Abstract
Hallucinations have been described in various clinical populations, but they are neither disorder nor disease specific. In schizophrenia patients, hallucinations are hallmark symptoms and auditory ones are described as the more frequent. In Parkinson’s disease, the descriptions of hallucination modalities are sparse, but the hallucinations do tend to have less negative consequences. Our study aims to explore the phenomenology of hallucinations in both hallucinating schizophrenia patients and Parkinson’s disease patients using the Psycho-Sensory hAllucinations Scale (PSAS). The main objective is to describe the phenomena of these clinical symptoms in those two specific populations. Each hallucinatory sensory modality significantly differed between Parkinson’s disease and schizophrenia patients. Auditory, olfactory/gustatory and cœnesthetic hallucinations were more frequent in schizophrenia than visual hallucinations. The guardian angel item, usually not explored in schizophrenia, was described by 46% of these patients. The combination of auditory and visual hallucinations was the most frequent for both Parkinson’s disease and schizophrenia. The repercussion index summing characteristics of each hallucination (frequency, duration, negative aspects, conviction, impact, control and sound intensity) was always higher for schizophrenia. A broader view including widespread characteristics and interdisciplinary works must be encouraged to better understand the complexity of the process involved in hallucinations.
Similar content being viewed by others
Introduction
Hallucinations have been defined by the Diagnostic and Statistical Manual of Mental Disorders fifth edition (DSM-5)1 as: “perception-like experiences that occur without an external stimulus. They are vivid and clear, with the full force and impact of normal perceptions, and not under voluntary control. They may occur in any sensory modality, but auditory hallucinations are the most common in schizophrenia and related disorders. Auditory hallucinations are usually experienced as voices, whether familiar or unfamiliar, that are perceived as distinct from the individual’s own thoughts. The hallucinations must occur in the context of a clear sensorium; those that occur while falling asleep (hypnagogic) or waking up (hypnopompic) are considered to be within the range of normal experience. Hallucinations may be a normal part of religious experience in certain cultural contexts” (Schizophrenia spectrum and other psychotic disorders section, p 87–88)1. However, this definition is somewhat restrictive for schizophrenia, as it is weak in terms of phenomenological aspects and specifically focused on auditory experiences. Despite the fact that hallucinations are present in the clinical criteria of various disorders (e.g. schizoaffective disorder, substance/medication-induced psychotic disorder, psychotic disorder due to other medical conditions, bipolar and related disorder, depressive disorder, anxiety disorder, etc), there is no other precise description taking into account their specificity related to the clinical context in DSM-5.
As underlined by Lowe2, “the variety in the manners in which hallucinations have been defined does not imply that any given definition is invalid, but it does confirm that hallucinations are complex phenomena, whose investigation almost certainly required multi-dimensional research designs and multiple initial criteria”.
Hallucinations must be considered as heterogeneous experiences, involving a wide variety of modalities and types including auditory, verbal, visual, olfactory, cenesthetic, gustatory and also multi-modal expression (hallucinations occurring simultaneously in more than one modality). If auditory hallucinations are the most frequently described and considered to be the most prevalent, especially in schizophrenia, other modalities or multi-modal expressions are probably underreported and more common than traditionally suggested3.
In a phenomenological perspective, auditory hallucinations can be better described using acoustic and linguistic properties, frequency, control, inner- vs outer-localization, content, personification, appraisals and change over time4. Perceptual qualities, temporal aspects, content, reality, sense of control, onset and triggers, reactions, beliefs and appraisals would help to describe visual hallucinations5.
In the last years, complementary approaches to the DSM have been raised. This is notably the case for the NIMH Research Domain Criteria (RDoC), which consider psychopathology in terms of maladaptive extremes along a continuum of normal functioning. Research Domain Criteria were judged optimal to promote a translational approach and encourage studying a dimension of interest in different groups, remaining “agnostic with regard to diagnosis”6. Recent works6,7 proposed that such a framework could be used to explore the complexity of auditory and visual hallucinations.
Hallucinations have been described in various clinical populations, but they are neither disorder nor disease specific. They are also frequent in non-clinical populations4, and an important interest has been developed for voice-hearing in the general population8.
In schizophrenia (SCZ) patients, hallucinations can be observed in any of the sensory modalities. In 59% of the cases they are auditory in nature, and in 27% of those cases visual hallucinations are also experienced at some point5. Other types of hallucinations are less prevalent.
