Abstract
Mycobacterium tuberculosis-infected macrophages and dendritic cells are limited in their ability to present antigen to CD4+ T cells suggesting that other mechanism of antigen presentation are driving the robust T cell response observed during an M. tuberculosis infection. These mechanisms could include antigens present in apoptotic bodies, necrotic debris, exosomes or even release of non-vesicular antigen from infected cells. However, there is limited data to support any of these mechanisms as important in driving T cell activation in vivo. In the present study we use Rab27a-deficient mice which show diminished trafficking of mycobacterial components to exosomes as well as M. tuberculosis strains that express recombinant proteins which traffic or fail to traffic to exosomes. We observed that exosomes released during a mouse M. tuberculosis infection contribute significantly to its T cell response. These finding imply that exosomes function to promote T cell immunity during a bacterial infection and are an important source of extracellular antigen.
Similar content being viewed by others
Introduction
Studies of animal models and tuberculosis (TB) patients indicate that a host mounts a robust T cell response to an M. tuberculosis (Mtb) infection and this response is essential for controlling the infection1. However, the mechanism(s) by which antigens are presented to T cells is still unclear. Possible mechanisms include direct antigen presentation by infected cells as well as uptake of necrotic cells or apoptotic bodies caring mycobacterial proteins. Recent studies suggest that “free” antigen can be released from infected cells and promote cross-priming2. Our published data also suggest that exosomes could play a role in Mtb antigen presentation3,4. Exosomes are membrane-bound vesicles of 30–150 nm in size that are released when multivesicular bodies (MVB) fuse with the plasma membrane releasing their intraluminal vesicles into the extracellular environment5,6. Exosomes generally function in intracellular communication and have been implicated in many physiological and pathological processes7. The release of exosomes has been implicated as a mechanism through which components derived from intracellular pathogens gain access to the immune system. Previous publications support this possibility as exosomes released from cells infected with intracellular pathogens such as Salmonella, Toxoplasma gondii, and M. tuberculosis, to name a few, contain pathogen components8. Furthermore, our earlier studies demonstrate that mice vaccinated with exosomes containing mycobacterial antigens can activate both CD4+ and CD8+ T cells and can protect these mice against infection to an extent comparable to M. bovis BCG3. Nevertheless, there is limited data to support any of the antigen presentation mechanisms as important in driving T cell activation in vivo and recent studies suggest that release of non-vesicular antigen from infected cells may in fact limit the T cell response9. Our inability to define the most relevant mechanisms of antigen presentation during the course of an Mtb infection stem, in part, from a lack of in vivo models where exosomes and other biological processes involved in antigen presentation can be blocked or modulated.
Previous published reports indicate that Rab27a and Rab27b may play an important role in exosome biogenesis at least in certain cell types10,11. Rab27 is a small molecular weight GTPase that is a member of the Ras GTPase superfamily. Through the use of a guanine-nucleotide dependent switch, they are known to regulate steps in membrane trafficking including: vesicle formation, vesicle trafficking, tethering, and fusion with target organelles12. Rab27a appears to mediate MVB docking to the plasma membrane during exosome biogenesis in FL3 and SLT4 metastatic cell lines, lung adenocarcinoma cells (A549) and the HeLa B6H4 tumor cell line10. Our present work extends these finding to macrophages where loss of Rab27a expression leads to diminished exosome production. These results suggest that Rab27a-deficient mice could serve as a useful model to evaluate exosome production during an infection. We found Mtb-infected Rab27a-deficient mice to have reduced exosome production and diminished activation of antigen-specific T cells as well as a diminished ability to control an Mtb infection compared to wild-type mice.
However, since Rab27a has been implicated in neutrophil degranulation as well as in other process that can impinge on immune function13,14, it was important to use additional approaches to evaluate exosomes as drivers of T cell activation during an in vivo Mtb infection. For this objective we generated BCG or Mtb H37Rv strains that expresses tagged DsRed or the mycobacterial protein HspX which differed in their trafficking to exosomes. When mice were infected with the different mycobacterial strains, increased T cell response to DsRed and HspX was observed when these proteins were targeted to exosomes. Altogether our data provides direct evidence for exosome-mediated T cell activation and suggest that cross presentation of antigen during an in vivo Mtb infection can be an important mechanism for eliciting an acquired immune response.
Results
Rab27a functions in exosome release in murine bone marrow-derived macrophages
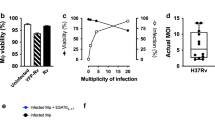
Rab27a had been identified in previous studies as an key regulator of MVB fusion with the plasma membrane, thereby regulating an important step in exosome biogenesis. However, the extent to which Rab27a mediates exosome release in macrophages had not been previously defined. To address this issue, bone marrow derived macrophages were isolated from Rab27a-deficient and wild-type C57BL/6 mice. Macrophages derived from Rab27a-deficient mice when infected with Mtb showed an 80% decrease in the number of exosomes in the culture media compared to the number of exosomes released from infected wild-type C57BL/6 macrophages (Fig. 1A,B). Furthermore, the protein markers found on exosomes released from Rab27a deficient cells may represent a unique subpopulation of exosomes whose biogenesis is mediated by alternative secretion mechanisms. As shown in Fig. 1C, the vesicles isolated from Rab27a-deficient macrophages featured a unique exosomal protein marker profile, notably a decreased CD63 expression which is consistent with previously reported studies10. Given that exosome secretion from Rab27a-deficient cells may represent a specific subpopulation, we sought to characterize the mycobacterial protein profile on these exosomes. Rab27a-deficient macrophages were infected with Mtb and probed for mycobacterial proteins using an antibody pool that was generated against Mtb culture filtrate proteins. We observed a general diminished presence of mycobacterial proteins but similar 19KDa lipoprotein concentration in/on exosomes secreted from Mtb infected Rab27a-deficient compared to wild-type macrophages (Fig. 1D). The loaded samples were normalized for protein concentration and therefore even a larger percentage of the total exosome material released from the Rab27a-infected relative to wild-type infected macrophages was used for the western blot. Importantly, we observed no difference in Mtb uptake or its survival between wild-type and Rab27a-deficient macrophages (Supplementary Figure 2). The diminished presence of mycobacterial proteins in exosome released from Rab27a-deficient macrophages suggest that these exosomes may have reduced immune system stimulatory activity. This prediction is supported by the observed limited TNF-α and RANTES production by macrophages treated with exosomes isolated from infected Rab27a-deficient macrophages as compared to exosomes from wild-type cells (Supplementary Figure 3).
