Abstract
Faces are an important cue to multiple physiological and psychological traits. Human preferences for exaggerated sex typicality (masculinity or femininity) in faces depend on multiple factors and show high inter-subject variability. To gain a deeper understanding of the mechanisms underlying facial femininity preferences in men, we tested the interactive effect of family structure (birth order, sibling sex-ratio and number of siblings) and parenthood status on these preferences. Based on a group of 1304 heterosexual men, we have found that preference for feminine faces was not only influenced by sibling age and sex, but also that fatherhood modulated this preference. Men with sisters had a weaker preference for femininity than men with brothers, highlighting a possible effect of a negative imprinting-like mechanism. What is more, fatherhood increased strongly the preference for facial femininity. Finally, for fathers with younger sisters only, the more the age difference increased between them, the more femininity preference increased. Overall our findings bring new insight into how early-acquired experience at the individual level may determine face preference in adulthood, and what is more, how these preferences are flexible and potentially dependent on parenthood status in adult men.
Similar content being viewed by others
Introduction
People undeniably pay attention to faces. Based on signals carried by a face, we almost instantaneously make numerous inferences, notably dealing with personality, dominance, attractiveness, emotional state, or aggressiveness1. Up-to-date studies on human faces cover two distinct topics: i) how the facial features of a certain individual transmit the information to an observer (e.g. masculinity/femininity, symmetry, health, see2) and ii) how an individual’s own traits (such as their own ethnicity, sex or age) affect the perception of faces of other people (see3).
Preferences for exaggerated sex typicality in faces (masculinity/femininity) depend on multiple factors and it has been proposed to reflect a long evolutionary history of sexual selection (but see4). Most studies in this field have however focused on women’s preferences. Men’s preferences for facial femininity were found to correlate with their self-reported mating value5,6, sex hormone levels7,8, mating context (e.g. short-term or long-term relationships5), health of the nation9, disease-related factors (e.g. pathogen disgust or cortisol level10) and partnership status11. Despite the fact that parenthood status is known to be associated with some of the above aspects (especially testosterone12 and sexual behaviours13,14), there are virtually no studies to date investigating the interaction between parenthood and other factors influencing men’s facial preferences.
Development of sexual preferences is a complex process which is affected by multiple causes ranging from genetics15 and innate factors16 to personal experience17. It has been suggested that during early development, there is a sensitive phase in which one acquires templates used in later life when searching for a partner18,19. Sensitive phases are limited time periods occurring long before the maturation of the sexual behaviour system, during which the brain is especially sensitive to certain kind of stimuli20. A template – or neural representation– is formed based on the appearance of individuals in the early environment (especially with kin members, but also with others in our entourage, e.g. classmates21), and it is not until sexual maturity that this memory becomes associated with sexual behaviour22. Such sexual imprinting mechanisms can be either positive or negative depending on whether an individual develops attraction or aversion towards their imprinted template23. Selection of mates that are moderately different from self or the imprinted template usually enables the individual to achieve an optimal balance between costs of outbreeding and inbreeding – defined as optimal outbreeding24.
Positive sexual imprinting has found support in a number of studies on non-human animals (e.g. refs 25,26). In humans, support for positive sexual imprinting has been found based on facial similarity between their other-sex parent and partner27,28,29 and also on preferences for parental characteristics in putative partners: similar hair and eye-colour6 and age characteristics30,31. However, interpretations of observed results as supporting positive sexual imprinting (based on appearance of parents) have some problems, particularly in the majority of cases it is impossible to rule out the influence of heritable facial preferences (for a review see23).
The opposite mechanism is called negative sexual imprinting. Due to the detrimental effects of inbreeding depression32, individuals should avoid mates who closely resemble kin based on the sensory template (especially visual and olfactory templates) acquired during development33. It was suggested that cohabitation during early development serves as a strong contextual cue to biological relatedness and is a basis for future sexual aversion towards individuals resembling siblings, the Westermarck effect34. There have been mixed results from studies of negative sexual imprinting in humans35,36,37. There is also evidence for plasticity in human inbreeding avoidance strategies including birth order effects. For example, earlier born children experience stronger sexual aversion to siblings than later born ones due to so called “maternal–perinatal association”. Older siblings have a chance to observe their own mother nurturing another new-born – this equips them with an extra tool to detect kinship (adding to co-residence experience38 (but see also39), which in turn increases self-reported rejection of consensual third-party sibling incest40 and leads to increased aversion towards faces similar to their siblings.