In the course of Parkinson’s disease (PD), hallucinations occur in approximately 30 to 60% of the subjects9. They are frequently considered to be visual in nature, with prevalence of this modality ranging from 22 to 38%10, and are less frequently auditory (8% prevalence) according to Inzelberg et al.11. Olfactory hallucinations have been described in 10% of PD patients12. More specifically, in this population “presence hallucinations” include the feeling of another person being present mostly to the side or behind the subject13. Multimodal experiences have been reported in up to 30% of cases14. An important aspect is that hallucinations in PD seem to have less negative valence and less impact on patient quality of life compared to SCZ patients12.
The assessment of hallucinations relies on different tools to evaluate each modality and integrate various phenomenological characteristics3. We recently developed a multimodal hetero-evaluation scale (i.e. the Psycho-Sensory hAllucinations Scale: PSAS) that includes four domains (auditory, visual, olfactory/gustatory, and coenesthetic modalities) and one specific item ‘guardian angel’ defined as the “feeling of presence to the vivid sensation that somebody is present nearby, when no one is actually there, in the absence of sensory clues revealing a presence”, to describe the “presence hallucinations”, previously mentioned. We validated this scale in different populations of hallucinating patients suffering from SCZ and PD. A dimensional analysis confirmed a four-factor structure in PD patients including a first factor grouping olfactory/gustatory hallucinations and coenesthetic hallucinations, a second factor with auditory hallucinations, a third one defined by visual hallucinations and a forth one with the ‘guardian angel’ item. In patients with SCZ, a three-factor solution was confirmed, including a first factor gathering auditory, gustatory and olfactory hallucinations, a second one including mainly visual hallucinations and a third one grouping “guardian-angel” and coenesthetic hallucinations15.
This study aimed to explore the phenomenology of hallucinations in SCZ and PD patients using the PSAS. The objectives were i) to describe the phenomenology of these clinical symptoms in those two specific populations, and ii) to compare their specificity in those two groups.
Methods
Procedure
We performed a multicenter study involving five psychiatric departments and two neurological departments in France during one year. All consecutive patients were screened for study participation in order to reduce selection bias. Hallucinating patients with a diagnosis of SCZ or PD were included consecutively and were evaluated during one session by investigators.
Evaluation tool
Hallucinations were assessed using the PSAS. It includes four domains related to the five sensory modalities (auditory, visual, olfactory/gustatory, and coenesthetic) but also another domain: ‘guardian angel’. This scale was found to have good internal consistency and good inter-rater reliability (i.e. for internal consistency Kuder–Richardson alpha coefficient 0.49 to 0.77 and for inter-rater reliability, agreement % = 0.78 to 1.0)15.
A repercussion index was calculated for each hallucination by adding the score obtained at the quantitative section of the PSAS: frequency (Fq), duration (Du), unpleasant or negative aspects (NA), conviction (C), impact (I), control (Ctrl) and sound intensity (SI) (the latter only for auditory hallucinations). Range of score was [0–27] for auditory hallucinations and [0–23] for visual, olfactory/gustatory and coenesthetic hallucinations.
Collected data
The evaluations were performed by psychiatrists specialized in the evaluation of SCZ patients (ICB, GB, DM, AMT, RS, CL, PML) and neurologists specialized in the evaluation of PD patients (GF, AM, BD, FD). A training session using a questionnaire-based diagnostic guidelines was conducted to reduce variability on rating.
Socio-demographic and therapeutic clinical data were collected for all of the participants during the interview for the administration of the PSAS.
The use of dopaminergic agonists, amantadine, anticholinergics, psychoactive drugs (antidepressants, antipsychotics including clozapine, anxiolytics and/or hypnotics) was recorded. The levodopa equivalent daily dose and chlorpromazine equivalent daily dose were calculated using published and validated equivalence schemes16,17.
Inclusion and exclusion criteria
Inclusion criteria were:
- Participants older than 18 years of age.
- For the SCZ group: schizophrenia, according to DSM-IV-TR diagnosis criteria including a positive score criterion for hallucination (A2: characteristic symptoms) and a score up to three for the ‘hallucinatory behavior’ item of the Positive and Negative Syndrome Scale (PANSS P3)18.
- For the PD group: a diagnosis of Parkinson’s disease based on the UK Brain Bank criteria19 and a score above one for the ‘thought disorder’ item of the modified version of Unified Parkinson’s Disease Rating Scale part 1 (UPDRS1 I2)20 (‘delusions’ and ‘florid psychosis’ terms have been deleted).