Exosomes were isolated from the cell culture supernatant of C57BL/6 and Rab27a-deficient BMMs infected either with Mtb at a 3:1 MOI or left uninfected. Purified exosomes were (A) quantified for protein concentration by BCA and (B) for vesicle number by Nanosight analysis. Shown is a representative Nanosight profile for exosomes released from infected or uninfected wild-type or Rab27a-deficient macrophages. (C) 10 μg of exosomes were probed for the presence of exosomal markers, Lamp-1, CD63, CD81 and Tsg-101 by western blot. (D) 20 μg of exosomes from Mtb infected BMM were assayed for mycobacterial proteins using an antibody that recognizes multiple Mtb culture filtrate proteins (CFP) and an antibody against the Mtb 19 kDa lipoprotein. The BCA data is the average protein concentration across 3 independent experiments +/−SD and statistical analysis was performed comparing Rab27a-deficient to wild-type infected macrophages (*p < 0.05). The NanoSight and western blot data are representative of three independent experiments. Uncropped western blots are shown in Supplementary Figure 1.
Rab27a-defcient mice show reduced exosomes concentrations in serum, which correlates with an increase in bacterial burden and decreased T cell activation
Most exosome studies which define their contribution to an immune response have been done in the context of autoimmunity and cancer biology, while little has been done to elucidate their role in modulating an immune response to infectious diseases. We have previously shown that exosome production in M. bovis BCG-infected C57BL/6 mice correlated with bacterial load15. Given that Rab27a deficiency results in decreased exosome release from cells, we sought to characterize the progression of disease in Rab27a-deficient mice infected with mycobacteria. Rab27a-deficient and wild-type C57BL/6 mice were infected with 106 Mtb or M. bovis BCG by retro-orbital injection and the mice were sacrificed at different times post-infection. Rab27a-deficient mice infected with M. bovis BCG or Mtb showed a marked increase in bacterial burden over time when compared to wild-type infected mice (Fig. 2A and Supplementary Figure 4) and this correlated with lower concentration of exosomes found in the serum (Fig. 2B and Supplementary Figure 4). Furthermore, compared to wild-type infected mice, cells isolated from the lung and spleen of Mtb-infected Rab27a-deficient mice showed diminished IFN-γ production when stimulated ex-vivo with Mtb whole cell lysate (Fig. 2C). Spleen CD4+ T-cell activation as measured by CD69 expression was also significantly diminished 12-days post infection (Fig. 2D). Histopathology of lung sections were generated to identify lymphocyte infiltration (Supplementary Figure 5). There was no significant differences in overall pathology between Rab27a-deficient and wild-type C57BL/6 mice although a general trend toward increased pathology in the Rab27a-infected mice was observed (Supplementary Figure 5). Altogether these results suggest a diminished T-cell immune response in Rab27a-deficient mice.
Bacterial load in the spleen (A) and serum exosome concentration (B) were defined at different times post-infection. (C) Cells isolated from lung and spleens of infected mice at the times indicated were stimulated ex vivo with Mtb whole cell lysate and the amount of secreted IFN-γ quantified by ELISA. The results are expressed as the IFN-ɣ concentration after stimulation of 1 × 106 cells with the Mtb cell lysate. (D) Splenocytes were isolated from wild-type C57BL/6 and Rab27a-deficient mice at different times post-infection with Mtb. The cells were stained with PE-conjugated anti-mouse CD69 and CD4 or with an isotype control antibody. CD4+ T cells were analyzed for CD69 surface expression with a Beckman Coulter flow cytometer, and the percentage of CD69 positive relative to total CD4+ cells was calculated. Results are defined for each mouse with +/−SD and statistical analysis was performed comparing Rab27a-deficient mice to wild-type infected mice (*p < 0.05). The data is representative of three independent experiments for a total of 9 mice per mouse strain per time point.
Exosomes isolated from M.tb infected Rab27a-deficient mice show a reduced capacity to elicit a pro-inflammatory response
Given our in vitro study which showed that exosomes derived from Rab27a-deficient macrophages have a diminished capacity to induce macrophage production of pro-inflammatory cytokines, we addressed whether this was also true for the in vivo derived exosomes. Exosomes were purified from serum of Rab27a-deficient and wild-type C57BL/6 infected mice and PBS-treated mice at days 10 and 20 post-infection. Pooled exosomes from these time points were used to treat BMMs. Serum-derived exosomes from wild-type and Rab27a-deficient mice were normalized to protein concentration (250 μg/mL, equal to ~2.5 × 1010 exosomes) and the BMMs were treated for 16 hours or left untreated. Supernatants were assayed for cytokine and chemokine levels, using a mouse cytokine array kit and pixel intensities were defined and plotted (Fig. 3A). Exosomes derived from the serum of wild-type C57BL/6 infected mice induced a significantly higher level of CCL1, IFN-γ, RANTES, MIP-2, IL1aR, and TNF-α as compared to exosomes isolated from infected Rab27a-deficient mice. To confirm the array results, BMMs were again treated with exosomes isolated from Rab27a-deficient or wild-type infected mice and the BMM supernatants were harvested 16 hours post-infection and analyzed for TNF-α concentration by ELISA. The results were comparable to the cytokine array, showing an approximate 30% reduction in TNF-α secreted from macrophages treated with exosomes isolated from infected Rab27a-deficient mice compared to infected wild-type mice (Fig. 3B). These results suggest that exosomes released from infected Rab27a-deficient mice are less pro-inflammatory.
Serum exosomes from Mtb-infected wild-type and Rab27a-deficient mice (3 mice/group) were purified and used at 250 μg/ml to treat BMMs for 16 hours. (A) The supernatants were harvested and assayed for specific proteins using a mouse cytokine array. The pixel densities for each spot of the array were calculated using ImageJ software and plotted. Results are defined for each individual mouse +/− SD and statistical analysis was performed comparing Rab27a-deficient mice to WT infected mice (*p < 0.05). (B) The culture supernatants were also analyzed for TNF-α by ELISA. The results are the mean of three separate experiments (9 mice total) with SD shown. Statistical analysis was performed comparing Rab27a-deficient to wild-type infected mice (*p < 0.05). Exo; exosomes. Rab; Rab27a.
