Abstract
Neurofibrillary tangles (NFTs) are the pathological hallmark of neurodegenerative diseases commonly known as tauopathies. NFTs result from the intracellular aggregation of abnormally and hyperphosphorylated tau proteins. Tau functions, which include the regulation of microtubules dynamics, are dependent on its phosphorylation status. As a consequence, any changes in tau phosphorylation can have major impacts on synaptic plasticity and memory. Recently, it has been demonstrated that AMP-activated protein kinase (AMPK) was deregulated in the brain of Alzheimer’s disease (AD) patients where it co-localized with phosphorylated tau in pre-tangle and tangle-bearing neurons. Besides, it was found that AMPK was a tau kinase in vitro. Here, we find that endogenous AMPK activation in mouse primary neurons induced an increase of tau phosphorylation at multiple sites, whereas AMPK inhibition led to a rapid decrease of tau phosphorylation. We further show that AMPK mice deficient for one of the catalytic alpha subunits displayed reduced endogenous tau phosphorylation. Finally, we found that AMPK deficiency reduced tau pathology in the PS19 mouse model of tauopathy. These results show that AMPK regulates tau phosphorylation in mouse primary neurons as well as in vivo and thus suggest that AMPK could be a key player in the development of AD pathology.
Similar content being viewed by others
Introduction
AMP-activated protein kinase (AMPK) is a master energy sensor whose activation following energy stress conditions helps maintain cellular energy levels. In this context, AMPK acts by phosphorylating key enzymes in metabolic pathways as well as transcriptional factors and cofactors. AMPK is a hetero-trimeric complex composed of a catalytic α subunit and two regulatory β and γ subunits. Several genes encode these subunits (α1, α2, β1, β2, γ1, γ2, γ3) giving rise to a multitude of complexes. The regulatory γ subunit possesses four CBS (cystathionine-beta-synthase) domains from which three of them are able to bind adenine nucleotides thereby allowing the kinase to sense and respond to AMP, ADP and ATP intracellular levels. In particular, AMP binding induces a conformational change in the α-subunit activation loop of the kinase that allows phosphorylation of the activating residue Thr172 by AMPK upstream kinases. Two main upstream kinases have been reported: LKB1 (liver kinase B1) and CaMKKβ (Ca2+/calmodulin dependent kinase kinase β). Although in the brain AMPK is mainly expressed in neurons1,2, its role in neuronal homeostasis and during neurodegeneration remains to be clearly established. In humans, it is becoming evident that AMPK is deregulated in major neurodegenerative diseases, including Huntington’s3, Parkinson’s4 and tauopathies5. It is acknowledged that the two α subunits of AMPK can have distinct roles. Indeed, it has been demonstrated that AMPKα2, which is the most highly expressed of the catalytic subunit in the brain2, was responsible for the detrimental effects observed following ischemic stroke in mice6. Whereas, AMPKα1 deregulation seems to play a key role in Huntington’s disease3.
Tauopathies - the best known of them being Alzheimer’s disease (AD) - are a subset of neurodegenerative diseases characterized by a common pathological process, aggregation of the microtubule-associated tau protein. Among these disorders, such aggregation leads to neurofibrillary tangles (NFTs), Pick bodies or argyrophilic grains. In all of these histopathological hallmarks, tau proteins are found abnormally and hyper-phosphorylated. Tau main functions include microtubules stabilization, neurite outgrowth and facilitation of organelles axonal transport including mitochondria7,8,9. These functions are regulated by tau phosphorylation status, for instance, residues Ser262 and Ser356 phosphorylation was shown to alter the microtubule binding properties of tau10. In addition tau phosphorylation is associated to changes in synaptic plasticity and memory11,12,13. As a consequence, a disequilibrium between tau kinases and phosphatases activities is believed to play a central role in the development of AD. In this context, several kinases have been reported to phosphorylate tau including glycogen synthase kinase 3 (GSK-3α and β)14,15,16, cyclin dependent kinase 5 (cdk5)16,17,18, protein kinase A (PKA)19, mitogen-activated protein kinases (MAPK)20 and the MARK family of protein kinases21,22. We and other also demonstrated that AMPK is a tau kinase in vitro5,23. In addition, activated AMPK is found accumulated in pre-tangle- and tangle-bearing neurons in AD and more largely in tauopathies. Further, expression levels of AMPK have been quantitatively shown to be elevated in AD24. The potential importance of AMPK in AD development is also supported by recent evidence. It was found that Amyloid-β (Aβ) oligomers, which are the core components of senile plaques, the other hallmark of AD, induce a rise in intracellular Ca2+ concentration thus leading to CaMKKβ activation and subsequent AMPKα1 phosphorylation23,25. In these conditions, AMPK was suggested to mediate the toxic effects of Aβ exposure on dendritic spine numbers25 and long-term potentiation (LTP)24. LTP impairments were also reported following AMPK activation mediated by AICAR (5-aminoimidazole-4-carboxamide ribonucleotide), 2-deoxy-D-glucose and metformin treatments26. However, the direct role of AMPK on tau phosphorylation in cell models remains controversial27,28 and the effect of AMPK activation mediated by metabolic stress on tau phosphorylation remains to be established. Indeed, metabolic impairments, including glucose hypometabolism and mitochondrial defects, are another early feature of AD that could participate to AMPK activation29,30,31.