Exclusion criteria were:
- An inability to understand the instructions because of language or an underlying severe pathology.
- Mini Mental State Examination (MMSE) < 24.
- A primary diagnosis of any psychiatric disorder Axis I (including Substance-related Disorder, Mood Disorder and Anxiety Disorder), apart from schizophrenia in the SCZ group.
Ethics
The study was conducted according to good clinical practice. This research has been evaluated and qualified by Ethics Committee (Committees of Protection of Persons Sud Est 6) as non-interventional clinical trial in accordance with French law that requires only a free, informed form. Written consent is not required in this case. CHU Clermont-Ferrand –FRANCE approved the experimental protocol.
Data Analysis
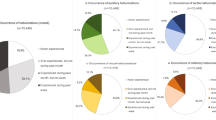
Statistical analysis was performed using Stata software, version 13 (StataCorp, College Station, TX, U.S.). The tests were two-sided, with a type I error set at α = 0.05. Continuous data were presented as the mean ± standard deviation or the median [interquartile range] according to statistical distribution (assumption of normality checked using normal probability plots and Shapiro-Wilk’s test). Comparisons between independent groups were analyzed using the Student t-test or Mann-Whitney test when conditions of the t-test were not met (normality and homoscedasticity verified by the Fisher-Snedecor test) for quantitative variables. Comparisons concerning categorical data were performed using the Chi-Squared test or Fisher’s exact test. For each pathology (SCZ and PD), a Venn diagram21 was proposed to illustrate the relationships between the dimensions of PSAS. More precisely, a five-set Venn diagram using congruent ellipses in a 5-fold rotationally symmetrical arrangement devised by Grünbaum was performed. A Venn diagram is constructed with a collection of simple closed curves drawn in a plane. The principle of these diagrams is that classes be represented by regions in such relation to one another that all the possible logical relations of these classes can be indicated in the same diagram. Venn diagrams comprise overlapping circles. The interior of the circle symbolically represents the elements of the set, while the exterior represents elements that are not members of the set. Venn diagrams do not generally contain information on the relative or absolute sizes of sets; i.e. they are schematic diagrams.
Results
Characteristics of the population
Socio-demographic and clinical characteristics are summarized in Table 1. The study included 100 PD and 100 SCZ patients (86 considered as early onset patients and 14 considered as late-onset according to the criteria defined by Howard et al.22). There was no difference between the two groups in regards to anxiolytics, antidepressant drug or sedative-hypnotic drug ratios.
Prevalence and associations between different hallucinatory experiences
Hallucination occurrences in every modality are summarized in Table 2. Each hallucination sensory modality significantly differed between PD and SCZ patients. The highest prevalence rate for “guardian angel” syndrome was observed in PD even though this syndrome was reported in half of the SCZ sample. Auditory, olfactory/gustatory and coenesthetic hallucinations were more frequent in SCZ compared to visual hallucinations which were more prevalent in PD. The combination of auditory and visual hallucinations was the most frequent for both PD and SCZ, but the distribution of the total number of sensory modalities significantly differed between the two groups.
Combinations of hallucination sets (Fig. 1) seemed specific for each disease. For SCZ the distribution of subsets of hallucinations was nearly well balanced. On the contrary, visual hallucinations predominated over mono- or multi-modalities in PD. For the two disorders, the guardian angel syndrome was always found combined with at least one other hallucination sensory modality. There’s no significant correlation between L-dopa equivalent daily dose or chlorpromazine equivalent daily dose and number of hallucinations.
Venn diagrams showing intersections between hallucination sensory modalities in Parkinson’s disease (PD) and schizophrenia (SCZ) patients (n = 200).
Understanding results guide For PD patients, 32 = Visual ∪Auditory ∩ Guardian angel ∪ Coenesthetic ∪ Olfactory For SCZ patients, 7 = Visual ∩ Auditory ∪ Guardian angel ∩ Coenesthetic ∩ Olfactory.
Repercussion index
Regarding the quantitative evaluation of hallucinations, only the repercussion index for olfactory/gustatory hallucination did not significantly differ between the groups (Table 3). For the other sensory modalities involved in hallucinations, the repercussion index varied significantly across groups, with the highest mean score in the SCZ group. Auditory hallucinations exhibited the highest repercussion index among sensory modalities, for both PD and SCZ groups. L-dopa equivalent daily dose was not significantly correlated with repercussion index of each sensory modalities. Chlorpromazine equivalent daily dose was only significantly and weakly correlated with auditory hallucinations repercussion index (r = 0.26, p = 0.01).
