Abstract
Changes in the chemical composition of soil organic carbon (SOC) might strongly affect the global carbon cycle as it controls the SOC decomposition rate. Vegetation change associated with long-term land use changes is known to strongly impact the chemical composition of SOC; however, data on the impacts of vegetation change following disturbance events of short durations and succession that occur frequently in forest ecosystems via diverse management objectives on SOC chemical composition are negligible. Here we examined the impacts of vegetation changes on the chemical composition of SOC by sampling soils of native broad-leaved forests, planted mixed broad-leaved and coniferous forests and tea gardens in eastern China. We used nuclear magnetic resonance spectroscopy to quantify SOC chemical composition. We found that among all components of SOC chemical composition, alkyl carbon (C) and aryl C were more liable to change with vegetation than other SOC components. Soil pH was negatively correlated to the relative abundances of alkyl C and N-alkyl C and Shannon’s index of overstory plant species was positively correlated to the relative abundances of phenolic C and aromaticity. Our results suggest that vegetation changes following short disturbance events and succession may strongly alter SOC chemical composition in forest ecosystems.
Similar content being viewed by others
Introduction
Globally, soil organic carbon (SOC) amounts to approximately 2,344 Gt, making it the largest terrestrial carbon (C) pool1,2,3. Even small changes in SOC pool decomposition might make significant changes in the atmospheric carbon (C) concentration4 and could lead to a strong positive feedback to climate change5. SOC decomposition rates are strongly associated with its chemical composition6, with decomposition rates decreasing from O-alkyl C to alkyl C7,8. Thus, shifts in SOC chemical composition affect SOC stabilization9,10.
The input of C to the soil profile is determined by C allocation, decomposition and production11. Vegetation change associated with long-term land use changes may result in SOC chemical compositional shifts12,13,14. For example, when natural forests were converted to hoop pine plantations, O-alkyl C decreased and alkyl C increased13. After native shrub lands were converted to chestnut plantations, both alkyl C and O-alkyl C decreased14. Vegetation change and the corresponding management may alter soil aggregates, which influence SOC chemical composition6. It also leads to lignin variation, which consequently impacts the yield of lignin-derived phenols and carboxyl15. Vegetation change affects lignin degradation through alterations in soil texture11 and increased yields of phenolic CuO oxidation products15. The soil texture consequently affects carbohydrates16 and O-alkyl C (mainly carbohydrate) was found be predominate in the larger fractions, whereas alkyl carbon (primarily polymethylene) dominated the clay fractions17. The influence of vegetation change and the corresponding management toward the initiation of changes in lignin and carbohydrates might be attributed to particular microbes18,19. As a result of the different growth characteristics and nutrient demands of specific microbial groups, the changes in lignin and carbohydrates may favor the growth of certain microbial groups over others20. Variations in SOC chemical composition associated with vegetation change might be attributed to the different patterns of mineralization of plant-derived microbial carbohydrate inputs15,21. However, it remains unclear whether vegetation change following disturbance events of short durations, which frequently occur naturally or via diverse management objectives in forest ecosystems, affects the chemical composition of SOC.
We sampled forest soils from native broadleaf forests, mixed evergreen broadleaved and coniferous forests and tea gardens in Fengyang Mountain nature reserve, Zhejiang Province, China, to examine the impact of vegetation change following disturbance events of short durations and succession on the chemical composition of SOC. The mixed broadleaved and coniferous forests and tea gardens were converted from native broadleaf forests during 1971–1973 and since 1975, the study forests have been protected from human disturbances. We hypothesized more similar SOC chemical compositions between native broadleaf forests and broadleaf tea gardens than those between native broadleaf forests and mixed broadleaved and coniferous forests, as plants play an important role in SOC dynamics22, mainly by means of litter quality23, which changes in the quantity and type of fresh organic matter that enters the soil1; thus, manipulating the SOC chemical composition accordingly21. We alternatively hypothesized that the conversion from native broadleaf forests to tea gardens has greater impacts on the chemical composition of SOC than that the conversion to mixed forests, as the conversion to tea garden involved complete clearance, which was a more intensive disturbance than the conversion from native forests to mixed forests via selective deforestation. We also hypothesized that overstory plant diversity, as well as soil physicochemical characteristics, may be attributable to the variation in SOC chemical composition. We addressed: (i) impacts of vegetation change from native broadleaved to mixed forests and tea garden on SOC chemical composition and (ii) associations between tree species diversity, soil physicochemical characteristics and SOC chemical composition.