The current study examined whether among adult men, both family structure (sibling age, number and sex) and parenthood status modulate their facial femininity preferences. Considering family structure in this study enabled testing the hypothesis of sexual imprinting-like mechanism whereas integrating the parenthood status of the participants allowed considering how the presence of another kin in adult life (own progeny) influences facial preferences.
To investigate our hypotheses, we used a large-scale sample of heterosexual men collected on-line. Based on facial femininity preferences expressed by participants, we tested two predictions. Firstly, we predicted that family structure (i.e. birth order and sibling sex ratio) influenced the preference for femininity. If so, an imprinting-like mechanism with a template based on siblings’ face could have been in place. Specifically, we wanted to test whether a negative sexual imprinting-like mechanism was activated – men who were more exposed to feminine faces (their other-sex siblings in childhood) would have a lower preference for facial femininity than men who were exposed to mainly masculine faces (same-sex siblings). Conversely, if positive sexual imprinting based on siblings was in place, we would observe the reverse effect: the presence of sisters during childhood would be related to increased preference for feminised faces. Secondly, we wanted to explore whether fatherhood status – exposure to another close kin, i.e. own child– would influence the preferences for exaggerated sex typicality in faces. Indeed, previous research suggested that i) transition to parenthood is associated with a decrease in baseline testosterone levels41,42 but see also43 and that, ii) men report stronger attraction to femininity in women’s faces if their testosterone levels are high8. We predicted that regardless of the presence and direction of imprinting-like mechanisms in place, fatherhood would lower the preference for femininity as a by-product of lower testosterone levels.
Results and Discussion
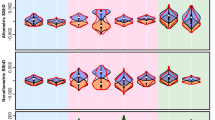
Overall, men in our study considered feminised women’s faces as more attractive than masculinised ones (on a scale from 0 – only masculinised faces chosen, to 1 – only feminised faces chosen as more attractive, 0.75 [0.67–0.83], Table S9 in ESM1), replicating the findings of previous studies44,45. We found direct and interactive effects of family structure (birth order, sibling sex-ratio and number of siblings, Tables S10–S12) and parenthood status on preference for facial femininity. The most important findings are displayed in Fig. 1.
The effects of family structure and parenthood status on preferences for facial femininity in men.
Mean and error bars represent 95% confidence intervals, and asterisks indicate significant differences (*p < 0.05, **p < 0.005). Shaded plots show the effect of fatherhood on femininity preference. (A) Effect of sibling sex ratio on the preference for facial femininity, showing that men with other-sex siblings had weaker preference for femininity than men with same-sex siblings. (B) Fatherhood induced a drop in preference for face femininity in men without sibling (Odds-ratio: 1.22 [1.08–1.38]) but a significant increase in preference among men with siblings (particularly when men have two siblings (Odds-ratio: 0.83 [0.71–0.97]). (C) Among men with female siblings, fatherhood influenced strongly the preference for femininity, especially in men with younger sisters. (D) For men with only younger sister(s), femininity preference as a function of the age difference between participants and their sister, and parenthood status (black: childless’ men; grey: men with child). Dashed lines were the curves predicted by logistic regression.
Sexual imprinting-like mechanism
Our results revealed an effect of sibling sex ratio on the preference for femininity (Fig. 1A). Indeed, men with sisters had weaker preference for femininity (0.74 [0.66–0.81]) than men with brothers (0.78 [0.70–0.84]; Chi2 = 8.05, df = 1, P = 0.005; OR = 0.81 [0.71–0.94], Table S13). For participants having younger siblings, men with brothers showed a higher preference for femininity than men with sisters (Chi2 = 4.72, df = 1, P = 0.029; OR = 1.16 [1.02–1.33]), but there was only a trend in same direction for men with older siblings (Chi2 = 3.8, df = 1, P = 0.051; OR = 1.16 [1.00–1.35]). Regardless of the fact that all men throughout their upbringing interact with a number of female individuals, men having sisters are more frequently exposed to feminine faces than men with brothers or without siblings. Lower preference for femininity in men with at least one sister could result from a negative sexual imprinting-like mechanism. Indeed, men who had sisters cohabited and interacted frequently with one or several girls during their upbringing. Such “over-exposure” to feminine faces in early environment during childhood could lead to developing later in life weaker preference towards facial femininity than men for whom such “feminine stimuli” was absent during childhood.