Quantitative evaluations of hallucinations are detailed in Fig. 2. The frequency of hallucinations was significantly higher for SCZ patients in the auditory and coenesthetic modalities. The duration of hallucinations was also significantly higher for SCZ in the auditory, visual and coenesthetic modalities. Negative contents were significantly more frequently reported by SCZ subjects in auditory and visual hallucinations. Conviction was found higher for SCZ only for visual hallucinations, while the feeling of being in control was higher in the same subjects only for coenesthetic experiences. Finally, the sound intensity of auditory hallucinations was higher for SCZ compared with PD patients.
Spider diagrams of repercussion factors of hallucinations in Parkinson’s disease (PD) and schizophrenia (SCZ) patients.
Frequency (Fq), duration (Du), unpleasant or negative aspects (NA), conviction (C), impact (I), control (Ctrl) and sound intensity (SI). For each spider diagram, data are expressed as means and standard deviations (SD) were placed in a box. *p < 0.05 (comparison between pathologic groups).
Discussion
To our knowledge, the present study is the first to qualitatively and quantitatively describe the phenomenological properties of hallucinatory experiences, across sensory modalities and categorical diagnoses (i.e. SCZ and PD). The main results can be summarized as follows:
-
i
In SCZ, auditory hallucinations were observed in 83% of the cases, but visual and coenesthetic hallucinations were also reported by more than 50% of patients. Different sensory modalities were combined in 81% of the patients, and within 55% of the cases two or three modalities were combined. In PD, visual hallucinations appeared predominant (88%), but 42% of the patients also described auditory experiences. In our study population, 80% of the PD patients described more than one sensory modality involved, with 44% associating two modalities (most frequently visual and auditory).
-
ii
The guardian angel item, which is usually not explored in SCZ, was described by 46% of SCZ patients.
-
iii
The repercussion index, created to describe the impact of hallucinations in the patients (i.e. this index gathered the scores for frequency, duration, intensity, negative content, beliefs and perceived control), demonstrated that SCZ patients were more affected by hallucinations compared with PD patients.
-
iv
Except for the olfactory and gustatory sensory modalities (which differed only in terms of impact), SCZ and PD significantly differed in terms of hallucination properties: i.e. frequency, duration, capacity of control, negative valence and impact.
Rates of hallucinatory experiences in the different sensory modalities appeared consistent with the literature both for SCZ23 and non-demented treated PD patients24. In our study, a major finding was the high rate of simultaneous multimodal hallucinations, which are poorly described in SCZ patients despite the fact that they are considered to be rather frequent in the non-clinical population25. A recent internet survey was conducted in the general population (n = 153) to phenomenologically explore auditory hallucinations26. The authors showed that 28% of the participants (n = 43) reported an association of auditory hallucinations with “distinct hallucinations in other senses”. Thirty-seven of the participants (24%) were diagnosed as SCZ or schizoaffective patients, and none were described as PD patients. No specific information was given about the multimodal expression of hallucinations in this sub-sample. The lower percentage of association of various modalities of hallucinations compared to our study can be related to the different study population and to differences in the method of hallucination evaluation.
In the literature22,27, late-onset schizophrenic patients are considered to complain more of visual, tactile and olfactory hallucinations, third-person running commentary, and accusatory or abusive auditory hallucinations, than early-onset patients. In our sample, if late-onset patients present more hallucinations with a smaller level of repercussion, compared to early-onset patients, these differences don’t reach statistical significance (respectively p = 0.06 and p = 0.08). This lack of difference can be related to the small size of our sample, and will have to be further studied.
For PD, our results are consistent with the findings of Goetz et al.9. These authors demonstrated that non-visual hallucinations emerged over time: after a ten-year observation period of 60 PD patients, 60% had multimodal hallucination symptomatology. In this case, the question arises whether dementia can have an impact but previous multivariate analysis28 and some univariate works29 already show that cognition impairment did not correlate as an independent variable significantly with Parkinson disease duration In our sample, the mean duration of PD was 10.76 years (SD 5.8), which is comparable with the study by Goetz et al. (9.0 ± 6.2 years).
Our two samples appear to be quite representative of SCZ and PD and haven’t any other clinical or treatment specificity compared to what was described in the literature that could be correlated to this observation. This result exemplifies the potential of the PSAS to systematically explore hallucinations in every modality.