Results
Vegetation change impacts on tree diversity and soil physicochemical characteristics
In this study, when evergreen broadleaved forests were converted to mixed forests and tea gardens, the Shannon index decreased significantly (Table 1). Soil physicochemical characteristics varied among vegetation types, primarily in bulk density and total phosphorus concentration. Bulk density significantly decreased from broadleaved forests to tea gardens, while total phosphorus concentration strongly decreased from broadleaved forests to mixed forests. The impacts of vegetation change on pH, total organic carbon and total nitrogen concentration were less apparent (Table 1).
Vegetation change impacts on SOC chemical composition
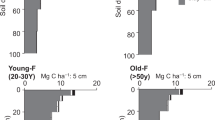
The integration of the major regions of 13C resonance revealed that alkyl C and O-alkyl C (C0−45 ppm and C45−110 ppm, respectively) were the dominant C components in all soils (Fig. 1). A multiple-response permutation procedures analysis (MRPP) showed the differences of SOC chemical composition among vegetation types with P = 0.003 (Table 2). The relative abundance of alkyl C and aryl C differed significantly among three vegetation types, with the highest relative abundance of alkyl C and lowest aryl C in mixed forests (Fig. 2, Table 3). The A/O-A also differed significantly among vegetation types, with the highest one in mixed forests, while the aromaticity in the mixed forest was lower than the other forest types (Fig. 3a,b, Table 3). However, there was no significant difference in SOC chemical composition between broadleaved forests and tea gardens. For the other components of SOC chemical composition, there were no significant differences among three vegetation types (Fig. 2, Table 3).
The NMDS ordination resulted in a final stress of 0.046 (Fig. 4). The distance between broadleaved forest and mixed forest was considerable and depicted along the NMDS axis 1 and axis 2 with P = 0.020 and P = 0.023, respectively. However, the distance between tea garden and broadleaved forest was not distinct along either the NMDS axis 1 or axis 2 (P = 0.640 and P = 0.541, separately). The chemical composition of SOC assemblages was strongly associated with all individual components, particularly along the NMDS axis 1 (Table 4). However, the chemical composition of SOC assemblages had statistically weak association with forest and soil properties. The NMDS axis 1 corresponded mainly to a gradient of increasing Shannon index and pH (P = 0.059 and P = 0.098, respectively). The NMDS axis 2 corresponded primarily to a gradient of increasing total soil phosphorus (TP) and decreasing total soil nitrogen (TN) (P = 0.085 and P = 0.104, respectively) (Fig. 5, Table 5).
Non-metric multi-dimensional scaling ordination of three vegetation types.
Site scores represent soil organic carbon chemical composition assemblages. The large symbols represent the centroids of all samples from three vegetation types and lines indicate the SE along each NMDS axis. Circles represent native broad-leaved forests; triangles represent mixed forests and squares represent tea gardens.
Non-metric multi-dimensional scaling ordination of soil organic carbon chemical composition in three vegetation types.
Black lines with long arrows indicate soil organic carbon chemical composition. Grey lines with short arrows indicate tree diversity and soil physicochemical characteristics. Shannon – Shannon index.
Influence of soil physicochemical characteristics and plant species diversity on SOC chemical composition
Soil pH was negatively correlated with alkyl C and N-alkyl C, with P = 0.043 and P = 0.039, respectively. The Shannon index was positively correlated with phenolic C and the aromaticity (P = 0.018 and 0.050, respectively). Soil physicochemical characteristics and Shannon index did not significantly correlate to the other components of chemical composition (P > 0.05) (Table 6).
Discussion
Vegetation controls the magnitude of SOC stocks as well as the composition of SOC in soils24 and is thus regarded as one of critical factors in SOC composition25. We found that the differences in the chemical composition of SOC are primarily represented by alkyl C and aryl C, which is similar to previous findings24,26,27,28. This indicated that alkyl C and aryl C were more sensitive to vegetation changes.