Macrophage infected with M. bovis BCG expressing Ag85A-DsRed release exosomes containing the fusion protein
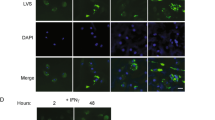
The interpretation of the Rab27a mouse experiments is complicated by Rab27a’s described role in other immune system functions16. Therefore, to further evaluate exosomes potential function in stimulating an antigen-specific T cell response during an in vivo Mtb infection, we generated a recombinant M. bovis BCG strains that expresses an Ag85A-DsRed fusion protein (Fig. 4A,B). Ag85A is a crucial component of the antigen 85 enzyme complex that functions as an acyltransferases and converts TMM (Trehalose monomycolate) to TDM (Trehalose dimycolate), and was previously identified in exosomes isolated from mycobacteria-infected macrophages4. In the recombinant BCG strain, Ag85A served as an exosome-targeting signal to facilitate DsRed trafficking to exosomes. M. bovis BCG strain expressing DsRed alone was also generated for this study. Both strains showed indistinguishable growth curves. As presented in Fig. 4 both the DsRed and Ag85A-DsRed were expressed by the recombinant M. bovis BCG. Moreover, both proteins were present in the macrophage cell lysate following infection with the recombinant M. bovis BCG (Fig. 4C). However, the DsRed was present in exosomes at significantly higher concentration in cells infected with BCG expressing the Ag85A-DsRed fusion protein compared to cells infected with BCG expressing DsRed alone. Interestingly there appeared to be some cleavage of the linker between Ag85A and DsRed within the exosome as we observed both intact fusion protein as well as DsRed alone in the exosomes. Exosomes derived from mammalian cells may be discriminated from other types of extracellular vesicles using various surface biomarkers including CD63 and CD917. To confirm that the DsRed-containing vesicles were exosomes, CD63-positive vesicles were captured on Protein G-conjugated sepharose resins and then analyzed by fluorescence microscope. As shown in Fig. 4D,E, CD63 positive extracellular vesicles released from macrophages infected with M. bovis BCG expressing Ag85A-DsRed were positive for DsRed fluorescence. No detectable fluorescence was observed for vesicles released from macrophages infected with M. bovis BCG expressing DsRed alone. Altogether the data indicates that Ag85A can be used to significantly increase transport of DsRed to exosomes. Therefore M. bovis BCG expressing either Ag85A-DsRed or DsRed provides a new tool to better address the importance of exosomes in driving a T cell response during an Mtb infection.
(A) Fluorescence microscopic and (B) western blot analysis of recombinant M. bovis BCG expressing DsRed or Ag85A-DsRed. (C) Western blot analysis of exosomes and RAW264.7 whole cell lysate (WCL) following a 72 hour infection with the recombinant M. bovis strains or from unifected cells. Tubulin and Lamp-1 were used as loading control for WCL and exosomes respectively. (D) Fluorescence microscopic analysis of Ds-Red positive exosomes captured on protein G Sepharose beads coupled to anti-CD63. (E) Quantitation of the DsRed-positive Sepharose beads observed in (D). Uncropped western blots are shown in Supplementary Figure 1.
Exosomes contribute to a DsRed specific T-cell response in mice infected with M. bovis BCG expressing Ag85A-DsRed
Wild-type C57BL/6 mice were intranasally infected with the M. bovis BCG strains expressing DsRed or Ag85A-DsRed and the number of DsRed-specific T cells that produce IFN-ɣ was measured by ELISPOT. As shown in Fig. 5A, spleen and lung cells isolated from mice following a two week infection with M. bovis BCG expressing Ag85A-DsRed contained a significantly higher number of IFN-ɣ positive T cells after ex vivo DsRed stimulation compared to cells isolated from mice infected with DsRed-expressing BCG. To exclude the effect of other antigen-delivering pathways, Rab27a-deficient mice were used. Analogous to our results with the Mtb antigens, the absence of functional Rab27a impaired the delivery of Ag85A-DsRed to exosomes. The abundance of Ag85A-DsRed in exosomes isolated from Rab27a deficiency BMMs was 5-fold lower than observed in exosomes from wild type BMMs (Supplementary Figure 6). Therefore we hypothesized that Rab27a-deficient mice would show a limited T cell response to DsRed even when infected with M. bovis BCG expressing Ag85A-DsRed. As predicted, spleen and lung cells isolated from Rab27a-deficient mice infected with Ag85A-DsRed expressing M. bovis BCG showed a reduced number of T cells producing IFN-ɣ upon DsRed stimulation when compared to similarly infected wild-type mice (Fig. 5B). Further, we observed no significant difference in the number of IFN-ɣ positive T cells following ex vivo DsRed stimulation when we used spleen or lung cells isolated from Rab27a-deficient mice that were infected with recombinant M. bovis BCG expressing either Ag85A-DsRed or DsRed alone (Fig. 5B). These results show that host-derived exosomes can function in promoting an antigen specific T cell response during a M. bovis BCG mouse infection.
(A) Wild-type mice were intranasally infected with M. bovis BCG expressing DsRed or Ag85A-DsRed and the lung and splenic cells were harvested 2 weeks post-infection and stimulated ex-vivo with DsRed. The number of IFN-ɣ producing T cells responding to the DsRed antigen was measured by ELISPOT. (B) Wild-type C57BL/6 or Rab27a-deficient mice were intranasally infected with M. bovis BCG expressing either DsRed or Ag85A-DsRed. The number of IFN-ɣ producing T cells responding to the antigen DsRed was determined by ELISPOT. Results are defined for each mouse (4 mice/group) +/−SD and statistical analysis was performed between BCG expressing DsRed or Ag85A-DsRed (A) or between infected WT and Rab27-deficient mice (B) (*p < 0.05). The data is representative of two independent experiments.
