Abstract
Tropical forests are major contributors to the terrestrial global carbon pool, but this pool is being reduced via deforestation and forest degradation. Relatively few studies have assessed carbon storage in degraded tropical forests. We sampled 37,000 m2 of intact rainforest, degraded rainforest and sclerophyll forest across the greater Wet Tropics bioregion of northeast Australia. We compared aboveground biomass and carbon storage of the three forest types, and the effects of forest structural attributes and environmental factors that influence carbon storage. Some degraded forests were found to store much less aboveground carbon than intact rainforests, whereas others sites had similar carbon storage to primary forest. Sclerophyll forests had lower carbon storage, comparable to the most heavily degraded rainforests. Our findings indicate that under certain situations, degraded forest may store as much carbon as intact rainforests. Strategic rehabilitation of degraded forests could enhance regional carbon storage and have positive benefits for tropical biodiversity.
Similar content being viewed by others
Introduction
Although tropical forests cover only ~6% of the global land surface, they are the largest single repository of above-ground biomass carbon (ABC) stores1,2,3, containing ~195 petagrams of carbon (PgC)4,5. In addition to their total ABC storage, tropical forests are also net carbon sinks5,6,7. As a consequence of their significant carbon storage and sink capacity, tropical forests play a critical role in climate change mitigation8,9. However, despite this valuable carbon storage and potential climate change mitigation capacity, tropical forests experience high levels of annual deforestation10,11, which has been estimated to have resulted in an annual, global ABC loss of 0.26 PgCYr−1 over the period 1993–20124. Moreover, deforestation of tropical forests is the second greatest contributor of green-house gas emissions to the atmosphere after the burning of fossil fuels12,13.
In addition to deforestation, much of the remaining tropical forested area experiences various forms of degradation with the area of degraded tropical forests now estimated to exceed 500 million hectares14. Moreover, regenerating forests are estimated to exceed primary forests as the predominant form of tropical forest cover worldwide15. Degraded tropical forests store less ABC than primary forests16,17,18 and as such forest degradation results in increased atmospheric CO2 emissions16,17,19. Alternatively, however, forest regrowth within degraded forests may remove large amounts of carbon from the atmosphere5. As such, it is becoming crucial to determine the impact of forest degradation and regeneration on net CO2 emissions and overall forest carbon storage capacity16,17,20,21,22.
One problem faced when determining the carbon storage capacity of degraded forests (and thus their net CO2 emissions) within complex tropical forest regions is that landscape-scale factors often determine the impact of forest degradation on carbon storage. These factors include: variability in the availability of constraining environmental resources23,24, differences in vegetation composition25,26, forest structural variation27,28, and the applied forest management regime8,29. As a whole, it is still unclear what impact each of these factors has on the dynamics of carbon storage in complex tropical forested landscapes23,30. As such, if the development of optimal management strategies to enhance landscape-scale carbon storage in complex tropical forested landscapes is to occur, determining the impact of individual landscape-scale factors on the carbon storage capacity of degraded forests is of utmost importance20,31.
The Wet Tropics bioregion of northeast Australia is a complex and contested (for land use) landscape32 which is primarily composed of one of the oldest rainforests on earth33. This region has been described as the second most irreplaceable natural world heritage area34, and the sixth most irreplaceable protected area on the planet35. The significant biodiversity values of the Wet Tropics bioregion are well documented36,37,38. However, the carbon storage values of the component vegetation types within the Wet Tropics bioregion is yet to be determined – precluding carbon storage within restoration plantings of the area39.
Here we evaluate, the biomass and carbon storage of intact closed-canopy forests (hereafter termed rainforest), degraded closed-canopy forests (hereafter termed degraded forest) and sclerophyll forests within a complex and heterogeneous landscape of the Wet Tropics bioregion. In addition, we examine the influence of forest structural features (e.g. tree size) and disturbance upon forest biomass and carbon storage. We also compare the impact of rainfall and elevational gradients upon the carbon storage of these vegetation types. Finally, we discuss mechanisms whereby appropriate site selection and management of degraded forests, may allow for potential policy interventions which enhance carbon storage in the tropical forests of this landscape and additionally aid biodiversity conservation.
Results
Structural variation across the forest types
We counted and measured a total of 1,438 trees in rainforests, 1193 trees in degraded forests and 693 trees in the sclerophyll forests. There was a significant difference in the number of trees ≥10 cm dbh among the examined forest types (χ2 = 18.269, df = 2, p2-tailed = <0.001), with a pairwise post hoc comparison, showing that rainforests (RF) had significantly more trees than degraded forest (DF) and sclerophyll forests (SF) (RF-DF p2-tailed = 0.013; RF- SF p2-tailed = <0.001) (Fig. 1). However, there was no significant difference in the number of trees between degraded forests and sclerophyll forests (p2-tailed = 1.00) (Fig. 1). The average tree dbh (cm) was significantly different among the three forest types (χ2 = 16.295, df = 2, p2-tailed = <0.001), with rainforests and degraded forests possessing significantly larger trees than sclerophyll forests (RF- SF p2-tailed = 0.002; DF- SF p2-tailed = 0.004) (Fig. 1). Analogously, the number of fallen logs (≥10 cm diameter) per ha was significantly different among the forest types (χ2 = 15.406, df = 2, p2-tailed = <0.001), with the post hoc pairwise comparison again finding that rainforests and degraded forests possessed more fallen logs than sclerophyll forests (RF- SF p2-tailed = 0.006; DF- SF p2-tailed = 0.002) (Fig. 1). Finally, the co-efficient of variance (CV) of tree dbh was also significantly different among the forest types (χ2 = 13.689, df = 2, p2-tailed = 0.001), which was again driven by the significantly larger values of CV of tree dbh in the rainforest and degraded forests compared to the sclerophyll forest (RF- SF p2-tailed = <0.001; DF- SF p2-tailed = 0.046) (Fig. 1).