As we anticipated, the conversion of subtropical native broadleaved evergreen forest to other vegetation types impacted the chemical composition of SOC, where the impact differed between vegetation types. We hypothesized that the conversion of native broadleaved evergreen forests to tea garden may result in stronger alterations in SOC chemical composition. It was considered that the disturbance of soil aggregates was much more potent during the conversion to tea gardens than the conversion to mixed forests, which might significantly impact the chemical composition of SOC6. Contrary to our hypothesis, the difference of SOC chemical composition in the conversion to mixed forests was more distinct.
This may be partially attributable to the different biochemical content (e.g., lignin or carbohydrates) in the litter of pine needles in the support of its preferred microbial community, as trees can alter soil properties through root-microbe interactions13. This resulted in a distinct difference of the chemical composition of SOC in mixed forests29. Smith, et al.30 deemed that the effects of vegetation or land use changes on C content of soil is tree species-dependent. That the conversion of native broadleaved forests to tea gardens resulted in no significant difference in the chemical composition of SOC may be attributed to the less differences in the organic carbon chemical composition in leaves or roots between broadleaved trees13. Collectively, these findings indicate that the vegetation type is a critical factor that influences SOC chemical composition.
The physiochemical characteristics of the soil may favor certain microbial groups over others, thereby leading to shifts in the composition of microbial communities20,31. In this study, alkyl C was found to be significantly correlated with soil pH and this may be attributed to the microbial community composition of different pH optima32, which controlled SOC formation processes accordingly21. As we anticipated, tree species diversity contributed to SOC chemical composition. The impact of plant diversity on SOC decomposition is chiefly through influential physicochemical and biological pathways2. Briefly, plant diversity may influence the quantity and quality of organic carbon inputs from litterfall and root decomposition, as well as microclimates, to subsequently alter physicochemical characteristics of the soil6,33,34,35. Subsequently, the belowground soil microbial biomass and microbial activities are stimulated36, leading to differences in SOC composition.
Our study highlights that vegetation change is a critical factor that impacts the SOC chemical composition in forests ecosystems, where the impacts were found to be variable from native broadleaved forests to other vegetation types following disturbances of short durations and natural succession. The impact of conversion from broadleaved forests to mixed forests on the chemical composition of SOC was stronger than the conversion to tea gardens. The difference of the chemical composition of SOC were primarily represented by alkyl C and aryl C, indicating that alkyl C and aryl C were more liable to change following vegetation changes. The relative abundance of alkyl C was significantly higher and aryl C was considerably lower in mixed forests than the other vegetation types. The physicochemical characteristics of the soil, as well as tree species diversity were correlated to SOC chemical composition, indicating that they contributed to SOC chemical compositional shifts associated with the vegetation changes. Our results suggest that the conversion of native broadleaved evergreen forests to the other vegetation types following disturbances of short durations and succession may shift the equilibrium states of SOC composition to a different degree, which may potentially drive alterations in SOC and other nutrient cycling in these ecosystems.
Methods
Study area
This study was conducted at the Fengyang Mountain nature reserve, Zhejiang Province, China (119°06′ E to 119°15′E, 27°46′ N to 27°58′ N, 600 m to 1929 m a.s.l.), which comprises an area of 15,171 ha. The nature reserve is characterized as a humid subtropical climate with ~2,400 mm of annual rainfall and an average annual temperature of 12.3 °C. Prior to 1970, this area was dominated by native evergreen broadleaved forests (composed mainly of Camellia japonica Linn., Cyclobalanopsis multiervis W. C. Cheng et T. Hong, Schima superba Gardn. et Champ., Eurya japonica Thunb. and Rhododendron simsii Planch.). From 1971 to 1973, intensive selective deforestation and reforestation was conducted and portions of the forests were converted to mixed evergreen broadleaved and coniferous forests (composed primarily of Schima superba Gardn. et Champ., Pinus taiwanensis Hayata, Camellia japonica Linn. and Eurya japonica Thunb.) and tea gardens (Table 1). Additionally, some were planted as pure conifer, such as Cunninghamia lanceolata (Lamb.) Hook., Cryptomeria fortune Hooibrenk ex Otto et Dietr. and bamboo plantations at different elevation following complete clearance. Subsequent to the establishment of the nature reserve in 1975, the entire study area, including the tea gardens has been protected from anthropogenic disturbances. There has since been no fire or insect infestation disturbances recorded as yet. An overview of the main vegetation composition of this study area is presented in Supplementary Information Table S1.