The prevalence of “presence hallucinations”, described by the “guardian angel” item of the PSAS, in PD is rather well-known, ranging from 34 to 40% in samples of consecutively examined patients10. In the present study, the percentage was higher (70%). This difference can be related to the specific inclusion of hallucinating PD patients in our study group, but also to the systematic identification of such experiences using the PSAS.
It’s noteworthy that, despite this high percentage of presence hallucinations, the mean L-dopa equivalent daily dose (LEDD) was very close to the one reported by Fenelon et al.10. (876 mg/d, SD 573 vs 892 mg/d, SD 389). The mean LEDD in the sub-group of patients (n = 70) with presence hallucinations was 864 (SD 564) mg/d.
Jaspers described this phenomenon in 1913, in patients suffering from dementia praecox (i.e. schizophrenia), under the name of “leibhatige Bewussheit”, which has been translated as “sense of presence”, but also ‘idea of presence’: “There are patients who have a certain feeling (in the mental sense) or awareness that someone is close by, behind them or above them, someone that they can in no way perceive with the external senses, yet whose actual/concrete presence is directly/clearly experienced” (translated in Koehler and Sauer, 198430). The discussion about the fact that it can be considered as a hallucination or a delusion can be related to the difficulty to operationalize in clinical practice the notions of “feeling in a mental way or awareness”30,31. In the DSM-5 criteria for schizophrenia, the “sense of presence of an unseen person” is considered to be one of the “unusual perceptive experiences”, part of the continuous disturbances described in the Criterion C (p 101). Presence hallucinations can be considered as residual symptoms. In our SCZ sample, the percentage of patients describing this experience is rather high (46%), but to our knowledge, there are no other studies to compare this level with. It probably deserves a more specific phenomenological evaluation in future studies.
By creating a composite index combining frequency, duration, intensity, negative content, beliefs and perceived control, we were able to describe in a reproducible manner the impact of hallucinations on patients. In our study, visual, auditory and coenesthetic hallucinations exhibited greater impacts on SCZ patients compared with PD patients. This can notably be correlated to the negative impact of hallucinations on SCZ patients in terms of quality of life and ability to function32. Nevertheless, the impact of positive symptoms on quality of life appears relatively weak in individuals in the early course of the illness compared to chronic patients33. The mean duration of illness in the sub-group of SCZ patients is 8.1 years (SD 6.8), and they can’t be considered to be in an early stage of schizophrenia. Specific interventions integrating coping training and cognitive-behavioral therapy and focusing on hallucinations induced a significant improvement in quality of life and ability to function34. On the other hand in PD patients, hallucinations do not seem to be a major predictor of impaired quality of life regardless cognitive profile35,36. Depression has the most negative influence on ability to function in those patients.
Perspectives
Conceptually, the present study, based on the PSAS, confirmed the possibility of exploring hallucinations transdiagnostically as recently proposed through the RDoC initiative. Indeed, to move beyond strict categorical diagnoses, it appears crucial to determine if hallucinations are the same phenomena in different clinical populations37. This is called the “equifinality” purpose in the RDoC framework6. The fact that SCZ and PD patients may share common hallucinatory experiences, but with different phenomenological properties (i.e. potentially linked with different RDoC domains) clearly raises new questions regarding their possible mechanisms. The present study is not able to answer such a complex question but the availability of transdiagnostic tools and data are a necessary starting point. The idea behind the RDoC domains is the possibility to link specific phenomenological features with cognitive or neurobiological mechanisms. Few studies have explored the neural basis of the phenomenological properties of hallucinations, as for example: the sense of reality38, the sensory modalities involved39, or the emotional content40. The next step is to combine these detailed studies of hallucination phenomenology with transdiagnostic approaches. We believe that tools such as the PSAS could help design such experiments in the future.
Strengths
To our knowledge, this work is the first to explore the hallucination phenomenon in a multimodal way, in two different clinical samples and using a specifically designed tool. These three points are the principal strengths of our study. This study analyzed patients with SCZ and PD, two illnesses in which hallucinations can be frequent, and are related to a dopaminergic dysfunction. The rather large size of our two sample populations, compared to previously published studies, can also be considered a strength as it allowed comparison of the different modalities of hallucinations between the two groups. The description of hallucination impact on patients, using the repercussion index, could be of interest to define individualized treatment goals for patients.
Limitations
One of the limitations of this study is the cross-sectional design, which doesn’t allow for study of the evolution of hallucinations over time or their relations, which is of interest as underlined by different authors41. The use of a structured tool can be considered another limitation to this study, as the previously made assumptions about the nature of hallucinations that were made to build this evaluation scale may limit the phenomenological description26.