Another possible explanation is that our results reflect a different mechanism that does not depend on cognitive processing of facial templates. It has been suggested that individuals in mixed-sex sibling pairs assimilate traits more typically associated with the other gender46. Boys with older sisters are considered to be substantially more feminine than their peers with older brothers47. Such role-assimilation mechanism paired with specific birth order, sex-ratio and number of siblings could also explain our results.
Our study is the first to show a strong effect of family structure on preferences for femininity in faces, highlighting a possible effect of a negative imprinting-like mechanism. As other social mechanisms could affect preferences for facial femininity, further research including more detailed description of the early family environment would be particularly valuable.
The interactive effect of family structure and fatherhood status
Previous research showed that parenthood induces a decrease in baseline testosterone levels (e.g. ref. 42) and that changes in testosterone levels contribute to the strength of men’s reported attraction to femininity in women’s faces8. Consistent with these findings we have suggested that parenthood would (indirectly) lower the preference for femininity. Unexpectedly, our results have shown that there is a trend for fathers having 1.1 times stronger preferences for feminine faces than childless men (OR = 1.11 [0.99–1.24]; F1,25725 = 3.57; P = 0.059, S14), but this almost significant effect varied strongly between men with different siblings’ age, number and sex (for additional details, see S15–S17). Interestingly, in interaction with parenthood status, men that had at least one sibling differed in the preference for feminine faces compared with men without siblings (F1,25447 = 8.89; P = 0.003). Whereas fatherhood induced a drop in preference for facial femininity in men without siblings, there was a significant increase in preference for femininity among men with siblings (but the effect is significant only in men with two siblings, Fig. 1B). Among men with sisters, parenthood status strongly affected (Chi2 = 9.23, df = 1, P = 0.003; OR = 0.84 [0.76–0.94]) the preference for femininity (childless men: 0.71 [0.62–0.79], father: 0.77 [0.69–0.84]). Nevertheless, this fatherhood effect was driven mostly by men with younger sisters (Chi2 = 10.76, df = 1, P = 0.001; OR = 1.64 [1.22–2.21], Fig. 1C). Altogether, these results seem to confirm our hypothesis stating that decreased preference for femininity is triggered by fatherhood but only among men who were only-children. They also suggest that having sisters could suppress the (direct) effect of sex steroid hormones. Three potential mechanisms could explain a higher preference for feminine faces among fathers than among childless men with sisters: mere exposure to a baby’s face; perceived maternal similarity of feminine faces; and a dormant imprinting-like mechanism (Fig. 1B,C).
First, exposure to a child’s face can influence the perception of attractiveness of other faces48. As it has been suggested that feminine faces bear multiple baby-shape face cues49, it is possible that men who see facial baby cues often develop preferences towards baby features via a mere exposure effect50 and hence perceive feminine faces as more attractive. However such mere exposure is not paired with co-residence during childhood or maternal perinatal association, and hence it does not have to trigger negative sexual imprinting.
A second hypothetical explanation could be that men who have children would direct their preferences towards women with stronger maternal orientation, triggering the observed increase in preference for facial dimorphism. As it was suggested that women with feminine faces are perceived to have stronger maternal instincts than their less feminine peers51, men who already have offspring could choose them more often. Nevertheless, the most compelling argument against these two hypotheses comes from the men who were only-children – their preference for feminine faces decreased with fatherhood. Moreover, these two explanations cannot explain why the preferences increased only for men with a younger sister and not among men who had an older sister or a brother (see Fig. 1C and Table S17).