Targeting HspX to exosomes during an M.tb mouse infection promotes the T cell response to this mycobacterial antigen
While the BCG experiments indicate that exosomes can promote an antigen-specific T-cell response, we sought to develop a model to evaluate whether exosomes can promote T-cell activation to a mycobacterial protein during an Mtb infection. Previous studies have shown that the mycobacterial protein heat shock protein X (HspX) is present in exosomes isolated from human TB patient serum18. Recent studies have shown that trafficking HspX to exosomes was dependent upon mono-ubiquitination of a lysine residue at position 85. Therefore, we expressed in M. smegmatis an HspX protein with a Lysine to Arginine amino acid substitution at position 85 as well as wild-type HspX. Although expressed in M. smegmatis at similar levels to wild-type HspX, the lysine mutant was not trafficked to exosomes19. Therefore, we transformed into an ΔHspX strain of H37Rv, expression clones containing the HspX gene coding for either wild-type or the K85R mutant protein. Similar to our observation with M. smegmatis, wild-type HspX but not the K85R mutant was trafficked to exosomes in macrophages infected with the transformed ΔHspX H37Rv (Fig. 6A). Importantly both wild-type and K85R HspX were expressed and could be detected within the infected macrophage cell lysates. As a control, macrophages were treated with recombinant His-tagged HspX and as observed previously19, the HspX was endocytosed and trafficked to exosomes (Fig. 6A). Since our experiments demonstrated that K85R HspX is expressed by the transformed H37Rv but not targeted to exosomes we retro-orbitally infected C57BL/6 mice with the various H37Rv strains and the number of splenic T-cells which produced IFN-ɣ upon ex vivo stimulation with purified HspX was measured by ELISPOT. Splenocytes isolated 10-days post-infection with the H37Rv expressing the K85R HspX showed a significantly reduced number of IFN-γ producing T-cells (~35% decrease) compared to splenocytes isolated from mice infected with H37Rv expressing wild-type HspX (Fig. 6B). At 15 days post-infection the difference in the T-cell IFN-γ recall response was limited to ~15% but was still statistical significant (Fig. 6D). However, by day 21 post-infection there was no statistical difference in IFN-γ production between HspX-stimulated splenocytes isolated from mice infected with Mtb expressing either wild-type or K85R HspX. To ensure that differences in the observed T-cell immune responses were not due to differences in the Mtb strains, splenocytes were isolated 10 or 15 days post-infection and stimulated ex vivo with Mtb whole cell lysate and the number of IFN-γ producing cells quantified. As shown in Fig. 6C and E all the splenocyte populations isolated from mice infected with the different H37Rv strains showed a similar response when exposed ex vivo to a mixed population of Mtb antigens. Moreover, in contrast to wild-type C57BL/6 mice, we detected no statistical difference in IFN-γ production by splenocytes following ex-vivo stimulation with HspX or Mtb whole cell lysate when we used Rab27a-deficient mice for the 10 day infection (Fig. 6F,G). Our findings were not limited to the spleen as lung cells isolated from mice infected intratracheally with H37Rv expressing the K85R HspX showed a significantly reduced number of IFN-γ producing T-cells (~33% decrease) compared HspX-stimulated lung cells isolated from mice infected with H37Rv expressing wild-type HspX (Supplementary Figure 7).
(A) RAW264.7 cells were infected with ∆HspX H37Rv or ∆HspX H37Rv expressing either His-tagged wild type HspX or K85R HspX and 72 hours post-infection exosomes were purified from the culture media. Raw264.7 cell lysates were also obtained. A separate well of Raw264.7 cells were treated with 40 μg/ml of purified His-tagged HspX (rHspX) and 24 hours post-treatment culture media and cell lysates were obtained. 10 μg of purified exosomes or cell lysates was analyzed for the His-tagged HspX using an anti-His antibody. Blots were also probed for Lamp1 as a loading control. (B–E) Splenocytes were harvested from wild-type C57BL/6 mice 10 days (B,C) or 15 days (D,E) post infection or from Rab27a-deficient mice 10 days post-infection (F,G). Mice (3 to 4 mice/group) were infected with either 106 CFU of wild-type H37Rv, ∆HspX H37Rv or ∆HspX H37Rv expressing either wild-type HspX or K85R HspX. The splenocytes were analyzed 20 h after ex vivo stimulation with 5 μg/ml HspX (B,D,F) or 10 μg/ml Mtb CFP (C,E,G). The number of T cells producing IFN-ɣ upon HspX antigen stimulation was determined by ELISPOT and the number of positive cells counted +/−SD between individual mouse infections. Significance between samples is indicated (*p < 0.05). Data is representative of three independent experiments. Uncropped western blots are shown in Supplementary Figure 1.
Discussion
Although there are approximately 2 billion people infected with Mtb, less than 10% of those infected will develop active disease in their life-time. This implies that in most individuals the immune response is sufficient to mediate protection. To understand this protective immune response and how it potential fails, requires a thorough knowledge of how PAMPs and Mtb antigens intersect with immune cells. Previous studies have demonstrated that exosomes released from infected macrophages are functionally associated with both the innate and acquired immune system. Exosomes are vesicles of 30–150 nm in size that are secreted from most nucleated cells and have been shown to function in intracellular communication. In the context of Mtb infection, we have identified a significant number of mycobacterial proteins on exosomes4,20. We have also shown that exosomes are immunogenic21,22. Exosomes derived from M. bovis BCG or Mtb infected macrophages upon adaptive transfer can activate antigen-specific CD4+ and CD8+ T cells in vivo and promote maturation of bone-marrow derived dendritic cells ex vivo21. Furthermore, we have shown that exosomes containing TB antigens can be used as an effective vaccine against an aerosolized Mtb infection in a mouse model3. While this data suggests exosomes can stimulate a T-cell immune response, there are several other processes through which antigen-specific T-cells may be primed including necrotic cells, apoptotic bodies, and free soluble antigen2,23,24. In the present study we developed new tools through which we could address the role of exosomes in stimulating innate and acquired immune responses during a natural infection.
One of the challenges in defining exosome function during the course of a natural infection is the lack of a mouse model in which exosome biogenesis is specifically inhibited. This is, in part, due to the fact that much of the machinery required for exosome biogenesis, such as the endosomal sorting complexes required for transport (ESCRT) machinery, mediates other important cellular processes and the loss of such machinery is embryonic lethal or has off target effects on cell/organ function. In the present study, we use a Rab27a-deficient mouse strain that was generated through a spontaneous mutation in the parental C3H strain and was back-crossed into the C57BL/6 background25. Rab27a, a member of the Rab GTPases, is known to mediate MVB fusion to the plasma membrane during exosome secretion10, although this may be cell-type specific. To evaluate Rab27a in the context of exosome biogenesis in macrophages, we compared exosome production by Rab27a deficient and wild-type bone marrow-derived macrophages and found significantly lower exosome concentration in the culture media of Rab27a-deficient macrophages. Interestingly, we observed an even greater difference in exosome concentration between wild-type and Rab27-deficient macrophages following an Mtb infection. This was due to an elevated concentration of exosomes released upon infection of wild-type macrophages while no significant increase was observed for infected Rab27a-deficient macrophages. Similar results were observed in vivo as serum concentration of exosomes increased only ~5 fold after a 40 day Mtb infection of Rab27a-deficient mice compared to an ~15 fold increase for wild-type mice infected with Mtb. It is likely that the decreased exosome concentration observed in the serum of infected Rab27a-deficient mice is due to combined decrease in exosomes released from infected macrophages as well as from other cell types whose exosome release is dependent on Rab27a. However, which cell types are producing exosomes during the course of an Mtb infection awaits further investigation.
In addition to decrease exosome production, the population of exosomes secreted from Mtb infected Rab27a-deficienct macrophages showed a reduced level of mycobacterial proteins and reduced capacity to stimulate TNF-alpha and RANTES production. Analysis of host exosomal markers showed that the exosomes secreted from the Rab27a deficient macrophages were CD81+ but CD63-, in part, reflecting an early study by Bobrie et al. which demonstrated Rab27a deficiency results in the secretion of exosomes that have altered host protein composition, most notably reduced CD63 expression26. At present, it is unclear whether the decreased expression of mycobacterial proteins in Rab27a-deficient exosomes reflects the inability of the macrophage to effectively secrete exosomes, or rather represents a distinct subpopulation of exosomes generated through a unique biogenesis and protein sorting pathway.