The average number of trees (≥10 cm dbh) per ha, average tree diameter at breast height (cm), average number of fallen logs (≥10 cm diameter) and average coefficient of variance (CV) of tree diameter at breast height (cm) in the examined plots of rainforest (RF) (n = 29), degraded forest (DF) (n = 13) and sclerophyll forest (SF) (n = 32) within the Wet Tropics bioregion of northeast Australia.
Above ground biomass and carbon stock
There was a significant difference among the amount of above ground biomass (Mg ha−1) and above ground carbon (Mg ha−1) stored within the examined rainforests, degraded forests and sclerophyll forests (χ2 = 33.064, df = 2, p2-tailed = <0.001 in each case). Using a pairwise post hoc comparison, we found that there was significantly more above ground biomass (Mg ha−1) and above ground carbon stored (Mg ha−1) within the rainforests and the degraded forests than within the sclerophyll forest (p2-tailed = <0.001 in each case) (RF- SF p2-tailed = <0.001; DF- SF p2-tailed = 0.009). However, there was no significant difference in the amount of above ground biomass and above ground carbon stored in the rainforests and degraded forests (RF- DF p2-tailed = 0.442) (Fig. 2).
Variation and distribution in carbon storage of the examined vegetation types
The Nonmetric Multidimensional Scaling (NMS) ordination of above ground carbon storage of plots in variable space explained 96.3% cumulative variation in the examined data, where the x-axis (axis 1) represented 87.7% and y axis (axis 2) 8.6% of the variation (Fig. 3). The x-axis showed a strong correlation with the number of trees and above ground carbon storage variables (r = 0.929, and 0.681 respectively), whilst the y axis showed a strong correlation with the average tree dbh and above ground carbon storage variables (r = −0.820, and −0.510 respectively) (Fig. 3).
Most of the rainforest plots had higher levels of above ground carbon storage than those of sclerophyll forests (Fig. 3), whilst the above ground carbon storage in the degraded forest plots varied from high levels similar to those of rainforests through to low levels similar to plots within sclerophyll forests (Fig. 3). As such, rainforests plots and sclerophyll forests plots occupied somewhat distinct ordination spaces whilst those of the degraded forest plots intergraded between the two; though they were, in general, more similar to those of the rainforest plots (Fig. 3).
Factors determining carbon stock
The backwards, stepwise, negative binomial generalized linear model (GLM) process identified three significant explanatory variables for determining the carbon storage of plots (R2 = 0.888; null deviance-residual deviance/null deviance): the number of fallen logs per plot, average tree diameter breast height per plot and the tree abundance per plot (Fig. 4a–c and Table 1). All of these explanatory variables displayed a positive correlation with the carbon storage (Mg ha−1) of plots (Fig. 4a–c and Table 1).
The significant relationship between (a) fallen logs (≥10 cm diameter) ha−1 (log transformed), (b) average tree diameter breast height (cm) and (c) tree abundance and carbon storage (Mg ha−1) of the examined plots, within the Wet Tropics bioregion northeast Australia. Filled circles represent the plot (50*10 m) values. The trend line was constructed using a binomial GLM with logit link function and shaded areas represent the 95% confidence intervals.
Discussion
We found that rainforests within the Wet Tropics bioregion store the highest levels of above ground biomass carbon (ABC) of the three examined vegetation types and when these forests experience degradation their storage capacity is reduced (Figs 2 and 3). However, the reduction in ABC storage values of plots in degraded forests compared with those of rainforest was not significant (Fig. 2) although degraded forest plots did display considerably more variation in ABC storage (Fig. 3). We suggest that it is likely that the examined degraded forests are in an advanced stage of regeneration given the lack of significant difference between their average tree size (dbh) and that of the rainforest. Moreover, the examined degraded forests on a global spectrum still store proportionately high values of ABC (241.04 ± 27.09 Mg ha−1) when compared with other degraded tropical forests. For example, our reported values of ABC for the examined degraded forests (Fig. 2) are higher than those of Ioki, et al.20 who estimated 52.18–229.11 Mg ha−1, and 136.00–382.59 Mg ha−1 ABC for the highly degraded and moderately degraded tropical rain forests of northern Borneo, and for those of Usuga, et al.27 who reported 99.6 Mg ha−1 and 85.7 Mg ha−1 ABC storage from Tropical Pine and Teak forest plantations in Colombia. Nonetheless, our observation of a non-significant decline in the ABC storage capacity of degraded forests compared to non-degraded rainforests is supported by findings reported elsewhere from within the tropics16,17,18,40.