The third possible and most probable explanation for our results is that parenthood status could affect face preference by a dormant imprinting-like mechanism. Early-acquired experience leaves permanent traces that work to preserve the plasticity of the face-processing system52. The effects of early-acquired experience with feminine faces (sisters) become dormant during development in the absence of continued experience and can be reactivated in adulthood by re-exposure to the original experience53. Here, we assumed that femininity preference of men who were exposed to more feminine faces by living side-by-side with their younger sisters in early childhood was modulated by life experiences. Before becoming a father and according to the inbreeding avoidance theory, these men would have lowered facial femininity preference. When becoming a father though, the preference for facial femininity is activated by re-exposure to an infant face (other close kin, his own child) and thus preference increases. When focusing our analyses on men who had at least one or two younger sisters among their siblings (N = 307), parenthood status had a significant positive effect on the preference for femininity (Chi2 = 14.3, df = 1, P < 0.001; OR = 1.34 [1.13–1.60]), but only for men with solely other-sex siblings (childless men: 0.70 [0.60–0.78], father: 0.76 [0.67–0.83). In addition, for fathers with solely other-sex siblings, the more sisters they had the more preference for femininity increased (N = 103, Chi2 = 6.41, df = 1, P = 0.011; OR = 1.49 [1.09–2.03]); two vs one sisters). Although our data support the dormant imprinting-like mechanism hypothesis, further research is needed to confirm it through a longitudinal study monitoring the effects of changing parenthood status among our participants and by incorporating direct measurements of hormone levels.
Sensitive phase
For men with only younger sister(s) (N = 171), there was a strong effect of the interaction between fatherhood status and the age difference between participants and their sister(s) (F1,3332 = 18.8, P < 0.0001, Fig. 1D). For fathers with younger sisters, the more the age difference increased, the more the preference for facial femininity increased (F1,1716 = 20.7, P < 0.0001; OR = 1.15 [1.08–1.22]). The opposite effect was observed in childless men (F1,1581 = 2.72, P = 0.099; OR = 0.95 [0.9–1.0]). This significant interaction starting from a 4–5 year age gap between siblings suggests a sensitive phase for negative imprinting like mechanisms in childless men. Namely, reactivation of the early-acquired experience of feminine faces seems to occur when men are at least 4–5 years older than their sisters (Fig. 1D).
Conclusion
Overall, our results clearly showed that (i) among childless men, having a sister was related to decreased preference for femininity; and that (ii) having progeny was related to an increase in femininity preference, especially if men had younger sisters. Based on a large sample of men, this study demonstrates for the first time that facial femininity preferences were not only influenced by family structure (highlighting a possible effect of a negative imprinting-like mechanism) but also that fatherhood and its interactive effect could modulate these preferences (highlighting a possible effect of a dormant imprinting-like mechanism).
Current findings show a robust interactive effect of family structure (birth order, sex-ratio, number of siblings, and age difference between siblings) and parenthood status on the preference for facial femininity in men. Although the mechanisms underlying this relationship remain to be fully elucidated, we believe that these findings shed a new light on how experience acquired during different life phases (especially in early childhood) may determine the degree to which plasticity for face preference varies.
Methods
Participants
Participants were 2517 men from 25 countries with a mean age of 26 years (for socio-demographic details see Supplementary Tables S1–S7). The study was conducted online and participants were recruited by following links from various universities and social media web-sites. Only heterosexual participants with two or less siblings were involved in the analysis (N = 1304). Participants were asked to report parenthood status (father or not), number of siblings (0 - only child, 1 or 2), sex of siblings (only other-sex sibling, only same-sex sibling and, both other- and same-sex siblings) and their birth-order (firstborn, middle-born, lastborn). To assess the relevance of sibling effects on the preference for femininity, we mixed the last three factors in order to create a single variable called Family Composition, with 15 modalities (see Supplementary Table S8) covering all possible sibling composition options.
Materials and procedure
We assessed men’s preferences for femininity in women’s faces using a method that is widely used in studies of individual differences in sexual dimorphism preferences (e.g. refs 9,54). Participants were presented with 20 pairs of women faces (aged 18–24 years, chosen randomly from a previous study55), each pair consisting of a masculinised and feminised version of the same individual (differing only in sexually dimorphic characteristics of face shape, see54; for more details and an example, see Supplementary Methods (c) and Supplementary Figure S1).