Sampling
In June 2013, we randomly sampled nine native evergreen broadleaved forest stands, six mixed-forest stands and four tea garden stands. All sample stands were located on well-drained and mesic sites with slopes of less than 5% to minimize the site effects in soil characteristics37,38. In each stand, we established a sample plot of 20 × 20 m2, in which all trees >2 cm in diameter at breast height (DBH, 1.3 m above root collar) were identified and counted. In each tea garden stand, the tea bush clusters were counted as the quantity of tea bush “individuals”.
Four sampling points were randomly determined within each plot, where sampling points were at least 8 m apart. At each sampling point, we collected soil samples for nuclear magnetic resonance (NMR) spectroscopy, pH, total organic carbon concentration, total nitrogen concentration and total phosphorus concentration analysis, using a sharp knife and a trowel at the depth from 0–10, 10–20, 20–30 cm after removing litter. For bulk density soil analysis, we carefully extracted soil samples with a metal corer at four randomly allocated sampling point (diameter: 5.5 cm, height: 5 cm)39. Samples for nuclear magnetic resonance (NMR) spectroscopy analysis were stored at −80 °C in laboratory until the analysis was conducted40.
Soil physicochemical properties analysis
Soil physicochemical properties of each plot were determined by the mean value of three layers (0–10, 10–20, 20–30 cm) of the four sampling points. Soil bulk density was determined by drying the samples in an oven at 105 °C until a constant weight was achieved, which was then corrected for root and stone volume38. Soil samples for other physicochemical analyses were air-dried in the laboratory and then sieved (2 mm mesh) and stored in air-tight plastic bags. Soil pH was measured using a PB-10 pH meter (Sartorius GmbH, Göttingen, Germany) at a 1:5 ratio of soil to water41. The total organic carbon concentration was determined using the sulfuric acid-potassium external heating method42. Total nitrogen and phosphorus concentrations were determined using a Bran+Luebbe Autoanalyser 3 Continuous Flow Analyzer (Bran+Luebbe GmbH, Norderstedt, Germany) according to the manufacturer’s instructions.
Nuclear magnetic resonance (NMR) analysis
The 13C NMR spectroscopy was conducted on composite samples, which were obtained by mixing three layers and four sampling points from each sampling plot for analysis14. Similar to physicochemical analysis, the mixed samples were air-dried in the laboratory and then sieved (2 mm mesh) and stored in air-tight plastic bags in a refrigerator at 4 °C for further investigation43. To enhance the signal to noise ratio of the instrument, the hydrofluoric acid (HF) pretreatment was performed to remove Fe3+ and Mn2+ from the soil44,45. Samples were processed according to previously published methods46. Briefly, 5 g of air-dried soil samples were weighed and transferred into a 100 ml plastic centrifuge tube for the HF pretreatment, followed by the addition of 50 ml HF solution (10% v/v) into the tube. Following vibratory agitation for 1 h and centrifugation for 10 min. (3000 r min−1), the supernatant liquid was removed from the tube and the residue was continuously treated with a HF solution. These steps were repeated 8 times, but varied in the vibration time (4 × 1 h, 3 × 12 h, 1 × 24 h). Soil samples were rinsed 4 times with double distilled water to remove any residual HF that remained. Subsequently, the residues were dried in oven at 40 °C. Following all of the above processes, the residues were ground to the extent that they could pass through a 60-mesh screen and then loaded into a bag for NMR measurement. The HF-treated soil samples were subjected to 13C NMR analysis with a Bruker (Spectrospin, Rheinstetten, Germany) Avance 600 MHz NMR spectrometer. The experiments were carried out using a 7 mm CPMAS probe, at a carbon frequency of 150 MHz, MAS spinning frequency at 6 kHz, contact time of 2 ms and a recycle delay time of 5 s.
