer, and 10 μl of respective restriction enzyme was added. Digestion was performed O/N at 37 °C. After completion of restriction digestion, chromatin was precipitated by absolute ethanol and dissolved in ChIP lysis buffer. ChIP experiment then was performed as described earlier.
Additional Information
Accession codes: GSE768558.
How to cite this article: Shariq, M. et al. The global regulator Ncb2 escapes from the core promoter and impacts transcription in response to drug stress in Candida albicans. Sci. Rep. 7, 46084; doi: 10.1038/srep46084 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Kumamoto, C. A. & Vinces, M. D. Contributions of hyphae and hypha-co-regulated genes to Candida albicans virulence. Cell Microbiol 7, 1546–1554 (2005).
Chandra, J. et al. Biofilm formation by the fungal pathogen Candida albicans: development, architecture, and drug resistance. J Bacteriol 183, 5385–5394 (2001).
Alby, K. & Bennett, R. J. Stress-induced phenotypic switching in Candida albicans. Mol Biol Cell 20, 3178–3191 (2009).
Wisplinghoff, H., Seifert, H., Wenzel, R. P. & Edmond, M. B. Current trends in the epidemiology of nosocomial bloodstream infections in patients with hematological malignancies and solid neoplasms in hospitals in the United States. Clin Infect Dis 36, 1103–1110 (2003).
Gudlaugsson, O. et al. Attributable mortality of nosocomial candidemia, revisited. Clin Infect Dis 37, 1172–1177 (2003).
Pappas, P. G. et al. A prospective observational study of candidemia: epidemiology, therapy, and influences on mortality in hospitalized adult and pediatric patients. Clin Infect Dis 37, 634–643 (2003).
Rex, J. H. et al. A randomized trial comparing fluconazole with amphotericin B for the treatment of candidemia in patients without neutropenia. Candidemia Study Group and the National Institute. N Engl J Med 331, 1325–1330 (1994).
Prasad, R., De Wergifosse, P., Goffeau, A. & Balzi, E. Molecular cloning and characterization of a novel gene of Candida albicans, CDR1, conferring multiple resistance to drugs and antifungals. Curr Genet 27, 320–329 (1995).
Coste, A. et al. A mutation in Tac1p, a transcription factor regulating CDR1 and CDR2, is coupled with loss of heterozygosity at chromosome 5 to mediate antifungal resistance in Candida albicans. Genetics 172, 2139–2156 (2006).
Dunkel, N., Blass, J., Rogers, P. D. & Morschhauser, J. Mutations in the multi-drug resistance regulator MRR1, followed by loss of heterozygosity, are the main cause of MDR1 overexpression in fluconazole-resistant Candida albicans strains. Mol Microbiol 69, 827–840 (2008).
Cannon, R. D. et al. Efflux-mediated antifungal drug resistance. Clin Microbiol Rev 22, 291–321, Table of Contents (2009).
Sa-Correia, I., dos Santos, S. C., Teixeira, M. C., Cabrito, T. R. & Mira, N. P. Drug:H+ antiporters in chemical stress response in yeast. Trends Microbiol 17, 22–31 (2009).
Morschhauser, J. Regulation of multidrug resistance in pathogenic fungi. Fungal genetics and biology: FG & B 47, 94–106, doi: 10.1016/j.fgb.2009.08.002 (2010).
Marie, C. & White, T. C. Genetic Basis of Antifungal Drug Resistance. Curr Fungal Infect Rep 3, 163–169 (2009).
Sanglard, D., Coste, A. & Ferrari, S. Antifungal drug resistance mechanisms in fungal pathogens from the perspective of transcriptional gene regulation. FEMS Yeast Res 9, 1029–1050 (2009).
Krishnamurthy, S., Gupta, V., Prasad, R. & Panwar, S. L. Expression of CDR1, a multidrug resistance gene of Candida albicans: transcriptional activation by heat shock, drugs and human steroid hormones. FEMS Microbiol Lett 160, 191–197 (1998).
Puri, N. et al. CDR1, a multidrug resistance gene from Candida albicans, contains multiple regulatory domains in its promoter and the distal AP-1 element mediates its induction by miconazole. FEMS Microbiol Lett 180, 213–219 (1999).
Chen, C. G., Yang, Y. L., Shih, H. I., Su, C. L. & Lo, H. J. CaNdt80 is involved in drug resistance in Candida albicans by regulating CDR1. Antimicrob Agents Chemother 48, 4505–4512 (2004).
Coste, A. T., Karababa, M., Ischer, F., Bille, J. & Sanglard, D. TAC1, transcriptional activator of CDR genes, is a new transcription factor involved in the regulation of Candida albicans ABC transporters CDR1 and CDR2. Eukaryot Cell 3, 1639–1652 (2004).
Karnani, N. et al. SRE1 and SRE2 are two specific steroid-responsive modules of Candida drug resistance gene 1 (CDR1) promoter. Yeast 21, 219–239 (2004).