The goal of our study was to determine whether AMPK is involved in tau phosphorylation in differentiated primary neurons and in vivo. To this end, we used a pharmacological approach to mimic energetic stress conditions to activate AMPK. We then evaluated the involvement of AMPK in vivo in the mouse brain by assessing endogenous tau phosphorylation levels in the brain of mice conditionally knock-out (KO) for the α2 subunit of AMPK and in AMPKα2 KO mice crossed with the PS19 tau transgenic mouse model of tauopathy.
Results
AMPK interacts with tau in neurons
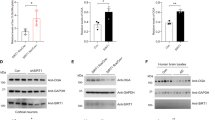
In the brain, AMPK and tau are mainly expressed in neurons therefore we used differentiated primary neurons as a model to perform our experiments. First, we determined the subcellular localization of AMPK and tau in our model. Total AMPK was localized in dendrites, cell body and nucleus (Fig. 1a), as previously reported5,32. Tau and activated AMPK (phosphorylated at Thr172) co-localized both in neurites and cell body (Fig. 1b). Primary neurons treated with the AMPK activator AICAR showed an increase of phosphorylated AMPK staining as compared to the control condition (Fig. S1a) while AMPK staining was completely abolished after incubation with an antibody blocking peptide, thus showing the specificity of the p-Thr172AMPK antibody (Fig. S1b). To further analyze the co-localization of tau with AMPK, we performed duolink proximity ligation assay (PLA) to detect in situ protein interactions using anti-tau5 (total tau) and anti-p-Thr172AMPK primary antibodies. PLA amplification occurs only if the antigens are located within 40 nm of proximity. PLA staining which is visualized as fluorescent dots was observed in cell body and neurites thereby reflecting AMPK-tau interactions (Fig. 1c). Together, these results suggest that endogenous AMPK and tau co-localize and interact in neurons supporting the notion that tau could be a neuronal target of AMPK.
AMPK co-localizes and interacts with tau in primary neurons.
(a,b) Immunofluorescence staining of total AMPK (AMPKα1/α2, green) and MAP2 (red) (a) or of activated AMPK (p-Thr172AMPK, green) and total tau (Tau5, red) (b) in primary neurons at 15 DIV. (c) PLA in primary neurons at 15 DIV using activated AMPK (p-Thr172AMPK) and total tau (Tau5) primary antibodies. PLA negative control (top panel). Green spots indicate interactions between AMPK and tau (bottom panel). Scale bars = 50 μm.
AMPK activation increases tau phosphorylation in primary neurons and affects tau binding to microtubules
We and others have previously demonstrated that AMPK could phosphorylate tau in vitro at several epitopes including Thr231, Ser262/356, Ser396, Ser409 and Ser422; whereas Ser199, Ser202, Ser235, Ser400 and Ser404 were not found to be direct AMPK substrates5,23. To study the involvement of AMPK on tau phosphorylation in a neuronal model, we used primary neuronal cultures at 15 DIV. At this differentiation time point, tau staining in Western-blot appears as two distinct bands corresponding to the two isoforms of tau containing 3 or 4 microtubules binding domains (3R and 4R isoforms). Neurons were challenged with drugs known to activate AMPK by mimicking an energy stress condition that are AICAR and metformin. AICAR is a cell-permeable nucleoside that is metabolically converted by adenosine kinase to AICA ribotide (ZMP), a purine precursor that acts as a 5′-AMP analogue whereas metformin activates indirectly AMPK through LKB1 by acting as a mild inhibitor of the respiratory chain Complex I, subsequently leading to a drop of intracellular ATP levels33.