Much of the decline in ABC storage within the degraded forests we examined appears to be due to increased disturbance. For instance, degraded forests when compared with non-degraded rainforests were found to display less trees (Fig. 1) and possess a higher number of fallen logs (Fig. 1). Furthermore, many of the degraded forests we examined were fragmented, a process which is known to result in higher levels of forest disturbance and tree loss through an increased susceptibility to wind damage41 and an altered microclimate on forest edges21,42. Moreover, both wind damage and microclimatic alterations within forests degraded by fragmentation are known to result in the disproportionate loss of large trees, especially on forest edges18,43,44. This loss of large trees can significantly alter the carbon storage capacity of degraded forests as large trees are known to drive tropical forest ABC storage45. In addition, forest fragmentation is known to increase wind damage susceptibility which may be particularly pertinent to lowland forests of this geographic region as they are exposed to regular cyclonic impacts, with the greater Wet Tropics bioregion experiencing 45 recorded east-to-west moving tropical cyclone impacts over the period 1858–201146,47. Therefore, strategies that minimize the disturbance of degraded forests and especially forest edges, might allow for enhanced ABC storage through successional recovery of the tree community and re-instatement of resilience to natural disturbances, particularly within larger fragments of forest. For instance, employing wind disturbance mitigation strategies such as wind-buffer plantings along the forest edges48 of the degraded forests of the region could substantially assist in decreasing forest disturbance41 and thus increase the ABC storage of these forests.
Sclerophyll forests within the study region had the smallest average tree size (dbh) (Fig. 1) and stored the least ABC of the three examined vegetation types (Fig. 2). The small average size (dbh) of trees within the sclerophyll forests compared to the other vegetation types is unsurprising given the less productive environmental envelope this vegetation type occupies (i.e. lower rainfall) and the fact that sclerophyll forests are pyrophytic and as such recruitment events are often determined by fire events and the time intervals between these49. Previous work50 has suggested that one of the main determinants of the distribution of the rainforest and sclerophyllous vegetation types within the examined region, may be fire, especially given the pyrophobic nature of the rainforest vegetation49,50,51. Therefore, management practices that aim to optimize ABC storage within the Wet Tropics bioregion whilst supporting the different species assemblages housed by rainforest and sclerophyll forests should, where practicable and appropriate, focus on the exclusion of fire from the rainforest component of the landscape to allow for the successional repair of the degraded rainforest vegetation type.
In addition to the exclusion of fire from the degraded forests, assisted restoration of these forests within the Wet Tropics bioregion may be an effective management strategy to allow for significant net ABC storage gains. This is suggested as the degraded forests within the region vary in their ABC storage capacity from values at a low end similar to sclerophyllous vegetation through to those of comparable non-degraded rainforests (Fig. 3). In particular, restoration of localized factors which support the retention of large trees and increase tree abundance would significant increase ABC storage across all forest types and within the degraded forest in particular (Figs 3 and 4, Table 1; Slik, et al.45). Although an increase in the number of fallen logs (as proxy for disturbance) was also found to increase the ABC storage capacity of the examined forests (Fig. 4); it is highly likely (given previous research on forest disturbance52,53) that increased disturbance within these forests would result in an asymptote and eventual negative relationship occurring between the number of fallen logs and ABC storage. As such, utilizing intermediate disturbance to attain increase ABC storage of forests in the studied region would be problematic and impractical.
Finally, the use of land within the Wet Tropics bioregion is highly contested32. Consequently, within this landscape, multi-value land usage strategies may maximize the likelihood of degraded forest retention. For instance, as well as their significant ABC storage values (Fig. 2), tropical rainforests are known to house the zenith of terrestrial biodiversity54. Additionally, remnant, fragmented and degraded forests can provide an important biodiversity repository for many complex tropical landscapes21,55,56. Consequently, degraded forest management within the Wet Tropics bioregion provides considerable opportunities for integrating ABC storage values with biodiversity conservation. In particular, the uplands of the Wet Tropics bioregion, as well as providing an area of low disturbance and thus optimal locations for ABC storage in both rainforest and degraded forests (Fig. 3), are also a known “hotspot” for endemism and diversity of numerous biota57,58,59 many of which are under threat38,60. Moreover, recent studies of forest restoration within this region suggest that secondary and degraded forests restoration may be passively enhanced through selection of sites in close proximity to primary forest61; although if maximal biodiversity outcomes are to be gained specific species may need to be actively restored62. Finally, rainforest restoration should not occur at the expense of other distinct, remnant vegetation types such as sclerophyll forests as a net biodiversity loss could occur due to the different species assemblages they support32.
Conclusion
To maximise ABC storage within the complex landscape of the Wet Tropics bioregion, it is optimal to conserve primary rainforests at sites that experience low levels of disturbance. Additionally, although degraded forests do not store as much ABC as non-degraded rainforest they play a considerable supplementary role in ABC storage and given appropriate management (i.e. disturbance minimization through fire exclusion and edge buffer plantings) and sufficient recovery, they can store as much ABC as non-degraded rainforest. Any additional ABC storage provided by degraded forests will come through the accumulation of additional carbon from atmosphere and thereby contribute to climate change mitigation. In addition, if degraded forests in close proximity to primary forests can be restored and supplementarily seeded with selected tree species they may also provide additional and considerable biodiversity conservation capacity.