Similar to previous NMR studies40,47, the NMR spectra were divided into the following regions: alkyl C region (0–45 ppm), N-alkyl C region (45–60 ppm), carbohydrate C region (60–90 ppm), di-O-alkyl C region (90–110 ppm), aryl C region (110–145 ppm), phenolic region (145–165 ppm), carboxyl C region (165–215 ppm). The relative intensities of the different SOC fractions were obtained through the measurement and integration of the area under the curve for each region46. Additionally, two indices of organic carbon stability were calculated: the ratio of alkyl C to O-alkyl C (A/O-A) =C0–45 ppm/C45–110 ppm, which is used as an indicator of organic carbon decomposition potential48,49 and aromaticity = C110–165 ppm/C0–165 ppm, which has been used to determine the enrichment in aromatic functional groups in SOC as an indicator of the advanced stages of decomposition46,50.
Data analysis
We quantified plant species diversity by employing the Shannon’s index (H′):

where pi is the proportion of the individuals of ith overstory species in the plot based on stem counts and n is the number of overstory species in the plot51 and the index was calculated for each sampling plot. To examine the impacts of vegetation changes on the Shannon index of overstory plants, soil physicochemical characteristics and each SOC chemical composition, one-way analysis of variance (ANOVA) was conducted, followed by a LSD test (P < 0.05).
Multiple-response permutation procedures analysis (MRPP) was performed to test the difference of SOC chemical composition of among three vegetation types. The relationship between SOC chemical composition, tree diversity and soil physicochemical characteristics was analyzed by non-multidimensional scaling (NMDS). Pearson correlation tests were also applied to explore the impacts of the chemical composition of SOC, tree diversity and soil physicochemical characteristics variables on SOC chemical composition assemblages of NMDS axes. This was achieved by calculating Pearson correlation coefficients between all of the variables and NMDS axes scores. To examine the chemical compositions of SOC, tree diversity and soil physicochemical characteristics variables, which influenced the chemical composition of SOC assemblages, linear regressions test were performed. To further discover the potential impact of tree diversity and soil physicochemical characteristics on SOC chemical composition, Pearson correlations analysis was also conducted between all components of SOC chemical composition, tree species diversity and soil physicochemical characteristics. All analyses were performed in R (vegan package)52.
Additional Information
How to cite this article: Guo, X. et al. Vegetation change impacts on soil organic carbon chemical composition in subtropical forests. Sci. Rep. 6, 29607; doi: 10.1038/srep29607 (2016).
References
Fontaine, S. et al. Stability of organic carbon in deep soil layers controlled by fresh carbon supply. Nature 450, 277–280, 10.1038/nature06275 (2007).
Schmidt, M. W. I. et al. Persistence of soil organic matter as an ecosystem property. Nature 478, 49–56, 10.1038/nature10386 (2011).
Wiesmeier, M. et al. Soil organic carbon stocks in southeast Germany (Bavaria) as affected by land use, soil type and sampling depth. Global Change Biol 18, 2233–2245, 10.1111/j.1365-2486.2012.02699.x (2012).
Albaladejo, J. et al. Land use and climate change impacts on soil organic carbon stocks in semi-arid Spain. J Soil Sediment 13, 265–277, 10.1007/s11368-012-0617-7 (2012).
Schuur, E. A. G. et al. Climate change and the permafrost carbon feedback. Nature 520, 171–179, 10.1038/nature14338 (2015).
Grandy, A. S. & Neff, J. C. Molecular C dynamics downstream: The biochemical decomposition sequence and its impact on soil organic matter structure and function. Sci Total Environ 404, 297–307, 10.1016/j.scitotenv.2007.11.013 (2008).
Leifeld, J. & Kögel-Knabner, I. Soil organic matter fractions as early indicators for carbon stock changes under different land-use? Geoderma 124, 143–155, 10.1016/j.geoderma.2004.04.009 (2005).
Baldock, J. et al. Assessing the extent of decomposition of natural organic materials using solid-state 13C NMR spectroscopy. Aust J Soil Res 35, 1061–1083, 10.1071/S97004 (1997).