Altogether our in vivo and in vitro data using the Rab27a deficient macrophages and mice suggest a role for this GTPase in activation of both the innate and acquired immune response following an Mtb infection. This decreased immune response correlates with diminished release of exosomes and limited transport of mycobacterial proteins to exosomes. This suggest that the effects of Rab27a deficiency on the immune response to Mtb stems in part from its effect on exosome production/composition. However, while it is known that Rab27a plays a role in exosome biogenesis, Rab27a deficiency is also associated with immune system dysregulation16. For example, it is known that Rab27a mediates exocytosis in neutrophils, CTLs, NK cells and mast cells, which in the context of immunity, is important for the secretion of antimicrobials. Rab27a associated defects in degranulation may lead to defects of adhesion, migration, chemotaxis of cells and defects in neutrophil mediated phagocytosis and reactive oxygen species production. However, characterization of the CD4+ T-cell responses in Rab27a- deficient mice have shown that the deficiency does not affect CD4+ T-cell proliferation and maturation and that T-cell receptor mediated IFN-y production is normal27. We also observed no difference in the number of T cells present in the spleens of Rab27a-deficient and wild-type mice prior to infection (data not shown). Therefore, we believe the model and the initial data establishes exosomes as a potential mediator of T-cell activation.
Nevertheless, as an alternative approach to evaluate exosomes in promoting T cell activation we developed a recombinant M. bovis BCG strain in which the fluorescent reporter protein DsRed was expressed either alone or fused to Ag85A. In this context, we used Ag85A as a protein to target DsRed to exosomes as we have previously shown Ag85A to be a major constituent of exosomes isolated from infected macrophages, mice and human TB patients4,18,20. Our BCG infection experiments indicate that targeting DsRed to exosomes enhanced the T cell response to this protein antigen. However, T-cell activation may also be mediated through other mechanisms such as free-soluble antigen or apoptotic blebs. Furthermore, a more recent study by Athman et al. suggests that bacterial membrane vesicles secreted directly from Mtb function to disseminate mycobacterial proteins which can subsequently interact with the immune system28. In order to exclude these other mechanisms of T-cell activation against DsRed, we applied our M. bovis BCG expressing Ag85A-DsRed or DsRed alone to our Rab27a-deficient mouse which as indicated previously has a markedly diminished exosome production relative to wild-type macrophages or mice. The number of DsRed-specific T cells was comparable in Rab27a-deficient mice whether they were infected with recombinant M. bovis BCG expressing Ag85A-DsRed or expressing DsRed alone. To address exosomes as carriers of antigen for T cell activation more directly and in the context of an Mtb infection, we generated an H37Rv strain that expressed either WT HspX or a mutant HspX which is not trafficked to exosomes upon macrophage infection19. When mice were infected with Mtb expressing WT HspX we observed a significantly higher number of IFN-ɣ positive T cells specific to HspX when compared to mice infected with Mtb expressing the K85R HspX. Interestingly, this exosome mediated T cell activation may be more relevant during the initial T cell response as we observed a diminished role for exosomes at 15 and 21 days post-infection relative to 10 days. Moreover, as observed with the BCG expressing Ag85a-DsRed, we again detected no significant difference in the production of IFN-γ by splenocytes isolated from Rab27a-deficient mice infected with H37Rv expressing either WT or K85R HspX, further suggesting that exosomes are driving a T cell response during a natural Mtb infection.
Although our data indicates that exosomes produced during a mouse Mtb infection can promote a T cell response, this is clearly only one of a number of mechanisms by which antigens can be made available for presentation to T cells. Recent studies by Srivastava et al. suggest that blocking release of non-vesicular “free” antigen from infected macrophages promotes antigen presentation by infected DC both in vitro and in vivo9. The authors suggest that releasing antigen out of infected DCs and macrophages is a virulence mechanism used by Mtb to limit T cell activation. However, it is unclear from these experiments how much of Mtb antigens are presented through cross-priming compared to presentation by infected cells during a natural infection. Our data shows that cross-presentation through exosomes occurs during an Mtb infection and our Rab27a data suggest that limiting exosome-mediated immune response leads to higher bacterial burden. Since greater than 90% of individuals infected with Mtb mount a T cell response sufficient to control the infection and the established observation that infected DCs/macrophages are poor presenters of antigen, it suggest that the various mechanisms of antigen cross-presentation including exosomes are required for the protective adaptive immune response. It is also possible that individual variations in exosome-mediated or other mechanisms of antigen presentation can be an important factor in those individuals who succumb to TB.
In summary, our data indicates that exosomes can mediate immune system activation during an in vivo infection. Furthermore, we demonstrate that exosomes enhance T-cell activation during an Mtb infection. However, the importance of exosome-mediated antigen delivery compared to other mechanisms of antigen presentation requires additional study and may vary depending on the stage or route of infection as well as on which antigen is being evaluated and its distribution inside the infected host cell.
Methods
Ethics Statement
The University of Notre Dame is accredited through the Animal Welfare Assurance (#A3093-01) and follows the guidelines described in “The Guide for the Care and Use of Laboratory Animals” and the “United States Department of Agriculture Animal Welfare Act and Animal Welfare Regulations”. All animal procedures were approved by the Institutional Animal Care and Use Committee (approval # 14-08-1969).
Animals
All wild type C57BL/6 mice and Rab27a-deficient mice in a C57Bl/6 background were housed at the institutional animal facility under specific-pathogen-free conditions during the experiments. The Rab27a-deficient mice were generously provide by Dr. Sergio Catz, Scripps Research Institute, CA. M. bovis BCG infections were carried out in the biosafety level-2 laboratory and Mtb infections were performed in a biosafety level-3 laboratory.
Bacteria and and Macrophage Cell Lines
M. bovis BCG and M. tuberculosis H37Rv strains were grown in Middlebrook 7H9 broth medium (Difco, Becton-Dickinson) containing 10% OADC (oleic acid/albumin/dextrose/catalase, 0.05% Tween 80) until exponential phase and then aliquoted and stored at −70 °C until use. Prior to use, the bacterial stocks were thawed and the mycobacteria were de-clumped by a brief sonication and passed through a syringe fitted with a 27-gauge needle at least 10 times. The macrophage cell line RAW 264.7 (ATCC, Manassas, VA) was maintained in Dulbecco modified Eagle’s minimal essential medium (DMEM, Cellgro, Manassas, VA) supplemented with 10% fetal bovine serum (Hyclone, South Logan, Utah), 25 mM Na-HEPES (ThermoScientific, Rockford, IL), 1 mM Sodium pyruvate (Lonza, Walkersville, MD), 100 U/mL penicillin and 100 U/mL streptomycin (Hyclone, South Logan, Utah) at 37 °C with 5% CO2. Bone marrow-derived macrophages (BMMs) were prepared from wild type C57BL/6 or Rab27a deficiency mice as described previously29.