Methods
The study area
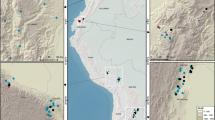
Our study was conducted in the Wet Tropics bioregion, northeast Australia (Fig. 5). The total area of the bioregion is ca. two million ha63, most of which experiences a seasonally wet tropical climate. The total mean annual rainfall ranges from 1200 mm to 4000 mm (although the highest mountain peaks may receive 8000 mm yr−1) and the mean annual temperature ranges from 17 °C to 31 °C63. The elevation of the bioregion ranges from a few meters above mean sea level (msl) to ~1000 m although the highest peak within the region is 1622 m. The heterogeneous and complex landscape of the region is dominated by contiguous rainforests and sclerophyll forests with environmentally defined boundaries50. The structure, composition and distribution of rainforests and sclerophyll forests in the Wet Tropics bioregion are largely determined by rainfall, elevation and soil types64,65.
The maps were created using Esri ArcMap 10.2. (http://www.arcgis.com).
Attributes of studied forest types
The rainforests of the Wet Tropics bioregion are the largest remaining rainforests in Australia66, contain globally recognized biodiversity35 and provide valuable ecosystem services67. The rainforests within the study area are the remnants (and regrowth) of a formerly much larger rainforest expanse that covered large portions of the greater Wet Tropics bioregion. Significant deforestation of the area began in approximately 1880 and proceeded rapidly during the next 5 decades68. Moreover, most of the remaining rainforests of this region have been selectively logged since 1880 for valuable hardwood timber species such as Red Cedar (Toona ciliata)69. However, all logging of approximately, 45% (894,420 ha) (mainly rainforest) of the Wet Tropics bioregion ceased with its inscription on the World Heritage list in 1988 as a property that fulfilled all four natural criteria for listing37,63. The examined rainforests within the study area can generally be described as complex mesophyll to notophyll vine forest (regional ecosystem 7.8.2 and 7.8.4) with drier areas transitioning into complex semi-evergreen notophyll vine forest (regional ecosystem 7.8.3)65,70. Within the complex mesophyll vine forest, multiple continuous canopies may be present with the upper canopy averaging a height of 20–40 m65. Deciduous tree species are rare, however woody lianas, epiphytes and ferns are quite common resulting in a complex forest structure65,70.The rainforests of this region are biologically diverse and dominated by trees from Lauraceae, Moraceae, Myrtaceae, Rutaceae and Sapindaceae families71. The sclerophyll forests of this region are relatively open, with a grassy understory and the canopy is dominated by trees of the following genera: Eucalyptus, Corymbia, Melaleuca, Acacia, Allocasuarina, Casuarina, Lophostemon and Syncarpia65. We defined degraded forest as rainforest that was in the process of recovering after extensive disturbance (e.g. logging or clearing) or were fragmented.
Sampling regime and data collection
We sampled a total of 74 plots: 29 rainforest, 32 sclerophyll forest and 13 degraded forest plots. In each forest types, sampling points were predetermined using ESRI ArcGIS 10.2 prior to field data collection to a) avoid creeks and water bodies; and b) minimize edge effects by maintaining at least 20 m distance between our plots and other land uses. Forest types in the Wet Tropics bioregion are distributed across a wide range of environmental gradients65. Therefore, we sampled sites across an: elevation gradient ranging from 12 meters above mean sea level (msl) to more than 1000 m; across a mean annual rainfall gradient ranging from less than 1000 mm to more than 3500 mm mean annual rainfall; and across a wide range of soils including: alluvium, genesis, fine sedimentary, laterite and andesite. Mean annual rainfall data was determined using long-term records for the region provided by the Wet Tropics Management Authority, Queensland, Australia72. Finally, all the sites were georeferenced and general environmental and landscape features such as slope and elevation recorded.
The size of each sampling plot was 0.05 ha (50 m * 10 m transect). We adopted 0.05 ha as our plot size as this has been previously found suitable to adequately sample widely spaced large trees, along with the relatively more smaller trees39,73. Moreover, for our data collection we used a transect method which has previously been determined suitable for estimating high densities of trees in rainforests39 and sclerophyll forests74. We measured the diameter at breast height (dbh) of all trees ≥10 cm within our plots to the nearest millimeter. In each of the plots we also counted the number of fallen logs ≥10 cm diameter on the forest floor (≤1 m above ground level).
Estimation of above ground biomass carbon
Preece, et al.39 compared the accuracy of biomass estimation methods for forests within the Wet Tropics bioregion and concluded that the Chave, et al.75 allometric provided the best and most reliable estimate for the region. As such, we estimated above ground biomass (AGB) following Chave’s allometric equation75. Moreover, the Chave, et al.75 allometric was especially developed for tropical forests using data from 27 sites across the tropics, where 2,410 trees ≥5 cm dbh (with maximum 156 cm) have been directly harvested75, and subsequently found suitable for small to large diameter range trees39. To convert AGB into biomass carbon storage we used a conversion factor of 0.47 which is the recommended value from the Intergovernmental Panel for Climate Change for tropical forests76. Like other regions in the world, wood density in the trees of Wet Tropics forests vary widely from species to species and across different forest types77 and due to the diversity of the examined tropical forest many species specific densities are still as yet unknown. To compensate for this uncertainty, in this study wood density estimates were calculated using the Australian Governments Department of Climate Change and Energy Efficiency reported default value for Australian tropical forests of 0.5 g cm−3 (500 kgm−3)78. Whilst this approach may underestimate or overestimate the above ground carbon storage in some plots it utilizes the best current knowledge on tropical Australian rainforest tree species average wood density whilst still remaining practicable. Moreover, the present study still provides a clear comparative analysis of above ground carbon storage across the examined forest types, though of course, were it available, using species level wood density would produce more precise results. Consequently, AGB was calculated using equation (1):

where AGB is measured in kg, dbh is measured in cm, and ρ is wood density measured in g cm−3.