Li, Z., Zhao, B., Wang, Q., Cao, X. & Zhang, J. Differences in chemical composition of soil organic carbon resulting from long-term fertilization strategies. Plos One 10, e0124359, 10.1371/journal.pone.0124359 (2015).
Rabbi, S. F. et al. Characterization of soil organic matter in aggregates and size-density fractions by solid state 13C CPMAS NMR spectroscopy. Commun Soil Sci Plan 45, 1523–1537, 10.1080/00103624.2014.904335 (2014).
Jobbágy, E. G. & Jackson, R. B. The vertical distribution of soil organic carbon and its relation to climate and vegetation. Ecol Appl 10, 423–436, 10.1890/1051-0761(2000)010[0423:TVDOSO]2.0.CO;2 (2000).
Quideau, S. A., Chadwick, O. A., Benesi, A., Graham, R. C. & Anderson, M. A. A direct link between forest vegetation type and soil organic matter composition. Geoderma 104, 41–60, 10.1016/S0016-7061(01)00055-6 (2001).
Chen, C. R., Xu, Z. H. & Mathers, N. J. Soil carbon pools in adjacent natural and plantation forests of subtropical Australia. Soil Sci Soc Am J 68, 10.2136/sssaj2004.2820 (2004).
Li, Y. et al. Converting native shrub forests to Chinese chestnut plantations and subsequent intensive management affected soil C and N pools. Forest Ecol Manag 312, 161–169, 10.1016/j.foreco.2013.10.008 (2014).
Guggenberger, G., Christensen, B. T. & Zech, W. Land-use effects on the composition of organic matter in particle-size separates of soil: I. Lignin and carbohydrate signature. Eur J Soil Sci 45, 449–458, 10.1111/j.1365-2389.1994.tb00530.x (1994).
Dalal, R. C. & Henry, R. J. Cultivation effects on carbohydrate contents of soil and soil fractions. Soil Sci Soc Am J 52, 10.2136/sssaj1988.03615995005200050029x (1988).
Oades, J., Vassallo, A., Waters, A. & Wilson, M. Characterization of organic matter in particle size and density fractions from a red-brown earth by solid state 13C NMR. Soil Res 25, 71–82, 10.1071/SR9870071 (1987).
Bardgett, R. D. & van der Putten, W. H. Belowground biodiversity and ecosystem functioning. Nature 515, 505–511, 10.1038/nature13855 (2014).
Jangid, K. et al. Land-use history has a stronger impact on soil microbial community composition than aboveground vegetation and soil properties. Soil Biol Biochem 43, 2184–2193, 10.1016/j.soilbio.2011.06.022 (2011).
Brant, J. B., Myrold, D. D. & Sulzman, E. W. Root controls on soil microbial community structure in forest soils. Oecologia 148, 650–659, 10.1007/s00442-006-0402-7 (2006).
Breulmann, M., Schulz, E., Weißhuhn, K. & Buscot, F. Impact of the plant community composition on labile soil organic carbon, soil microbial activity and community structure in semi-natural grassland ecosystems of different productivity. Plant Soil 352, 253–265, 10.1007/s11104-011-0993-6 (2011).
Janssens, I. A. et al. Reduction of forest soil respiration in response to nitrogen deposition. Nature Geosci 3, 315–322, 10.1038/ngeo844 (2010).
Bird, J. A. & Torn, M. S. Fine roots vs. needles: a comparison of 13C and 15N dynamics in a ponderosa pine forest soil. Biogeochemistry 79, 361–382, 10.1007/s10533-005-5632-y (2006).
Helfrich, M., Ludwig, B., Buurman, P. & Flessa, H. Effect of land use on the composition of soil organic matter in density and aggregate fractions as revealed by solid-state 13C NMR spectroscopy. Geoderma 136, 331–341, 10.1016/j.geoderma.2006.03.048 (2006).
Chuai, X., Huang, X., Wang, W., Wu, C. & Zhao, R. Spatial simulation of land use based on terrestrial ecosystem carbon storage in coastal Jiangsu, China. Sci Rep 4, 5667, 10.1038/srep05667 (2014).
Zech, W., Haumaier, L. & Reinhold, H. In Humic Substances in Soil and Crop Sciences: Selected Readings (eds MacCarthy, P., Clapp, C. E., Malcolm, R. L. & Bloom, P. R. ) (Soil Science Society of America, 1990).