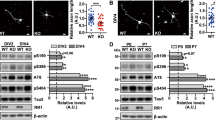
Primary neurons were treated with AICAR and metformin for up to 24 hours without inducing any toxicity (Fig. 2k and Fig. S2k). As expected, AICAR and metformin induced AMPK activation as shown by the increased phosphorylation of AMPK at Thr172 and of acetyl-CoA carboxylase (ACC) at Ser79, a direct cytosolic target of AMPK (Fig. 2a–d and Fig. S2a–d). Importantly, AMPK activation led to a significant increase of tau phosphorylation at several epitopes including Ser262/356 and Thr231, while phosphorylation at Ser202 was not changed (Fig. 2e–j and Fig. S2e–j). Similar results were obtained following metformin treatment (Fig. S2). Overall our results show that AMPK activation increased tau phosphorylation at epitopes relevant to what was previously reported in vitro.
AICAR-mediated AMPK activation induces tau phosphorylation in primary neurons.
Primary neurons at 15 DIV were treated for the indicated times with the AMPK activator AICAR (1 mM). AMPK and ACC expression and activation were monitored by Western-blot (WB) using antibodies against AMPK, p-Thr172AMPK (pAMPK), ACC, p-Ser79ACC (pACC) and actin (a). Quantifications of the ratios AMPK/actin (b) pAMPK/AMPK (c) and pACC/ACC (d). WB analysis (e) and quantification of total tau (f) and phosphorylated tau at epitopes Ser262/356 (g), Thr231 (h), Ser396/404 (i) and Ser202 (j) expressed in ratios. Cytotoxicity assay following 6 h and 24 h AICAR treatments in primary neurons at 15 DIV determined using the lactate dehydrogenase (LDH) test (k). Treatment with 0.9% Triton X-100 was used as a positive control. Results represent mean ± SD, n = 4–6. a.u., arbitrary units. *p < 0.05, **p < 0.01, ***p < 0.001 compared to Ctrl, One way ANOVA with Bonferroni’s post hoc test.
We next assessed the consequences of AMPK-mediated tau phosphorylation on tau ability to bind microtubules. To investigate the effect of AMPK activation on tau binding to microtubules, we used a microtubule fractionation assay34. Following this assay, free tau is recovered in the cytosol fraction whereas microtubule-associated tau is present in the microtubule fraction. Upon AMPK activation using AICAR or metformin, tau levels in the microtubule fraction were significantly decreased (Fig. 3) therefore suggesting that AMPK-mediated phosphorylation of tau affected its interaction with microtubules.
AMPK-mediated tau phosphorylation impairs tau affinity with microtubules.
Primary neurons were treated with AICAR (1 mM) or metformin (2.5 mM) for 2 h prior to being subjected to cell fractionation into cytosol and microtubule fractions. WB analysis on cytosol and microtubule fractions of acetylated-tubulin, tubulin, total tau and pSer262/356 tau (a). Percentage of 4R (b) and 3R (c) tau proteins observed in the microtubule fraction upon treatment with AICAR or meformin. Results represent mean ± SD, n = 5. *p < 0.05, compared to Ctrl, One way ANOVA with Bonferroni’s post hoc test.
AMPK inhibition decreases tau phosphorylation in neurons
To rule out the possibility that the increased in tau phosphorylation following AICAR treatment could be indirectly mediated by AMPK, we used several inhibitors of the main known tau kinases. Primary neurons were pre-treated with AMPK, MARK1, cdk5, MEK, PKA, mTOR and PI3K inhibitors before being subjected to AICAR treatment. Phosphorylation of ACC, the direct substrate of AMPK, was used as a control of AMPK inhibition. Except for the co-treatment with Compound C, an AMPK inhibitor, neither of the inhibitors used could reverse the increased phosphorylation of tau that was observed following AICAR treatment (Fig. 4). In addition, Compound C was able to reduce tau phosphorylation below basal levels (Fig. 4a; control condition, lane 1) suggesting that AMPK controls basal tau phosphorylation levels. Indeed, we found that Compound C led to a rapid decrease in tau phosphorylation levels (Fig. 5a–f) at the same epitopes that were found to be phosphorylated following AMPK activation. These results suggest that AMPK is one of the main tau kinase phosphorylating tau at the epitopes Ser262/356 and Thr231 at basal conditions in neurons. While after 24 h of treatment we observed a slight toxicity of Compound C, this toxicity could not be solely responsible for the decrease of tau phosphorylation since Compound C was not toxic up to 6 h of treatment (Fig. 5g) and the decreased phosphorylation of tau was already observed after 30 min of treatment.