Above ground biomass estimates were then converted to carbon estimates using equation (2):

Statistical analyses
All statistical analyses were conducted in IBM SPSS 20, PCORD 6 and Program R79. We used independent Kruskal-Wallis tests (2-tailed, α = 0.05) to compare above ground biomass, above ground carbon storage and forest structural attributes (number of stems (≥10 cm dbh), average tree dbh, fallen logs (≥10 cm diameter), and CV of tree dbh) between the rainforest, degraded forest and sclerophyll forest types. A NMS ordination was performed (in PCORD 6) to investigate the plot based variation in above ground carbon storage in relation to the other examined attributes of the examined forests. In the NMS ordination analysis we utilized Sorensen and Orthogonal Principal Axis (rotation).
The significant explanatory variables dictating the carbon storage of plots were determined using a binomial generalized linear model (GLM) with a logit link function, followed by a backwards, stepwise regression comparison. Prior to creating the global model and candidate model comparisons we performed data exploration and checked for (and removed) correlated predictor variables following the protocol of Zuur, et al.80. We selected a priori a global model in which the carbon storage of plots was a function of: elevation (m), slope (degree), number of fallen logs (≥10 cm diameter), canopy cover (%), mean annual rainfall (mm), tree diameter breast height (cm), tree abundance and forest type (rainforest, degraded forest or sclerophyll). The best model was then determined through a backwards stepwise model comparison whereby nested models were compared using the drop1 function and AIC model values, and the best model was that which contained only significant variables and the lowest AIC model value.
Additional Information
How to cite this article: Alamgir, M. et al. Degraded tropical rain forests possess valuable carbon storage opportunities in a complex, forested landscape. Sci. Rep. 6, 30012; doi: 10.1038/srep30012 (2016).
References
Laurance, W. F. Can Carbon Trading Save Vanishing Forests? BioScience 58, 286–287, doi: 10.1641/b580402 (2008).
Lewis, S. L. Tropical forests and the changing earth system. Philosophical Transactions of the Royal Society of London B: Biological Sciences 361, 195–210 (2006).
Saatchi, S. S. et al. Benchmark map of forest carbon stocks in tropical regions across three continents. Proceedings of the National Academy of Sciences 108, 9899–9904, doi: 10.1073/pnas.1019576108 (2011).
Liu, Y. Y. et al. Recent reversal in loss of global terrestrial biomass. Nature Clim. Change 5, 470–474, doi: 10.1038/nclimate2581 http://www.nature.com/nclimate/journal/v5/n5/abs/nclimate2581.html#supplementary-information (2015).
Pan, Y. et al. A Large and Persistent Carbon Sink in the World’s Forests. Science 333, 988–993 (2011).
Lewis, S. L. et al. Increasing carbon storage in intact African tropical forests. Nature 457, 1003–1006, doi: http://www.nature.com/nature/journal/v457/n7232/suppinfo/nature07771_S1.html (2009).
Baker, T. R. et al. In Tropical Forests and Global Atmospheric Change (eds Y. Mahli & O. Phillips ) (Oxford University Press, 2005).
Fahey, T. J. et al. Forest carbon storage: ecology, management, and policy. Frontiers in Ecology and the Environment 8, 245–252, doi: 10.1890/080169 (2009).
Carrasco, L. R. & Papworth, S. K. A ranking of net national contributions to climate change mitigation through tropical forest conservation. Journal of Environmental Management 146, 575–581, doi: http://dx.doi.org/10.1016/j.jenvman.2014.08.016 (2014).
Achard, F. et al. Determination of Deforestation Rates of the World’s Humid Tropical Forests. Science 297, 999–1002, doi: 10.1126/science.1070656 (2002).
Hansen, M. C. et al. High-Resolution Global Maps of 21st-Century Forest Cover Change. Science 342, 850–853 (2013).
Laurance, W. F., Laurance, S. G. & Delamonica, P. Tropical forest fragmentation and greenhouse gas emissions. Forest Ecology and Management 110, 173–180, doi: http://dx.doi.org/10.1016/S0378-1127(98)00291-6 (1998).
Pütz, S. et al. Long-term carbon loss in fragmented Neotropical forests. Nat Commun 5, doi: 10.1038/ncomms6037 (2014).
Ghazoul, J., Burivalova, Z., Garcia-Ulloa, J. & King, L. A. Conceptualizing Forest Degradation. Trends in Ecology & Evolution 30, 622–632, doi: 10.1016/j.tree.2015.08.001 (2015).
Chazdon, R. L. Second growth: The promise of tropical forest regeneration in an age of deforestation. (University of Chicago Press, 2014).