Construction of Plasmids
To generate plasmid pMV261-Ag85A::DsRed, the DNA fragment encoding DsRed was amplified by PCR from plasmid, pMSP12-DsRed2, using Easy-A high-fidelity DNA polymerase (Agilent Technologies, CA) and primers DsRed-Forward (NheI), 5′-CTT GCT AGC ATG GCC TCC TCC GAG AAC GT-3′, and DsRed-Reverse (Sna BI), 5′-CTT TAC GTA CTA CAG GAA CAG GTG GTG GC-3′. Ag85A ORF was amplified from M. tuberculosis genomic DNA with the primers Ag85A-Forward (BamHI), 5′-CTT GGA TCC ATG CAG CTT GTT GAC AGG GTT CG-3′, and Ag85A-Reverse (NheI), 5′-CTT GCT AGC AGG TCC GGC GCC CTG GGG CGC G-3′. The PCR products were cloned into pGEM-T easy vector to generate plasmids pGEM-DsRed and pGEM-Ag85A, respectively. The DsRed fragment was cut from pGEM-DsRed with restriction endonucleases NheI and NdeI and inserted into plasmid pGEM-Ag85A at the same sites to create plasmid, pGEM- Ag85A::DsRed. Finally, the Ag85A::DsRed fragment was cut from pGEM- Ag85A::DsRed with restriction endonucleases BamHI and SnaI and cloned in the vector pMV261.hgy at the sites BamHI and HpaI to generate plasmid pMV261-Ag85A::DsRed. To generate plasmid pMV261-DsRed, the ORF of DsRed was amplified from plasmid,pMSP12-DsRed2 with primers DsRed-Forward (BamHI), 5′-CTT GG ATCC ATG GCC TCC TCC GAG AAC GT-3′, and DsRed-Reverse (Sna BI), 5′-CTT TAC GTA CTA CAG GAA CAG GTG GTG GC-3′. The PCR product was cloned into pGEM-T Easy vector to create plasmid pGEM-DsRed, and then the DsRed fragment was cut from pGEM-DsRed with restriction endonucleases BamHI and SnaI and cloned into the vector pMV261.hgy at the sites BamHI and HpaI to generate plasmid pMV261-DsRed.
The pMV261-HspX and pMV261-HspX mutant plasmids construction was generated as previously described19.
Electroporation of Mycobacteria
M. bovis BCG and Mtb were grown in Middlebrook 7H9 broth plus 10% OADC at 37 °C until OD600 at a range of 0.5–1.0. The cells (50 ml culture) were washed with 50 ml 10% glycerol three times at 2000 × g, 15 min, RT and cell pellet was finally resuspended in 500 μl 10% glycerol. For transformation, 200 μl competent mycobacteria were gently mixed with 2 μg of plasmid DNA for BCG or with 1 μg of plasmid DNA for Mtb and then incubated in ice 30 min. The DNA-Cell mixture was transferred into a 0.2 cm electroporation cuvette and pulsed with BioRad GenePulser Xcell™ Electroporation Systems set to 2.5 kV, 1000 ohms and 25 μF. The cells were then transferred into a 15 ml snap cap tube containing 5 ml Middlebrook 7H9 broth containing 10% OADC and incubated at 37 °C over-night. The transformation was than plated on Middlebrook 7H10 agar plates containing 10% OADC and 50 ug/ml hygromycin and incubated at 37 °C for 3–4 weeks.
Exosome Preparation and Purification
RAW 264.7 cells or BMMs were infected with M. bovis BCG strains or Mtb as described previously15. After 72 hours in exosome-depleted medium, cell culture supernatant was collected and centrifuged at 350 × g for 15 min at 4 °C to remove cells and large debris. The supernatant was then passed through a 0.22 um polythersulfone filter (Corning, NY, USA), followed by centrifugation at 10,000 × g for 1 hour at 4 °C. The culture supernatant was further ultra-centrifuged at 100,000 × g for 1 h at 4 °C to spin down expected exosomes. The pellets were resuspended in PBS and washed 3X with PBS. The final pellets were resuspended in PBS and stored at −80 °C.
NanoSight Analysis
The exosomes were characterized for size distribution and quantitated by NanoSight LM10, using light scatter from the 635-nm red laser, as well as NTA 2.3 Analytical software as described30.
Cytokine Array and TNF-alpha, RANTES and IFN-ɣ ELISA
Exosomes were isolated from H37Rv-infected WT or Rab27a infected mice or PBS-injected control mice. Primary bone marrow macrophages (1 × 106 cells) were stimulated with the serum-derived exosomes at 250 mg/ml for 16 hours. The cell culture supernatants were harvested, and particulates were removed by centrifugation. The supernatants were tested immediately for cytokine/chemokine levels, using the Mouse Cytokine Array Panel A kit (R&D Systems, Minneapolis, MN) according to the manufacturer’s instructions. Briefly, cell culture supernatants were mixed with a blend of biotinylated detection antibodies (Abs) and incubated with the nitrocellulose membrane that contains 40 different anti-cytokine/chemokine capture Abs spotted in duplicate. Any cytokine/Ab complex formed is bound to the immobilized capture Ab. The membranes were incubated with chemiluminescent substrate (Thermo Scientific [Pierce]. In separate experiments, the cell culture supernatants from the exosome treated macrophages were also tested for TNF-alpha by ELISA (Biosource) per the manufacturer’s instructions. RANTES levels in exosome treated macrophages was measured by ELISA per manufacturer’s instructions (R&D Systems, Camarillo, CA).
Wild type C57BL/6 mice and Rab27a-deficient mice were retro-orbitally infected with WT Mtb H37Rv, and cells from lungs and spleens of mice were harvested at various time points post-infection as described previously3. Isolated cells were stimulated ex vivo with 5 μg/ml Mtb whole cell lysate at 37 °C with 5% CO2. After 72 h, INF-γ released in cell-free culture supernatant was measured by ELISA (eBioScience, San Diego, CA) according to the manufacturer’s instruction.
Histology of lung sections
The lung sections were fixed in 10% neutral buffered formalin overnight and then transferred to 70% ethyl alcohol. The samples were processed using a Shandon Citadell 2000 automated tissue processor. After fixation, samples were dehydrated, embedded in paraffin, and sectioned at 4 mm, using a Leica RM 2155 automated microtome. Sections were H&E stained to examine general tissue and cellular morphology. Individual sections were scored for pathology as described3.