Boeni, M. et al. Organic matter composition in density fractions of Cerrado Ferralsols as revealed by CPMAS 13C NMR: Influence of pastureland, cropland and integrated crop-livestock. Agr Ecosyst Environ 190, 80–86, 10.1016/j.agee.2013.09.024 (2014).
Pérez-Cruzado, C. et al. Organic matter properties in soils afforested with Pinus radiata. Plant Soil 374, 381–398, 10.1007/s11104-013-1896-5 (2013).
Nsabimana, D., Haynes, R. J. & Wallis, F. M. Size, activity and catabolic diversity of the soil microbial biomass as affected by land use. Appl Soil Ecol 26, 81–92, 10.1016/j.apsoil.2003.12.005 (2004).
Smith, C. K., de Assis Oliveira, F., Gholz, H. L. & Baima, A. Soil carbon stocks after forest conversion to tree plantations in lowland Amazonia, Brazil. Forest Ecol Manag 164, 257–263, 10.1016/S0378-1127(01)00599-0 (2002).
Kaiser, C. et al. Belowground carbon allocation by trees drives seasonal patterns of extracellular enzyme activities by altering microbial community composition in a beech forest soil. New Phytol 187, 843–858, 10.1111/j.1469-8137.2010.03321.x (2010).
Shah, Z., Adamst, W. A. & Haven, C. D. V. Composition and activity of the microbial population in an acidic upland soil and effects of liming. Soil Biol Biochem 22, 257–263, 10.1016/0038-0717(90)90095-H (1990).
Baumann, K. et al. Changes in litter chemistry and soil lignin signature during decomposition and stabilisation of 13C labelled wheat roots in three subsoil horizons. Soil Biol Biochem 67, 55–61, 10.1016/j.soilbio.2013.07.012 (2013).
Cotrufo, M. F., Wallenstein, M. D., Boot, C. M., Denef, K. & Paul, E. The microbial efficiency-matrix stabilization (MEMS) framework integrates plant litter decomposition with soil organic matter stabilization: do labile plant inputs form stable soil organic matter? Global Change Biol 19, 988–995, 10.1111/gcb.12113 (2013).
Six, J., Conant, R. T., Paul, E. A. & Paustian, K. Stabilization mechanisms of soil organic matter: Implications for C-saturation of soils. Plant Soil 241, 155–176, 10.1023/a:1016125726789 (2002).
Lange, M. et al. Plant diversity increases soil microbial activity and soil carbon storage. Nat Commun 6, 10.1038/ncomms7707 (2015).
Hackl, E., Pfeffer, M., Donat, C., Bachmann, G. & Zechmeister-Boltenstern, S. Composition of the microbial communities in the mineral soil under different types of natural forest. Soil Biol Biochem 37, 661–671, 10.1016/j.soilbio.2004.08.023 (2005).
Chen, H. Y. H. & Shrestha, B. M. Stand age, fire and clearcutting affect soil organic carbon and aggregation of mineral soils in boreal forests. Soil Biol Biochem 50, 149–157, 10.1016/j.soilbio.2012.03.014 (2012).
Laganière, J. et al. Stability of Soil Carbon Stocks Varies with Forest Composition in the Canadian Boreal Biome. Ecosystems 16, 852–865, 10.1007/s10021-013-9658-z (2013).
Tas, N. et al. Impact of fire on active layer and permafrost microbial communities and metagenomes in an upland Alaskan boreal forest. Isme J 8, 1904–1919, 10.1038/ismej.2014.36 (2014).
Peverill, K. I., Sparrow, L. & Reuter, D. J. Soil analysis: an interpretation manual. (CSIRO publishing, 1999).
Faithfull, N. T. Methods in agricultural chemical analysis: a practical handbook. (CABI, 2002).
Zhang, T. et al. Converting paddy fields to Lei bamboo (Phyllostachys praecox) stands affected soil nutrient concentrations, labile organic carbon pools and organic carbon chemical compositions. Plant Soil 367, 249–261, 10.1007/s11104-012-1551-6 (2012).