Achard, F., Eva, H. D., Mayaux, P., Stibig H.-J. & Belward, A. Improved estimates of net carbon emissions from land cover change in the tropics for the 1990s. Global Biogeochem. Cycles, 18, GB2008 (2004).
Bustamante, M. M. C. et al. Towards an integrated monitoring framework to assess the effects of tropical forest degradation and recovery on carbon stocks and biodiversity. Global Change Biology In press (2015).
Laurance, W. F. et al. Biomass Collapse in Amazonian Forest Fragments. Science 278, 1117–1118 (1997).
Asner, G. P. et al. Selective Logging in the Brazilian Amazon. Science 310, 480–482 (2005).
Ioki, K. et al. Estimating above-ground biomass of tropical rainforest of different degradation levels in Northern Borneo using airborne LiDAR. Forest Ecology and Management 328, 335–341, doi: http://dx.doi.org/10.1016/j.foreco.2014.06.003 (2014).
Magnago, L., Rocha, M., Meyer, L., Martins, S. & Meira-Neto, J. Microclimatic conditions at forest edges have significant impacts on vegetation structure in large Atlantic forest fragments. Biodiversity and Conservation 24, 2305–2318, doi: 10.1007/s10531-015-0961-1 (2015).
Malhi, Y. & Grace, J. Tropical forests and atmospheric carbon dioxide. Trends in Ecology & Evolution 15, 332–337, doi: http://dx.doi.org/10.1016/S0169-5347 (00)01906-6 (2000).
Tian, H. et al. Climatic and biotic controls on annual carbon storage in Amazonian ecosystems. Global Ecology and Biogeography 9, 315–335, doi: 10.1046/j.1365-2699.2000.00198.x (2000).
Cramer, W. et al. Tropical forests and the global carbon cycle: impacts of atmospheric carbon dioxide, climate change and rate of deforestation. Vol. 359 (2004).
Corlett, R. T. & Primack, R. B. Tropical rainforests and the need for cross-continental comparisons. Trends in Ecology & Evolution 21, 104–110, doi: http://dx.doi.org/10.1016/j.tree.2005.12.002 (2006).
Glenday, J. Carbon storage and emissions offset potential in an East African tropical rainforest. Forest Ecology and Management 235, 72–83, doi: http://dx.doi.org/10.1016/j.foreco.2006.08.014 (2006).
Usuga, J. C. L., Toro, J. A. R., Alzate, M. V. R. & de Jesús Lema Tapias, Á. Estimation of biomass and carbon stocks in plants, soil and forest floor in different tropical forests. Forest Ecology and Management 260, 1906–1913, doi: http://dx.doi.org/10.1016/j.foreco.2010.08.040 (2010).
Zhang, J. et al. Carbon storage by ecological service forests in Zhejiang Province, subtropical China. Forest Ecology and Management 245, 64–75, doi: http://dx.doi.org/10.1016/j.foreco.2007.03.042 (2007).
Turner, D. P. et al. Decadal trends in net ecosystem production and net ecosystem carbon balance for a regional socioecological system. Forest Ecology and Management 262, 1318–1325, doi: http://dx.doi.org/10.1016/j.foreco.2011.06.034 (2011).
Zuidema, P. A. et al. Tropical forests and global change: filling knowledge gaps. Trends in Plant Science 18, 413–419, doi: http://dx.doi.org/10.1016/j.tplants.2013.05.006 (2013).
Fearnside, P. M. Are climate change impacts already affecting tropical forest biomass? Global Environmental Change 14, 299–302, doi: http://dx.doi.org/10.1016/j.gloenvcha.2004.02.001 (2004).
Stork, N. & Turton, S. M. Living in a Dynamic Tropical Forest Landscape. (Blackwell Publishing 2008).
Byrne, M. et al. Decline of a biome: evolution, contraction, fragmentation, extinction and invasion of the Australian mesic zone biota. Journal of Biogeography 38, 1635–1656, doi: 10.1111/j.1365-2699.2011.02535.x (2011).
Bertzky, B. et al. Terrestrial biodiversity and the World Heritage list: Identifying broad gaps and potential candidate sites for inclusion in the natural World Heritage network., xiv+70pp (Cambridge, UK, 2013).
Le Saout, S. et al. Protected Areas and Effective Biodiversity Conservation. Science 342, 803–805, doi: 10.1126/science.1239268 (2013).
Hilbert, D. W., Ostendorf, B. & Hopkins, M. S. Sensitivity of tropical forests to climate change in the humid tropics of north Queensland. Austral Ecology 26, 590–603, doi: 10.1046/j.1442-9993.2001.01137.x (2001).
UNESCO. Decision-12COM XIV.A-Inscription: Wet Tropics of Queensland. World Heritage List. (Available at: http://whc.unesco.org/en/list/486 (accessed 7 October 2015). 1988).
Williams, S. E., Bolitho, E. E. & Fox, S. Climate change in Australian tropical rainforests:an impending environmental catastrophe. Proc. R. Soc. Lond. B. 270, 1887–1892 (2003).
Preece, N. D., Crowley, G. M., Lawes, M. J. & van Oosterzee, P. Comparing above-ground biomass among forest types in the Wet Tropics: Small stems and plantation types matter in carbon accounting. Forest Ecology and Management 264, 228–237, doi: http://dx.doi.org/10.1016/j.foreco.2011.10.016 (2012).