Effect of main tau kinases inhibition on AICAR-mediated tau phosphorylation.
Primary neurons at 15 DIV were pre-treated (30 min) with various kinases inhibitors including, Compound C (Comp C, 10 μM), MARK/Par1 (20 μM), roscovitine (Rosco, 10 μM), PD98059 (PD98, 10 μM), H-89 (20 μM), Rapamycin (Rapa, 10 nM) and LY294002 (LY29, 20 μM) prior to AICAR treatment (1 mM, 2 h). Cell lysates were analyzed by WB for ACC, p-Ser79ACC (pACC), phosphorylated tau at epitopes Ser262/356 and Thr231, total tau and actin (a). Quantification of phosphorylated tau at epitopes Ser262/356 (b) and Thr231 (c) expressed in ratio of Ctrl. Results represent mean ± SD, n = 4. a.u, arbitrary units. ***p < 0.001 compared to AICAR, One way ANOVA with Bonferroni’s post hoc test.
AMPK inhibition decreases tau phosphorylation in neurons.
Primary neurons at 15 DIV were treated for the indicated times with the AMPK inhibitor Compound C (10 μM). WB analysis (a) and quantification of total tau (b) and phosphorylated tau at epitopes Ser262/356 (c), Thr231 (d), Ser396/404 (e) and Ser202 (f) expressed in ratios. Cytotoxicity was assessed after 6 h and 24 h of Compound C treatment using LDH assay (g). Results represent mean ± SD, n = 4–6. a.u., arbitrary units. *p < 0.05, **p < 0.01, ***p < 0.001 compared to Ctrl, One way ANOVA with Bonferroni’s post hoc test.
AMPKα2 deficiency reduces endogenous tau phosphorylation
To go further and to determine whether, in vivo, AMPK was also involved in tau phosphorylation, we assessed endogenous tau phosphorylation in mice that were deficient for one of the two AMPK catalytic subunits, the AMPKα2−/− mice35. Using subunits specific antibodies, we confirmed that the AMPKα2 subunit was the predominant subunit in the mouse brain, accounting for at least 65% of total AMPKα (Fig. 6a,b,d). Additionally, while there is a slight increase in AMPKα1 subunit levels, it did not compensate for the loss of the AMPKα2 subunit (Fig. 6a,c). To quantitatively assess different epitopes of tau relevant to AD, we used ultra-sensitive ELISAs36. Using these previously well characterized highly sensitive assays we observed a decrease of endogenous tau phosphorylation in the brain of the AMPKα2−/− compared to wild type mice while no significant differences were observed for the levels of total tau (Fig. 6e–h). These results suggest that AMPK can also regulate endogenous tau phosphorylation in vivo.
Endogenous levels of phosphorylated tau in AMPK α2 KO brains.
WB analysis (a) and quantification of AMPKα1/α2 (b), AMPKα1 (c) and AMPKα2 (d) expressed in ratio of actin. Results represent mean ± SEM, n = 6 (WT) and 6 (α2 KO) of 8-months old WT and AMPKα2−/− (α2 KO) mouse brains. (e–h) ELISA quantifications of endogenous total tau (e) and phosphorylated tau at epitopes Thr231 (f), Ser396/404 (g) and Ser202 (h) in heat stable fraction of 6-months old WT and AMPKα2−/− (α2 KO) mouse brains. Results represent mean ± SEM, n = 17 (WT) and 20 (α2 KO); *p < 0.05, **p < 0.01, ***p < 0.001 (Student’s t test). a.u., arbitrary units.