Manoharlal, R., Gaur, N. A., Panwar, S. L., Morschhauser, J. & Prasad, R. Transcriptional activation and increased mRNA stability contribute to overexpression of CDR1 in azole-resistant Candida albicans. Antimicrob Agents Chemother 52, 1481–1492 (2008).
Tsao, S. et al. Positive regulation of the Candida albicans multidrug efflux pump Cdr1p function by phosphorylation of its N-terminal extension. The Journal of antimicrobial chemotherapy 71, 3125–3134, doi: 10.1093/jac/dkw252 (2016).
de Micheli, M., Bille, J., Schueller, C. & Sanglard, D. A common drug-responsive element mediates the upregulation of the Candida albicans ABC transporters CDR1 and CDR2, two genes involved in antifungal drug resistance. Mol Microbiol 43, 1197–1214 (2002).
Gaur, N. A. et al. Identification of a negative regulatory element which regulates basal transcription of a multidrug resistance gene CDR1 of Candida albicans. FEMS Yeast Res 4, 389–399 (2004).
Murad, A. M. et al. Transcript profiling in Candida albicans reveals new cellular functions for the transcriptional repressors CaTup1, CaMig1 and CaNrg1. Mol Microbiol 42, 981–993 (2001).
Talibi, D. & Raymond, M. Isolation of a putative Candida albicans transcriptional regulator involved in pleiotropic drug resistance by functional complementation of a pdr1 pdr3 mutation in Saccharomyces cerevisiae. J Bacteriol 181, 231–240 (1999).
Znaidi, S. et al. Genomewide location analysis of Candida albicans Upc2p, a regulator of sterol metabolism and azole drug resistance. Eukaryot Cell 7, 836–847 (2008).
Bannister, A. J. & Kouzarides, T. Regulation of chromatin by histone modifications. Cell research 21, 381–395, doi: 10.1038/cr.2011.22 (2011).
Struhl, K. Histone acetylation and transcriptional regulatory mechanisms. Genes Dev 12, 599–606 (1998).
Sellam, A. et al. Genome-wide mapping of the coactivator Ada2p yields insight into the functional roles of SAGA/ADA complex in Candida albicans. Mol Biol Cell 20, 2389–2400, doi: 10.1091/mbc.E08-11-1093 (2009).
Zawel, L. & Reinberg, D. Initiation of transcription by RNA polymerase II: a multi-step process. Prog Nucleic Acid Res Mol Biol 44, 67–108 (1993).
Roeder, R. G. The role of general initiation factors in transcription by RNA polymerase II. Trends Biochem Sci 21, 327–335 (1996).
Albert, T. K. et al. Global distribution of negative cofactor 2 subunit-alpha on human promoters. Proc Natl Acad Sci USA 104, 10000–10005 (2007).
Thomas, M. C. & Chiang, C. M. The general transcription machinery and general cofactors. Crit Rev Biochem Mol Biol 41, 105–178 (2006).
Chitikila, C., Huisinga, K. L., Irvin, J. D., Basehoar, A. D. & Pugh, B. F. Interplay of TBP inhibitors in global transcriptional control. Mol Cell 10, 871–882 (2002).
Geisberg, J. V., Holstege, F. C., Young, R. A. & Struhl, K. Yeast NC2 associates with the RNA polymerase II preinitiation complex and selectively affects transcription in vivo . Mol Cell Biol 21, 2736–2742 (2001).
Meisterernst, M. & Roeder, R. G. Family of proteins that interact with TFIID and regulate promoter activity. Cell 67, 557–567 (1991).
Inostroza, J. A., Mermelstein, F. H., Ha, I., Lane, W. S. & Reinberg, D. Dr1, a TATA-binding protein-associated phosphoprotein and inhibitor of class II gene transcription. Cell 70, 477–489 (1992).
Goppelt, A., Stelzer, G., Lottspeich, F. & Meisterernst, M. A mechanism for repression of class II gene transcription through specific binding of NC2 to TBP-promoter complexes via heterodimeric histone fold domains. Embo J 15, 3105–3116 (1996).
Butryn, A. et al. Structural basis for recognition and remodeling of the TBP:DNA:NC2 complex by Mot1. eLife 4, doi: 10.7554/eLife.07432 (2015).
Kamada, K. et al. Crystal structure of negative cofactor 2 recognizing the TBP-DNA transcription complex. Cell 106, 71–81 (2001).
Schluesche, P., Stelzer, G., Piaia, E., Lamb, D. C. & Meisterernst, M. NC2 mobilizes TBP on core promoter TATA boxes. Nat Struct Mol Biol 14, 1196–1201 (2007).
Shukla, S., Yadav, V., Mukhopadhyay, G. & Prasad, R. Ncb2 is involved in activated transcription of CDR1 in azole-resistant clinical isolates of Candida albicans. Eukaryot Cell 10, 1357–1366 (2011).
Creton, S., Svejstrup, J. Q. & Collart, M. A. The NC2 alpha and beta subunits play different roles in vivo . Genes Dev 16, 3265–3276 (2002).