Pregitzer, K. S. & Euskirchen, E. S. Carbon cycling and storage in world forests: biome patterns related to forest age. Global Change Biology 10, 2052–2077, doi: 10.1111/j.1365-2486.2004.00866.x (2004).
Laurance, W. F. & Curran, T. J. Impacts of wind disturbance on fragmented tropical forests: A review and synthesis. Austral Ecology 33, 399–408 (2008).
Williams-Linera, G., Domínguez-Gastelú, V. & García-Zurita, M. E. Microenvironment and Floristics of Different Edges in a Fragmented Tropical Rainforest. Conservation Biology 12, 1091–1102, doi: 10.1046/j.1523-1739.1998.97262.x (1998).
Laurance, W. F., Delamonica, P., Laurance, S. G., Vasconcelos, H. L. & Lovejoy, T. E. Conservation: Rainforest fragmentation kills big trees. Nature 404, 836–836 (2000).
Oliveira, M. A., Santos, A. M. M. & Tabarelli, M. Profound impoverishment of the large-tree stand in a hyper-fragmented landscape of the Atlantic forest. Forest Ecology and Management 256, 1910–1917, doi: http://dx.doi.org/10.1016/j.foreco.2008.07.014 (2008).
Slik, J. W. F. et al. Large trees drive forest aboveground biomass variation in moist lowland forests across the tropics. Global Ecology and Biogeography 22, 1261–1271, doi: 10.1111/geb.12092 (2013).
Turton, S. M. Landscape-scale impacts of Cyclone Larry on the forests of northeast Australia, including comparisons with previous cyclones impacting the region between 1858 and 2006. Austral Ecology 33, 409–416, doi: 10.1111/j.1442-9993.2008.01896.x (2008).
Turton, S. M. Securing Landscape Resilience to Tropical Cyclones in Australia’s Wet Tropics under a Changing Climate: Lessons from Cyclones Larry (and Yasi). Geographical Research 50, 15–30, doi: 10.1111/j.1745-5871.2011.00724.x (2012).
Goosem, S. & Tucker, N. I. J. Repairing the rainforest (second edition). (Wet Tropics Management Authority and Biotropica Australia Pty Ltd, Cairns., 2013).
Bowman, D. M. J. S. Australian rainforests: islands of green in a land of fire. (Cambridge University Press., 2000).
Ash, J. The Location and Stability of Rainforest Boundaries in North-Eastern Queensland, Australia. Journal of Biogeography 15, 619–630, doi: 10.2307/2845440 (1988).
Fensham, R. J., Fairfax, R. J., Butler, D. W. & Bowman, D. M. J. S. Effects of fire and drought in a tropical eucalypt savanna colonized by rain forest. Journal of Biogeography 30, 1405–1414, doi: 10.1046/j.1365-2699.2003.00934.x (2003).
Laurance, W. F. et al. Rapid decay of tree-community composition in Amazonian forest fragments. Proceedings of the National Academy of Sciences 103, 19010–19014, doi: 10.1073/pnas.0609048103 (2006).
Nascimento, H. E. M. & Laurance, W. F. Biomass dynamics in Amazonian forest fragments. Ecological Applications 14, 127–138, doi: 10.1890/01-6003 (2004).
Gibson, L. et al. Primary forests are irreplaceable for sustaining tropical biodiversity. Nature 478, 378–381, doi: http://www.nature.com/nature/journal/v478/n7369/abs/nature10425.html#supplementary-information (2011).
Arroyo-Rodríguez, V. & Mandujano, S. The Importance of Tropical Rain Forest Fragments to the Conservation of Plant Species Diversity in Los Tuxtlas, Mexico. Biodivers Conserv 15, 4159–4179, doi: 10.1007/s10531-005-3374-8 (2006).
Edwards, D. P. & Laurance, W. F. Biodiversity Despite Selective Logging. Science 339, 646–647 (2013).
Williams, S. E., Pearson, R. G. & Walsh, P. J. Distributions and biodiversity of the terrestrial vertebrates of Australia’s Wet Tropics: a review of current knowledge. Pacific Conservation Biology 2, 327–362 (1995).
Williams, S. E. & Pearson, R. G. Historical rainforest contractions, localized extinctions and patterns of vertebrate endemism in the rainforests of Australia’s wet tropics. Proceedings of the Royal Society B: Biological Sciences 264, 709–716 (1997).
Yeates, D. K., Bouchard, P. & Monteith, G. B. Patterns and levels of endemism in the Australian Wet Tropics rainforest: evidence from flightless insects. Invertebrate Systematics 16, 605–619 (2002).
Costion, C. M. et al. Will tropical mountaintop plant species survive climate change? Identifying key knowledge gaps using species distribution modelling in Australia. Biological Conservation 191, 322–330, doi: http://dx.doi.org/10.1016/j.biocon.2015.07.022 (2015).
Sloan, S., Goosem, M. & Laurance, S. Tropical forest regeneration following land abandonment is driven by primary rainforest distribution in an old pastoral region. Landscape Ecology 1–18, doi: 10.1007/s10980-015-0267-4 (2015).
Shoo, L. P., Freebody, K., Kanowski, J. & Catterall, C. P. Slow recovery of tropical old field rainforest regrowth and the value and limitations of active restoration. Conservation Biology, In press, doi: 10.1111/cobi.12606 (2015).