The authors confirm that the methods were carried out in accordance with the approved guidelines. All experimental protocols were approved by the Ethics in Research Committee of Daugavpils University (Latvia) and written informed consent was obtained from all subjects included in this study.
Data Analysis
Data were analysed using multiple logistic regression, predicting the likelihood of making the feminine face choice. Before performing our multiple logistic regression analyses, we checked for potential problems of collinearity. As a result of collinearity issues between variables (e.g., birth-order and number of siblings, see Supplementary Tables S2–S7), we had to create a single explanatory variable called Family Composition to analyse our data. Explanatory variables for the model included Family Composition (15 modalities, see Table S8) and parenthood status (father or not). All predictors were considered fixed-effects except for participants, pairs’ identification, and country which were considered random-effects. Planned contrast analyses were performed to examine the specific effects of Family Composition or parenthood status. Means and odd-ratios (OR) were computed with the 95% confidence interval in brackets. Statistical analyses were conducted using SAS Version 9.2 (see Supplementary Methods (d) for additional explanation of data analysis).
Additional Information
How to cite this article: Marcinkowska, U. M. et al. Imprinting and flexibility in human face cognition. Sci. Rep. 6, 33545; doi: 10.1038/srep33545 (2016).
References
Todorov, A., Olivola, C. Y., Dotsch, R. & Mende-Siedlecki, P. Social Attributions from Faces: Determinants, Consequences, Accuracy, and Functional Significance. Annual Review of Psychology 66, 519–545 (2015).
Little, A. C., Connely, J., Feinberg, D. R., Jones, B. C. & Roberts, S. C. Human preference for masculinity differs according to context in faces, bodies, voices, and smell. Behav. Ecol 22, 862–868 (2011).
Perrett, D. I. In your face, the new science of human attraction. (Palgrave Macmillan, 2010).
Scott, I. M. et al. Human preferences for sexually dimorphic faces may be evolutionarily novel. Proc. Natl. Acad. Sci. USA 111, 14388–14393 (2014).
Burriss, R. P., Welling, L. L. M. & Puts, D. A. Men’s attractiveness predicts their preference for female facial femininity when judging for short-term, but not long-term, partners. Pers. Indiv. Differ. 50, 542–546 (2011).
Little, A. C., Penton-Voak, I. S., Burt, D. M. & Perrett, D. I. Investigating an imprinting-like phenomenon in humans Partners and opposite-sex parents have similar hair and eye colour. Evol. Hum. Behav. 24, 43–51 (2003).
Bobst, C. & Lobmaier. J. S. Men’s preference for the ovulating female is triggered by subtle face shape differences. Horm Behav. 62, 413–417 (2012).
Welling, L. L. M. et al. Men report stronger attraction to femininity in women’s faces when their testosterone levels are high. Horm. Behav. 54, 703–708 (2008).
Marcinkowska, U. M. et al. Cross-cultural variation in men’s preference for sexual dimorphism in women’s faces. Biol. Lett. 10, 20130850 (2014).
Jones, B. C. et al. Salivary cortisol and pathogen disgust predict men’s preferences for feminine shape cues in women’s faces. Biol. Psychol. 92, 233–240 (2013).
Little, A. C., Jones, B. C., Feinberg, D. R. & Perrett, D. I. Men’s strategic preferences for femininity in female faces. Brit. J. Psychol. 105, 364–381 (2014).
Gray, P. B., Kahlenberg, S. M., Barrett, E. S., Lipson, S. F. & Ellison, P. T. Marriage and fatherhood are associated with lower testosterone in males. Evol. Hum. Behav. 23, 193–201 (2002).
Gray, P. B. et al. Sexuality among fathers of newborns in Jamaica. BMC Pregnancy. Childbirth 15, 44 (2015).
von Sydow, K. Sexuality during pregnancy and after childbirth: A metacontent analysis of 59 studies. J. Psychosom. Res. 47, 27–49 (1999).
Mitchem, D. G. et al. Estimating the Sex-Specific Effects of Genes on Facial Attractiveness and Sexual Dimorphism. Behav. Genet. 44, 270–281 (2014).