Conclusion
The phenomenological specificity of hallucinations regarding pathologies can contribute significantly towards their comprehension, and ongoing publications26,42,43 show promising results. A broader view including widespread characteristics, such as age, gender, cognitive deficits or treatments and interdisciplinary works, must be encouraged to better understand the complexity of the process involved in hallucinations.
Additional Information
How to cite this article: Llorca, P. M. et al. Hallucinations in schizophrenia and Parkinson’s disease: an analysis of sensory modalities involved and the repercussion on patients. Sci. Rep. 6, 38152; doi: 10.1038/srep38152 (2016).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
American Psychiatric Association. Fifth Edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-5). (New School Library, 2013).
Lowe, G. R. The phenomenology of hallucinations as an aid to differential diagnosis. Br J Psychiatry 123, 621–633 (1973).
Laroi, F., Van der Linden, M., DeFruyt, F., van Os, J. & Aleman, A. Associations between delusion proneness and personality structure in non-clinical participants: comparison between young and elderly samples. Psychopathology 39, 218–226, doi: 10.1159/000093922 (2006).
Laroi, F. et al. The characteristic features of auditory verbal hallucinations in clinical and nonclinical groups: state-of-the-art overview and future directions. Schizophr Bull 38, 724–733, doi: 10.1093/schbul/sbs061 (2012).
Waters, F. et al. Visual hallucinations in the psychosis spectrum and comparative information from neurodegenerative disorders and eye disease. Schizophr Bull 40 Suppl 4, S233–245, doi: 10.1093/schbul/sbu036 (2014).
Ford, J. M. et al. Studying hallucinations within the NIMH RDoC framework. Schizophr Bull 40 Suppl 4, S295–304, doi: 10.1093/schbul/sbu011 (2014).
Badcock, J. C. & Hugdahl, K. A synthesis of evidence on inhibitory control and auditory hallucinations based on the Research Domain Criteria (RDoC) framework. Front Hum Neurosci 8, 180, doi: 10.3389/fnhum.2014.00180 (2014).
Beavan, V., Read, J. & Cartwright, C. The prevalence of voice-hearers in the general population: a literature review. J Ment Health 20, 281–292, doi: 10.3109/09638237.2011.562262 (2011).
Goetz, C. G., Stebbins, G. T. & Ouyang, B. Visual plus nonvisual hallucinations in Parkinson’s disease: development and evolution over 10 years. Mov Disord 26, 2196–2200, doi: 10.1002/mds.23835 (2011).
Fenelon, G. & Alves, G. Epidemiology of psychosis in Parkinson’s disease. J Neurol Sci 289, 12–17, doi: 10.1016/j.jns.2009.08.014 (2010).
Inzelberg, R., Kipervasser, S. & Korczyn, A. D. Auditory hallucinations in Parkinson’s disease. J Neurol Neurosurg Psychiatry 64, 533–535 (1998).
Bannier, S. et al. Prevalence and phenomenology of olfactory hallucinations in Parkinson’s disease. J Neurol Neurosurg Psychiatry 83, 1019–1021, doi: 10.1136/jnnp-2012–302414 (2012).
Fenelon, G., Mahieux, F., Huon, R. & Ziegler, M. Hallucinations in Parkinson’s disease: prevalence, phenomenology and risk factors. Brain 123 (Pt 4), 733–745 (2000).
Katzen, H. et al. Multi-modal hallucinations and cognitive function in Parkinson’s disease. Dement Geriatr Cogn Disord 30, 51–56, doi: 10.1159/000314875 (2010).
de Chazeron, I. et al. Validation of a Psycho-Sensory hAllucinations Scale (PSAS) in schizophrenia and Parkinson’s disease. Schizophr Res 161, 269–276, doi: 10.1016/j.schres.2014.11.010 (2015).
Leucht, S. et al. Dose Equivalents for Second-Generation Antipsychotics: The Minimum Effective Dose Method. Schizophr Bull, doi: 10.1093/schbul/sbu001 (2014).
Thobois, S. Proposed dose equivalence for rapid switch between dopamine receptor agonists in Parkinson’s disease: a review of the literature. Clin Ther 28, 1–12 (2006).
Kay, S. R., Fiszbein, A. & Opler, L. A. The positive and negative syndrome scale (PANSS) for schizophrenia. Schizophr Bull 13, 261–276 (1987).