Goosem, S. Update of original Wet tropics of Queensland nomination dossier. (Wet Tropics Management Authority Cairns, Queensland, Australia., 2002).
Webb, L. J. Environmental relationship of the structural types of Australian rainforest vegetation. Ecology 49, 296–311 (1968).
Tracey, J. G. (CSIRO, Melbourne, 1982).
Stork, N. E., Goosem, S. & Turton, S. M. In Biodiversity Hotspots (eds F. E. Zachos & J. C. Habel ) 311–332 (Springer-Verlag 2011).
Alamgir, M., Turton, S. M., Macgregor, C. J. & Pert, P. L. Assessing regulating and provisioning ecosystem services in a contrasting tropical forest landscape. Ecological Indicators 65, 319–334 (2016).
Turton, D. In Living in a Dynamic Tropical Forest Landscape (eds N. E. Stork & S. M. Turton ) 71–80 (Blackwell, 2008).
Society, E. H. Eacham Shire, Atherton Tableland, North Queensland: Yesterday and Today. Eacham Historical Society, Milla Milla., (1979).
Magrach, A., Rodríguez-Pérez, J., Campbell, M. & Laurance, W. F. Edge effects shape the spatial distribution of lianas and epiphytic ferns in Australian tropical rain forest fragments. Applied Vegetation Science 17, 754–764. (2014).
Adam, P. History and ecology of rainforest diversity. Trends in Plant Science 3, 287–288, doi: http://dx.doi.org/10.1016/S1360-1385 (98)01286-2 (1998).
WTMA. Vegetation of the wet tropics of Queensland. (Wet Tropics Management Authority (WTMA). Cairns, Australia, 2009).
Pearson, T., Walker, S. & Brown, S. Sourcebook for Land Use, Land-Use Change and Forestry Projects., (BioCarbon Fund of the World Bank & Winrock International., 2005).
Burrows, W. H. et al. Growth and carbon stock change in eucalypt woodlands in northeast Australia: ecological and greenhouse sink implications. Global Change Biology 8, 769–784, doi: 10.1046/j.1365-2486.2002.00515.x (2002).
Chave, J. et al. Tree allometry and improved estimation of carbon stocks and balance in tropical forests. Oecologia 145, 87–99, doi: 10.1007/s00442-005-0100-x (2005).
IPCC. IPCC Guidelines for National Greenhouse Gas Inventories. (Prepared by the National Greenhouse Gas Inventories Programme; Institute for Global Environmental Strategies, Kanagawa, Japan., 2006).
Kanowski, J. & Catterall, C. P. Carbon stocks in above-ground biomass of monoculture plantations, mixed species plantations and environmental restoration plantings in north-east Australia. Ecological Management & Restoration 11, 119–126, doi: 10.1111/j.1442-8903.2010.00529.x (2010).
Department of Climate Change and Energy Efficiency. Australian National Greenhouse Accounts National Inventory Report 2008. 276 (Department of Climate Change and Energy Efficiency, Canberra, 2010).
R. Core Team. R: A language and environment for statistical computing. (R Foundation for Statistical Computing, Vienna, Austria, 2015).
Zuur, A. F., Ieno, E. N. & Elphick, C. S. A protocol for data exploration to avoid common statistical problems. Methods in Ecology and Evolution 1, 3–14 (2010).
Acknowledgements
We acknowledge funding for this research project from the Australian Research Council (Discovery grant awarded to William Laurance), Skyrail Rainforest Foundation, Australia, Wet Tropics Management Authority, Australia, Cowan Grant Trust and the Graduate Research Scheme of James Cook University, Australia. Additionally, we would like to thank Wimolwan Khanthap, Dr. Ainhoa Magrach and volunteers Edison Salas Castelo, Dale Bennett, Darren Jiron, Bjoernar Hauge and Kylie Lamb, James Cook University, and Pamela Schultz, Envirocare for their assistance with field data collection.
Author information
Authors and Affiliations
Contributions
M.A., M.J.C., S.M.T., P.L.P. and W.F.L. conceived the idea and designed the project. M.A. and M.J.C. collected and analysed data, and wrote the manuscript. W.E. provided support in statistical analysis. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Alamgir, M., Campbell, M., Turton, S. et al. Degraded tropical rain forests possess valuable carbon storage opportunities in a complex, forested landscape. Sci Rep 6, 30012 (2016). https://doi.org/10.1038/srep30012
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep30012
This article is cited by
-
Effects of forest degradation classification on the uncertainty of aboveground carbon estimates in the Amazon
Carbon Balance and Management (2023)
-
RETRACTED ARTICLE: Experimental study on the mechanical properties of unsaturated soils and the effects of dry-wet cycles—taking coastal cities as examples
Arabian Journal of Geosciences (2021)
-
RETRACTED ARTICLE: Analysis of atmospheric climate characteristics in coastal areas and the influence of educational cognition on industrial structure changes
Arabian Journal of Geosciences (2021)
-
Water resources of Balochistan, Pakistan—a review
Arabian Journal of Geosciences (2021)
-
Forest canopy height estimation using satellite laser altimetry: a case study in the Western Ghats, India
Applied Geomatics (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.