Schmidt, M. W. I., Knicker, H., Hatcher, P. G. & Kogel-Knabner, I. Improvement of 13C and 15N CPMAS NMR spectra of bulk soils, particle size fractions and organic material by treatment with 10% hydrofluoric acid. Eur J Soil Sci 48, 319–328, 10.1111/j.1365-2389.1997.tb00552.x (1997).
Mathers, N. J., Xu, Z., Berners-Price, S. J., Senake Perera, M. C. & Saffigna, P. G. Hydrofluoric acid pre-treatment for improving 13C CPMAS NMR spectral quality of forest soils in south-east Queensland, Australia. Soil Res 40, 665–674, 10.1071/SR01073 (2002).
Berhe, A. A., Suttle, K. B., Burton, S. D. & Banfield, J. F. Contingency in the direction and mechanics of soil organic matter responses to increased rainfall. Plant Soil 358, 371–383, 10.1007/s11104-012-1156-0 (2012).
Mathers, N. J., Jalota, R. K., Dalal, R. C. & Boyd, S. E. 13C-NMR analysis of decomposing litter and fine roots in the semi-arid Mulga Lands of southern Queensland. Soil Biol Biochem 39, 993–1006, 10.1016/j.soilbio.2006.11.009 (2007).
Chavez-Vergara, B., Merino, A., Vázquez-Marrufo, G. & García-Oliva, F. Organic matter dynamics and microbial activity during decomposition of forest floor under two native neotropical oak species in a temperate deciduous forest in Mexico. Geoderma 235–236, 133–145, 10.1016/j.geoderma.2014.07.005 (2014).
Baldock, J., Preston, C., McFee, W. & Kelly, J. In Carbon forms and functions in forest soils. 89–117 (Soil Science Society of America Inc.).
Rovira, P. & Vallejo, V. R. Labile and recalcitrant pools of carbon and nitrogen in organic matter decomposing at different depths in soil: an acid hydrolysis approach. Geoderma 107, 109–141, 10.1016/S0016-7061(01)00143-4 (2002).
Shannon, C. E. A mathematical theory of communication. Bell Syst. Tech. J. 27, 379–423, 10.1002/j.1538-7305.1948.tb01338.x (1948).
Oksanen, J. et al. Vegan: Community ecology package, https://cran.r-project.org, https://github.com/vegandevs/vegan (2016).
Acknowledgements
We would like to thank Bo Zhang and Jiasen Wu for their constructive suggestions on sampling design. Financial support for this study by the National Natural Science Foundation of China (Grant No. 31470709), National Special Fund for Forestry Scientific Research in the Public Interest (Grant No. 201504406), Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD), Doctorate Fellowship Foundation of Nanjing Forestry University is gratefully acknowledged.
Author information
Authors and Affiliations
Contributions
J.Z. and X.G. designed the experiements. X.G. and M.M. conducted the experiments. All authors wrote the early version of manuscript draft. H.Y.H.C., M.M. and X.G. wrote the revised version. H.Y.H.C. and X.G. conducted all the data analysis. X.G. and M.M. prepared all figures. All authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Guo, X., Meng, M., Zhang, J. et al. Vegetation change impacts on soil organic carbon chemical composition in subtropical forests. Sci Rep 6, 29607 (2016). https://doi.org/10.1038/srep29607
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep29607
This article is cited by
-
No Evidence of an Elevation Effect Caused by Temperature Differences on Soil Microbial Properties in a Walnut Fruit Forest in Kyrgyzstan
Journal of Soil Science and Plant Nutrition (2023)
-
Effects of environmental factors and tree species mixtures on the functional groups of soil organic carbon across subtropical plantations in southern China
Plant and Soil (2022)
-
Impact of short-rotation willow as riparian land-use practice on soil organic carbon fractions and composition from two contiguous wetland systems in the prairie pothole region
Agroforestry Systems (2022)
-
Effects of land use change from natural forest to plantation on C, N and natural abundance of 13C and 15N along a climate gradient in eastern China
Scientific Reports (2019)
-
Converting natural evergreen broadleaf forests to intensively managed moso bamboo plantations affects the pool size and stability of soil organic carbon and enzyme activities
Biology and Fertility of Soils (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.