Hughes, A. J., Daniel, S. E., Kilford, L. & Lees, A. J. Accuracy of clinical diagnosis of idiopathic Parkinson’s disease: a clinico-pathological study of 100 cases. J Neurol Neurosurg Psychiatry 55, 181–184 (1992).
The Unified Parkinson’s Disease Rating Scale (UPDRS): status and recommendations. Mov Disord18, 738–750, doi: 10.1002/mds.10473 (2003).
Henderson, D. W. Venn diagrams for more than four classes. American Mathematical Monthly 70, 424–426 (1963).
Howard, R., Rabins, P. V., Seeman, M. V. & Jeste, D. V. Late-onset schizophrenia and very-late-onset schizophrenia-like psychosis: an international consensus. The International Late-Onset Schizophrenia Group. Am J Psychiatry 157, 172–178, doi: 10.1176/appi.ajp.157.2.172 (2000).
Aleman, A. & Laroi, F. Hallucinations: The Science of Idiosyncratic Perception. (American Psychological Association, 2008).
Burghaus, L., Eggers, C., Timmermann, L., Fink, G. R. & Diederich, N. J. Hallucinations in neurodegenerative diseases. CNS Neurosci Ther 18, 149–159, doi: 10.1111/j.1755–5949.2011.00247.x (2012).
Laroi, F., Marczewski, P. & Van der Linden, M. Further evidence of the multi-dimensionality of hallucinatory predisposition: factor structure of a modified version of the Launay-Slade Hallucinations Scale in a normal sample. Eur Psychiatry 19, 15–20, doi: 10.1016/s0924-9338(03)00028-2 (2004).
Woods, A., Jones, N., Alderson-Day, B., Callard, F. & Fernyhough, C. Experiences of hearing voices: analysis of a novel phenomenological survey. Lancet Psychiatry 2, 323–331, doi: 10.1016/s2215-0366(15)00006-1 (2015).
Harish, M. G., Suresh, K. P., Rajan, I., Reddy, Y. C. & Khanna, S. Phenomenological study of late-onset schizophrenia. Indian J Psychiatry 38, 231–235 (1996).
Zhu, K., van Hilten, J. J. & Marinus, J. Predictors of dementia in Parkinson’s disease; findings from a 5-year prospective study using the SCOPA-COG. Parkinsonism Relat Disord 20, 980–985, doi: 10.1016/j.parkreldis.2014.06.006 (2014).
Braak, H., Rub, U., Jansen Steur, E. N., Del Tredici, K. & de Vos, R. A. Cognitive status correlates with neuropathologic stage in Parkinson disease. Neurology 64, 1404–1410, doi: 10.1212/01.wnl.0000158422.41380.82 (2005).
Koehler, K. & Sauer, H. Jaspers’ sense of presence in the light of Huber’s basic symptoms and DSM-III. Compr Psychiatry 25, 183–191 (1984).
Fenelon, G., Soulas, T., Cleret de Langavant, L., Trinkler, I. & Bachoud-Levi, A. C. Feeling of presence in Parkinson’s disease. J Neurol Neurosurg Psychiatry 82, 1219–1224, doi: 10.1136/jnnp.2010.234799 (2011).
Rocca, P., Castagna, F., Mongini, T., Montemagni, C. & Bogetto, F. Relative contributions of psychotic symptoms and insight to quality of life in stable schizophrenia. Psychiatry Res 177, 71–76, doi: 10.1016/j.psychres.2009.01.030 (2010).
Eack, S. M. & Newhill, C. E. Psychiatric symptoms and quality of life in schizophrenia: a meta-analysis. Schizophr Bull 33, 1225–1237, doi: 10.1093/schbul/sbl071 (2007).
Wiersma, D., Jenner, J. A., Nienhuis, F. J. & van de Willige, G. Hallucination focused integrative treatment improves quality of life in schizophrenia patients. Acta Psychiatr Scand 109, 194–201 (2004).
Behari, M., Srivastava, A. K. & Pandey, R. M. Quality of life in patients with Parkinson’s disease. Parkinsonism Relat Disord 11, 221–226, doi: 10.1016/j.parkreldis.2004.12.005 (2005).
McKinlay, A. et al. A profile of neuropsychiatric problems and their relationship to quality of life for Parkinson’s disease patients without dementia. Parkinsonism Relat Disord 14, 37–42, doi: 10.1016/j.parkreldis.2007.05.009 (2008).