Troisi, A. Sexual disorders in the context of Darwinian psychiatry. J. Endocrinol Invest. 26, 54–57 (2003).
McGuire, R. J., Carlisle, J. M. & Young, B. G. Sexual deviations as conditioned behaviour: a hypothsis. Behav. Res. Ther. 3, 185–190 (1965).
Enquist, M., Aronsson, H., Ghirlanda, S., Jansson, L. & Jannini, E. A. Exposure to mother’s pregnancy and lactation in infancy is associated with sexual attraction to pregnancy and lactation in adulthood. J. Sex. Med. 8, 140–147 (2011).
Enquist, M., Ghirlanda, S., Lind, J. & Aronsson, H. Parental influences on sexual preferences: The case of attraction to smoking. J. Evol. Psychol. 9, 21–41 (2011).
Knudsen, E. I. Sensitive periods in the development of the brain and behavior. J. Cognitive Neurosci. 16, 1412–1425 (2004).
Saxton, T. K., Little, A. C., DeBruine, L. M., Jones, B. C. & Roberts, S. C. Adolescents’ preferences for sexual dimorphism are influenced by relative exposure to male and female faces. Pers. Indiv. Differ. 47, 864–868 (2009).
Immelmann, K. Introduction to ethology. 1–237 (Pleneum Press, 1980).
Rantala, M. J. & Marcinkowska, U. M. The role of sexual imprinting and the Westermarck effect in mate choice in humans. Behav. Ecol. Sociobiol. 65, 859–873 (2011).
Bateson, P. Sexual imprinting and optimal outbreeding. Nature 273, 659–660 (1978).
Bischof, H. J. & Clayton, N. Stabilization of sexual preferences by sexual experience in male Zebra Finches Taeniopygia-guttata castanotis. Behaviour 118, 144–155 (1991).
ten Cate, C. & Vos, D. R. Sexual imprinting and evolutionary processes in birds: A reassessment. In Advances in the Study of Behavior (eds P. J. B. Slater, J. S. Rosenblat, C. T. Snowden & T. J. Roper ) 1–31 (Academic Press, 1999).
Bereczkei, T., Gyuris, P., Koves, P. & Bernath, L. Homogamy, genetic similarity, and imprinting; parental influence on mate choice preferences. Pers. Indiv. Differ. 33, 677–690 (2002).
Bereczkei, T., Gyuris, P. & Weisfeld, G. E. Sexual imprinting in human mate choice. P. Roy. Soc. B-Biol. Sci. 271, 1129–1134 (2004).
Marcinkowska, U. M. & Rantala, M. J. Sexual Imprinting on Facial Traits of Opposite-Sex Parents in Humans. Evol. Psychol. 10, 621–630 (2012).
Perrett, D. I. et al. Facial attractiveness judgements reflect learning of parental age characteristics. P. Roy. Soc. B-Biol. Sci. 269, 873–880 (2002).
Zei, G., Astolfi, P. & Jayakar, S. D. 1981 Correlation between fathers age and husbands age - a case of imprinting. J. Biosoc. Sci. 13, 409–418.
Keller, L. F. & Waller, D. M. Inbreeding effects in wild populations. Trends Ecol. Evol. 17, 230–241 (2002).
Schmitt, D. P. & Pilcher, J. J. Evaluating evidence of psychological adaptation - How do we know one when we see one? Psychol. Sci. 15, 643–649 (2004).
Lieberman, D., Tooby, J. & Cosmides, L. The architecture of human kin detection. Nature 445, 727–731 (2007).
Fessler, D. M. T. Neglected natural experiments Germane to the Westermarck hypothesis - The Karo Batak and the Oneida community. Hum. Nature-Int. Bios. 18, 355–364 (2007).
Shepher, J. Mate selection amond second generation Kibbutz adoloscents and adults - incest avoidance and negative imprinting. Arch. Sex. Behav. 1, 293–307 (1971).
Marcinkowska, U. M., Moore, F. R. & Rantala, M. J. An experimental test of the Westermarck effect: sex differences in inbreeding avoidance. Behav. Ecol. 24, 842–845 (2013).
Lieberman, D. Inbreeding, incest, and the incest taboo: The state of knowledge at the turn of the century. Evol. Hum. Behav. 28, 211–213 (2007).