Leach, M. D. et al. Hsf1 and Hsp90 orchestrate temperature-dependent global transcriptional remodelling and chromatin architecture in Candida albicans. Nature communications 7, 11704, doi: 10.1038/ncomms11704 (2016).
van Werven, F. J. et al. Cooperative action of NC2 and Mot1p to regulate TATA-binding protein function across the genome. Genes Dev 22, 2359–2369 (2008).
Palii, S. S., Chen, H. & Kilberg, M. S. Transcriptional control of the human sodium-coupled neutral amino acid transporter system A gene by amino acid availability is mediated by an intronic element. The Journal of biological chemistry 279, 3463–3471, doi: 10.1074/jbc.M310483200 (2004).
Pennacchio, L. A., Bickmore, W., Dean, A., Nobrega, M. A. & Bejerano, G. Enhancers: five essential questions. Nature reviews. Genetics 14, 288–295, doi: 10.1038/nrg3458 (2013).
Maston, G. A., Evans, S. K. & Green, M. R. Transcriptional regulatory elements in the human genome. Annual review of genomics and human genetics 7, 29–59, doi: 10.1146/annurev.genom.7.080505.115623 (2006).
Bonocora, R. P., Smith, C., Lapierre, P. & Wade, J. T. Genome-Scale Mapping of Escherichia coli sigma54 Reveals Widespread, Conserved Intragenic Binding. PLoS genetics 11, e1005552, doi: 10.1371/journal.pgen.1005552 (2015).
Barrett, L. W., Fletcher, S. & Wilton, S. D. Regulation of eukaryotic gene expression by the untranslated gene regions and other non-coding elements. Cellular and molecular life sciences: CMLS 69, 3613–3634, doi: 10.1007/s00018-012-0990-9 (2012).
Whitfield, T. W. et al. Functional analysis of transcription factor binding sites in human promoters. Genome biology 13, R50, doi: 10.1186/gb-2012-13-9-r50 (2012).
Liu, T. T. et al. Genome-wide expression and location analyses of the Candida albicans Tac1p regulon. Eukaryot Cell 6, 2122–2138 (2007).
Dhamgaye, S. et al. RNA sequencing revealed novel actors of the acquisition of drug resistance in Candida albicans. BMC genomics 13, 396, doi: 10.1186/1471-2164-13-396 (2012).
Sellam, A., Tebbji, F. & Nantel, A. Role of Ndt80p in sterol metabolism regulation and azole resistance in Candida albicans. Eukaryot Cell 8, 1174–1183 (2009).
Schubert, S. et al. Regulation of efflux pump expression and drug resistance by the transcription factors Mrr1, Upc2, and Cap1 in Candida albicans. Antimicrob Agents Chemother 55, 2212–2223, doi: 10.1128/AAC.01343-10 (2011).
Cannon, R. D. et al. Candida albicans drug resistance another way to cope with stress. Microbiology 153, 3211–3217 (2007).
Xie, J., Collart, M., Lemaire, M., Stelzer, G. & Meisterernst, M. A single point mutation in TFIIA suppresses NC2 requirement in vivo . Embo J 19, 672–682, doi: 10.1093/emboj/19.4.672 (2000).
Cang, Y. & Prelich, G. Direct stimulation of transcription by negative cofactor 2 (NC2) through TATA-binding protein (TBP). Proc Natl Acad Sci USA 99, 12727–12732, doi: 10.1073/pnas.202236699 (2002).
Spedale, G. et al. Tight cooperation between Mot1p and NC2beta in regulating genome-wide transcription, repression of transcription following heat shock induction and genetic interaction with SAGA. Nucleic acids research 40, 996–1008, doi: 10.1093/nar/gkr784 (2012).
Acknowledgements
MS is thankful to UGC for Dr. DS Kothari postdoctoral fellowship. RN acknowledges DST for providing the DST-INSPIRE senior research fellowship. The work has been supported by a grant from Department of Science and Technology (SB/SO/BB-130/2013).
Author information
Authors and Affiliations
Contributions
R.P., G.M., and M.S. conceived the experiments. M.S. S.D., R.N., N.G., and V.J. conducted the experiments. R.P., G.M., M.S., A.K.M., and A.M. analyzed the results. R.P. and M.S. wrote the manuscript. All author reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Shariq, M., Dhamgaye, S., Nair, R. et al. The global regulator Ncb2 escapes from the core promoter and impacts transcription in response to drug stress in Candida albicans. Sci Rep 7, 46084 (2017). https://doi.org/10.1038/srep46084
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep46084
This article is cited by
-
The negative cofactor 2 complex is a key regulator of drug resistance in Aspergillus fumigatus
Nature Communications (2020)
-
Identification of genome-wide binding sites of heat shock factor 1, Hsf1, under basal conditions in the human pathogenic yeast, Candida albicans
AMB Express (2018)
-
Identification of di-substituted ureas that prevent growth of trypanosomes through inhibition of translation initiation
Scientific Reports (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.