Goghari, V. M., Harrow, M., Grossman, L. S. & Rosen, C. A 20-year multi-follow-up of hallucinations in schizophrenia, other psychotic, and mood disorders. Psychol Med 43, 1151–1160, doi: 10.1017/s0033291712002206 (2013).
Jardri, R. & Deneve, S. Circular inferences in schizophrenia. Brain 136, 3227–3241, doi: 10.1093/brain/awt257 (2013).
Jardri, R., Thomas, P., Delmaire, C., Delion, P. & Pins, D. The neurodynamic organization of modality-dependent hallucinations. Cereb Cortex 23, 1108–1117, doi: 10.1093/cercor/bhs082 (2013).
Escarti, M. J. et al. Increased amygdala and parahippocampal gyrus activation in schizophrenic patients with auditory hallucinations: an fMRI study using independent component analysis. Schizophr Res 117, 31–41, doi: 10.1016/j.schres.2009.12.028 (2010).
McCarthy-Jones, S. et al. A new phenomenological survey of auditory hallucinations: evidence for subtypes and implications for theory and practice. Schizophr Bull 40, 231–235, doi: 10.1093/schbul/sbs156 (2014).
Gras, A., Amad, A., Thomas, P. & Jardri, R. [Hallucinations and borderline personality disorder: a review]. Encephale 40, 431–438, doi: 10.1016/j.encep.2014.07.002 (2014).
McCarthy-Jones, S. & Longden, E. Auditory verbal hallucinations in schizophrenia and post-traumatic stress disorder: common phenomenology, common cause, common interventions? Front Psychol 6, 1071, doi: 10.3389/fpsyg.2015.01071 (2015).
Hoehn, M. M. & Yahr, M. D. Parkinsonism: onset, progression and mortality. Neurology 17, 427–442 (1967).
Acknowledgements
The study was supported by a Hospital Program for Clinical Research (Local funding), CHU Clermont-Ferrand –FRANCE.
Author information
Authors and Affiliations
Contributions
Llorca P.-M., Jardri R. and de Chazeron I. wrote the main manuscript text. Pereira B. wrote the statistical part of the manuscript and prepared Figure 1 and 2. Chereau-Boudet I., Brousse G., Misdrahi D., Fénelon G., Tronche A.-M., Schwan R., Lançon C., Marques A., Ulla M., Derost P., Debilly B. and Durif F reviewed the manuscript.
Ethics declarations
Competing interests
Author Llorca P.-M. reports grants from Lilly, personal fees from Janssen, nonfinancial support from Roche, and grants from Otsuka, all outside the submitted work. Author Renaud Jardri reports personal fees for lectures: Lundbeck, Astra-Zeneca, Janssen-Cilag, Otsuka pharmaceutical, Shire and GlaxoSmithKline, all outside of the submitted work Author Brousse G. reports personal fees from Lundbeck, Janssen-Cilag and Otsuka pharmaceutical, all outside of the submitted work. Author Fénelon G. reports personal fees from Lundbeck and Teva Pharma, all outside the submitted work. Author Schwan R. reports grants from Boucharad and personal fees from Lundbeck, Janssen-Cilag, Otsuka pharmaceutical and Reckitt-Bekinson, all outside of the submitted work. Author Durif F. reports grants from CHU Clermont Ferrand, during the conduct of the study; grants from Novartis SA, Lundbeck, and Merz; and personal fees from Novartis SA, Lundbeck, Allergan, Aguettant, Teva Pharma, and Servier, all outside the submitted work. All other authors declare that they have no conflicts of interest.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Llorca, P., Pereira, B., Jardri, R. et al. Hallucinations in schizophrenia and Parkinson’s disease: an analysis of sensory modalities involved and the repercussion on patients. Sci Rep 6, 38152 (2016). https://doi.org/10.1038/srep38152
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep38152
This article is cited by
-
Non-visual hallucinations in Parkinson’s disease: a systematic review
Journal of Neurology (2023)
-
The diagnostic performance of functional dopaminergic scintigraphic imaging in the diagnosis of dementia with Lewy bodies: an updated systematic review
European Journal of Nuclear Medicine and Molecular Imaging (2023)
-
Neuroscience robotics for controlled induction and real-time assessment of hallucinations
Nature Protocols (2022)
-
Hearing voices as a feature of typical and psychopathological experience
Nature Reviews Psychology (2022)
-
Ursachen visueller Halluzinationen bei der Parkinson-Krankheit
Der Nervenarzt (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.