Kaminski, G., Ravary, F., Graff, C. & Gentaz, E. Firstborns’ Disadvantage in Kinship Detection. Psychol. Sci. 21, 1746–1750 (2010).
De Smet, D., Van Speybroeck, L. & Verplaetse, J. The Westermarck effect revisited: a psychophysiological study of sibling incest aversion in young female adults. Evol. Hum. Behav. 35, 34–42 (2014).
Berg, S. J. & Wynne-Edwards, K. E. Changes in testosterone, cortisol, and estradiol levels in men becoming fathers. Mayo Clin. Proc. 76, 582–592 (2001).
Gettler, L. T., McDade, T. W., Feranil, A. B. & Kuzawa, C. W. Longitudinal evidence that fatherhood decreases testosterone in human males. Proc. Natl. Acad. Sci. USA 108, 16194–16199 (2011).
Jasienska, G. & Ellison, P. T. Father’s education is a crucial predictor of the direction of relationship between the number of children and testosterone levels in Polish men. Am. J. Phys. Anthropol. 50, 134–134 (2010).
Perrett, D. I. et al. Effects of sexual dimorphism on facial attractiveness. Nature 394, 884–887 (1998).
Rhodes, G. The evolutionary psychology of facial beauty. Annu. Rev. Psychol. 57, 199–226 (2006).
Brim, O. G. 1958 Family-structure and sex-role learning by children - a further analysis of Koch, Helen data. Sociometry 21, 1–16 (1958).
Okudaira, H., Kinari, Y., Mizutani, N., Ohtake, F. & Kawaguchi, A. Older sisters and younger brothers: The impact of siblings on preference for competition. Pers. Indiv. Differ. 82, 81–89 (2015).
Luo, L., Kendrick, K. M., Li, H. & Lee, K. Adults with siblings like children’s faces more than those without. J. Exp. Child Psychol. 129, 148–156 (2015).
Jones, D. Sexual selection, physical attractiveness and facial neoteny-cross cultural evidence and implications. Curr. Anthropol. 36, 723–748 (1995).
Zajonc, R. B. Attitudinal effects of mere exposure. J. Pers. Soc. Psychol. 9, 1–27 (1968).
Law Smith, M. J. et al. Maternal tendencies in women are associated with estrogen levels and facial femininity. Horm. Behav. 61, 12–16 (2012).
Pascalis, O., de Haan, M. & Nelson, C. A. Is face processing species-specific during the first year of life? Science 296, 1321–1323 (2002).
Macchi Cassia, V., Kuefner, D., Picozzi, M. & Vescovo, E. Early experience predicts later plasticity for face processing: Evidence for the reactivation of dormant effects. Psychol. Sci. 20, 853–959 (2009).
DeBruine, L. M., Jones, B. C., Smith, F. G. & Little, A. C. Are attractive men’s faces masculine or feminine? The importance of controlling confounds in face stimuli. J. Exp. Psychol. Hum. Percept. Perform. 36, 751–758 (2010).
Rantala, M. J. et al. Evidence for the stress-linked immunocompetence handicap hypothesis in humans. Nat. Commun. 3, 694 (2012).
Acknowledgements
The study was supported by Turku University Foundation to UMM and by a grant from the French National Research Agency to G.K. (grant no ANR-13-JSH2-0006). We are grateful to David Perrett for insightful comments on this manuscript and to Keith Morgan for language support.
Author information
Authors and Affiliations
Contributions
U.M.M. carried out and coordinated the experimental work. G.K. carried out experimental work and was responsible for the statistical analysis. All authors participated in the design of the study and in manuscript preparation.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/4.0/ Metadata associated with this Data Descriptor is available at http://www.nature.com/sdata/ and is released under the CC0 waiver to maximize reuse.
About this article
Cite this article
Marcinkowska, U., Terraube, J. & Kaminski, G. Imprinting and flexibility in human face cognition. Sci Rep 6, 33545 (2016). https://doi.org/10.1038/srep33545
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep33545
This article is cited by
-
An exploratory, cross-cultural study on perception of putative cyclical changes in facial fertility cues
Scientific Reports (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.