Western blots
Exosomes (10 μg or 109 particles) were resuspended in PBS with protease inhibitors. The suspension was mixed with Laemmli buffer, heated at 95 °C for 5 min, and chilled on ice for 5 min before loading onto SDS gel. Immunoblots probed with antibodies for proteins: Tsg-101 (C-2, 1:1000, Santa Cruz), CFP (C192, 1:1000, ATCC), 19kDa-lipoprotein (IT-19, 1:1000, ATCC), His-tag (1:500, Santa Cruz), CD81 (1:500, SBI), CD63 (1:500, Systems Bioscience), and, DsRed (632393, 1:1000, Clontech). Primary antibody incubation was followed with HRP-conjugated secondary antibodies (1:25,000, Pierce) and detected using enhanced chemiluminecence kit (Pierce). As loading controls, primary mouse mAbs against either alpha-Tubulin, 1/1000 dilution (Cat. T9026, Sigma) or Lamp-1 1/500 dilution (SC-17768, Santa Cruz Biotechnology or 1D4B, DHSB) were used.
Macrophage Infection and CFU determination
Mycobacteria were thawed and incubated with 10% horse serum for 2 hours at 37 °C to opsonize bacteria for infection. Rab27a or WT BMMs were seeded at 106 cells/well in a 6-well plate. Bacteria were added to the cells at indicated concentrations and were allowed to infect for 4 hours at 37 °C. Cells were washed three times with phosphate-buffered saline (PBS) before fresh complete media without antibiotics was added. Macrophages were lysed immediately after the 4 hour infection or 76 hours post infection. Bacteria was plated on 7H11 agar and CFUs were determined after 4 weeks at 37 °C.
Mouse Infection and CFU determination
C57BL/6 mice of Rab27a-deficient mice (8–10 weeks old) were infected retro-orbitally with H37Rv expressing wild-type or mutant HspX at (106 bacilli per mouse) or injected with an equal volume of PBS. Mice were sacrificed at 10, 15 and 21 days post infection (three to four mice per group), and serum and spleens were collected. Exosomes were purified from mouse serum. Spleens from infected mice were homogenized and passed through a 70-mm cell strainer. The cell suspension was treated with RBC lysis buffer, followed by PBS washes. The suspension was serially diluted in PBS+ Tween 80 (0.05% v/v) and plated on 7H10 agar supplemented with oleic albumin dextrose catalase. CFUs were determined after 4 weeks at 37 °C.
For BCG infection, WT C57BL/6 mice and Rab27a-deficient mice (8–10 weeks old) were intranasally immunized with M. bovis BCG expressing DsRed or Ag85A-DsRed (1 × 106 CFU/mouse) or PBS alone at an injection volume of 30 μl (15 μl/nostril) as described previously30. There were four mice per treatment group. Mouse lungs and spleens were isolated two weeks post-infection and used for ELISPOT analysis.
Flow Cytometry
The cells were rinsed with Dulbecco’s PBS and gently scraped and counted on a hemacytometer, using trypan blue to assess viability. The cells were washed in FACS buffer and blocked with 10% mouse serum and stained with PE-conjugated anti-mouse CD69 (BD Pharmingen, San Diego, CA), FITC-conjugated CD4, or using isotype Abs as controls. Cells were analyzed for protein surface expression with a Beckman Coulter flow cytometer, and the percentage of positive cells was calculated.
Isolation of Lymphocytes and ELISPOT
Mouse lung and spleen cells were isolated after an Mtb, or M. bovis BCG infection as described previously3. The IFN-ɣ assay was performed using the Mouse IFN-ɣ ELISPOT Ready-SET-Go! Kit (eBioScience, CA) following the manufacturer’s protocol. Spleen and lung cells (5 × 105 cells/well) were analyzed 20 h after ex vivo re-stimulation with recombinant DsRed (STA-202, Cell Biolabs, INC) or HspX (Colorado State University) at a final concentration of 5 μg/ml or M. tuberculosis CFPs at 10 μg/ml.
CD63-conjugated Bead-binding Assay
Protein G-Sepharose 4B resins (Cat. 10–1241, Invitrogen, CA) was first washed and incubated with blocking buffer (cat. 00-4202-56, ebioscience, CA) on a rotator at 4 °C for 5 hrs. Prepared resins were coupled with CD63 Abs (Cat. 15363, Santa Cruz, CA) at 4 °C overnight, followed by a wash step with blocking buffer. The resin was resuspended in blocking buffer and incubated with purified exosomes at 4 °C overnight. After washed with blocking buffer 3 times, the resins were analyzed by Zeiss AxioObserver. Z1 Fluorescence Microscope.
Statistical Methods
The data obtained was analyzed by student paired T-test. A value of p ≤ 0.05 was considered significant. The computer program GraphPad PRISM 5 was used for the analysis.
Additional Information
How to cite this article : Smith, V. L. et al. Exosomes function in antigen presentation during an in vivo Mycobacterium tuberculosis infection. Sci. Rep. 7, 43578; doi: 10.1038/srep43578 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Cooper, A. M. T cells in mycobacterial infection and disease. Current opinion in immunology 21, 378–384 (2009).
Srivastava, S. & Ernst, J. D. Cell-to-cell transfer of M. tuberculosis antigens optimizes CD4 T cell priming. Cell host & microbe 15, 741–752, doi: 10.1016/j.chom.2014.05.007 (2014).
Cheng, Y. & Schorey, J. S. Exosomes carrying mycobacterial antigens can protect mice against Mycobacterium tuberculosis infection. Eur J Immunol 43, 279–290. (2013).
Giri, P. K., Kruh, N. A., Dobos, K. M. & Schorey, J. S. Proteomic analysis identifies highly antigenic proteins in exosomes from M. tuberculosis-infected and culture filtrate protein-treated macrophages. Proteomics 10, 3190–3202 (2010).
Schorey, J. S. & Bhatnagar, S. Exosome function: from tumor immunology to pathogen biology. Traffic 9, 871–881 (2008).
Hurley, J. H. ESCRTs are everywhere. The EMBO journal 34, 2398–2407, doi: 10.15252/embj.201592484 (2015).
Raposo, G. & Stoorvogel, W. Extracellular vesicles: exosomes, microvesicles, and friends. The Journal of cell biology 200, 373–383 (2013).
Schorey, J. S., Cheng, Y., Singh, P. P. & Smith, V. L. Exosomes and other extracellular vesicles in host-pathogen interactions. EMBO reports 16, 24–43, doi: 10.15252/embr.201439363 (2015).
Srivastava, S., Grace, P. S. & Ernst, J. D. Antigen Export Reduces Antigen Presentation and Limits T Cell Control of M. tuberculosis. Cell host & microbe 19, 44–54, doi: 10.1016/j.chom.2015.12.003 (2016).
Ostrowski, M. et al. Rab27a and Rab27b control different steps of the exosome secretion pathway. Nature cell biology 12, 19–30; sup pp 11–13 (2011).
Pfeffer, S. R. Two Rabs for exosome release. Nature cell biology 12, 3–4, doi: 10.1038/ncb0110-3 (2010).
Stenmark, H. Rab GTPases as coordinators of vesicle traffic. Nature reviews. Molecular cell biology 10, 513–525, doi: 10.1038/nrm2728 (2009).
Johnson, J. L. et al. Rab27a and Rab27b regulate neutrophil azurophilic granule exocytosis and NADPH oxidase activity by independent mechanisms. Traffic 11, 533–547, doi: 10.1111/j.1600-0854.2009.01029.x (2010).
Catz, S. D. The role of Rab27a in the regulation of neutrophil function. Cellular microbiology 16, 1301–1310, doi: 10.1111/cmi.12328 (2014).
Singh, P. P., Smith, V. L., Karakousis, P. C. & Schorey, J. S. Exosomes isolated from mycobacteria-infected mice or cultured macrophages can recruit and activate immune cells in vitro and in vivo . Journal of immunology 189, 777–785 (2012).
Catz, S. D. Regulation of vesicular trafficking and leukocyte function by Rab27 GTPases and their effectors. Journal of leukocyte biology 94, 613–622, doi: 10.1189/jlb.1112600 (2013).
Choi, D. S., Kim, D. K., Kim, Y. K. & Gho, Y. S. Proteomics, transcriptomics and lipidomics of exosomes and ectosomes. Proteomics 13, 1554–1571, doi: 10.1002/pmic.201200329 (2013).
Kruh-Garcia, N. A. et al. Use of Multiple Reaction Monitoring Mass Spectrometry to Enhance Discovery of Tuberculosis Biomarkers in Exosomes. PloS one 9, e103811 doi: 10.1371/journal.pone.0103811 (2014).
Smith, V. L., Jackson, L. & Schorey, J. S. Ubiquitination as a Mechanism To Transport Soluble Mycobacterial and Eukaryotic Proteins to Exosomes. Journal of immunology, doi: 10.4049/jimmunol.1403186 (2015).
Dobos, K., Schorey, J. S. & Kruh-Garcia, N. Tuberculosis Biomarkers: Prospects from the bench to the clinic Vol. 1 (InTech - Open Access Publisher, 2011).
Giri, P. K. & Schorey, J. S. Exosomes derived from M. Bovis BCG infected macrophages activate antigen-specific CD4+ and CD8+ T cells in vitro and in vivo . PloS one 3, e2461, doi: 10.1371/journal.pone.0002461 (2008).
Bhatnagar, S., Shinagawa, K., Castellino, F. J. & Schorey, J. S. Exosomes released from macrophages infected with intracellular pathogens stimulate a proinflammatory response in vitro and in vivo . Blood 110, 3234–3244 (2007).
Divangahi, M., Behar, S. M. & Remold, H. Dying to live: how the death modality of the infected macrophage affects immunity to tuberculosis. Adv Exp Med Biol 783, 103–120 (2013).
Behar, S. M. et al. Apoptosis is an innate defense function of macrophages against Mycobacterium tuberculosis. Mucosal immunology 4, 279–287, doi: 10.1038/mi.2011.3 (2011).
Wilson, S. M. et al. A mutation in Rab27a causes the vesicle transport defects observed in ashen mice. Proceedings of the National Academy of Sciences of the United States of America 97, 7933–7938, doi: 10.1073/pnas.140212797 (2000).
Bobrie, A. et al. Rab27a supports exosome-dependent and -independent mechanisms that modify the tumor microenvironment and can promote tumor progression. Cancer research 72, 4920–4930, doi: 10.1158/0008-5472.CAN-12-0925 (2012).
Haddad, E. K., Wu, X., Hammer, J. A. 3rd & Henkart, P. A. Defective granule exocytosis in Rab27a-deficient lymphocytes from Ashen mice. The Journal of cell biology 152, 835–842 (2001).
Athman, J. J. et al. Bacterial Membrane Vesicles Mediate the Release of Mycobacterium tuberculosis Lipoglycans and Lipoproteins from Infected Macrophages. Journal of immunology 195, 1044–1053, doi: 10.4049/jimmunol.1402894 (2015).
Roach, S. K. & Schorey, J. S. Differential regulation of the mitogen-activated protein kinases by pathogenic and nonpathogenic mycobacteria. Infection and immunity 70, 3040–3052 (2002).
Cheng, Y. & Schorey, J. S. Targeting soluble proteins to exosomes using a ubiquitin tag. Biotechnology and bioengineering 113, 1315–1324, doi: 10.1002/bit.25884 (2016).
Acknowledgements
We thank Dr. Sergio Catz at the Scipps Research Institute for providing the Rab27a-deficient mice. We thank Dr. Karen Dobos (Colorado State) and Dr. Russ Karl (University of George) for the ΔHspX H37Rv strain. Funds provided to JSS through a grant (AI052439) from the National Institute of Allergy and Infectious Diseases.
Author information
Authors and Affiliations
Contributions
V.L.S. designed and performed the experiments shown in Figures 1–3 and 6 and co-wrote the manuscript. Y.C. designed and performed the experiments shown in Figures 4 and 5 and wrote the result and methods sections associated with these figures. B.R.B. worked with V.L.S. on the experiments shown in Figures 2 and 3. J.S.S. helped design the experiments and co-wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Smith, V., Cheng, Y., Bryant, B. et al. Exosomes function in antigen presentation during an in vivo Mycobacterium tuberculosis infection. Sci Rep 7, 43578 (2017). https://doi.org/10.1038/srep43578
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep43578
This article is cited by
-
Bovine Milk Derived Exosomes Affect Gut Microbiota of DSS-Induced Colitis Mice
Indian Journal of Microbiology (2024)
-
Immune cells-derived exosomes function as a double-edged sword: role in disease progression and their therapeutic applications
Biomarker Research (2022)
-
Bedaquiline reprograms central metabolism to reveal glycolytic vulnerability in Mycobacterium tuberculosis
Nature Communications (2020)
-
Host defense mechanisms against Mycobacterium tuberculosis
Cellular and Molecular Life Sciences (2020)
-
The Messenger Apps of the cell: Extracellular Vesicles as Regulatory Messengers of Microglial Function in the CNS
Journal of Neuroimmune Pharmacology (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.