AMPKα2 deficiency reduces tau pathology in PS19 transgenic mice
In order to determine if AMPKα2 deficiency could impact tau pathology and tangle formation in vivo, AMPKα2−/− mice were crossed with the PS19 tau transgenic mice37. The resulting AMPKα2−/−:tauP301S mice were compared to AMPKα2+/+:tauP301S mice. Heat stable and insoluble tau fractions were obtained from 8 months old animals and analyzed by ELISA as previously described38. We did not observe any significant effect of AMPKα2 deficiency on total or phosphorylated tau levels in the heat-stable preparations (Fig. 7a–d and Fig. S3). However, we observed a significant decrease of the conformational tau epitope MC1 staining in the hippocampus, cortex and midbrain of the AMPKα2−/−:tauP301S mice as compared to AMPKα2+/+:tauP301S mice (Fig. 7e–n). This decrease of MC1 staining was accompanied by a significant reduction in insoluble total tau in the AMPKα2−/−:tauP301S mice as compared to AMPKα2+/+:tauP301S controls (Fig. 7o and Fig. S3). These results suggest that AMPK reduction could moderate tau pathology.
Impact of AMPK α2 deficiency on tau pathology.
(a–d) ELISA quantifications of total tau (a) and phosphorylated tau at epitopes Thr231 (b), Ser396/404 (c) and Ser202 (d) in heat stable fraction of 8-months old AMPKα2+/+: tauP301S (Tg) and AMPKα2−/−:tauP301S (Tg × α2 KO) mouse brains. (e–j) Immunohistochemistry of MC1 in Tg (e–g) and Tg × α2 KO (h–j) mouse brains in hippocampus (×5) (e,h), cortex (×20) (f,i) and midbrain (× 20) (g,j). Scale bars = 100 μm. (k–n) Quantifications of MC1 positive staining in dentate gyrus (DG, k) CA1 (l) cortex (m) and midbrain (n) expressed in percentage. Results represent mean ± SEM, n = 6, *p < 0.05 (Student’s t test). (o) ELISA quantifications of total tau in insoluble fraction of Tg and Tg × α2 KO mouse brains. Results represent mean ± SEM, n = 22, *p < 0.05 (Student’s t test).
Discussion
We previously demonstrated that AMPK was highly deregulated in the brain of AD and other major tauopathies patients where activated AMPK co-localized with phosphorylated tau in pre-tangle and tangle bearing neurons. An abnormal staining of AMPK was also observed in neurons not yet presenting tau hyper-phosphorylation5. In addition, in vitro studies demonstrated that AMPK was a tau kinase5,23. Together these results led us to postulate that AMPK deregulation could be an upstream event in AD and other tauopathies. In the present study, we demonstrated that AMPK was a tau kinase in primary neurons, participating in tau phosphorylation at Ser262/356 and Thr231 thereby regulating tau affinity for microtubules. In addition, we used a mouse model in which the AMPKα2 subunit was deficient to assess in vivo the impact of AMPK on tau phosphorylation and pathology. Concordant with the results we obtained in primary neurons, we found that AMPK could regulate endogenous tau phosphorylation in vivo in the mouse brain. However, we did not observe a significant decrease of tau phosphorylation in the PS19 mouse model which might be related to the presence of the P301S pathological tau mutation. Indeed, tau phosphorylation status is altered by the presence of pathological mutations39. It is interesting to note that phosphorylation of the Ser202 and Ser396/404 epitopes that were not or slightly regulated by AMPK in vitro or in primary neurons were found to be significantly decreased in vivo in the brain of AMPKα2 deficient mice. Therefore it is possible that, in vivo, AMPK could also indirectly regulate tau phosphorylation. Indeed, Thr231 phosphorylation was suggested to induce local conformational change in tau that would alter the inhibitory activity of the N-terminus of tau. As a consequence, Thr231 might serve as a priming site for GSK3β40, thereby allowing for increased tau phosphorylation at GSK3β epitopes including Ser396/404. In addition, our results show that the phosphorylation pattern of tau Thr231 is different from that of p-Thr172AMPK following AICAR treatment. These results suggest that tau Thr231 phosphorylation induced by AMPK activation might be transient. It is possible that phosphorylation at this site is also controlled by other mechanisms, including dephosphorylation. Indeed, Thr231 was described to be the target for Pin1, a peptidyl-cis-trans isomerase that was reported to favor Thr231 dephosphorylation by PP2A41,42. It is also possible that AICAR and metformin might be regulating other pathways that would eventually lead to tau dephosphorylation. It was shown for example that metformin induces tau dephosphorylation by directly activating PP2A43. In addition, a recent study showed that AMPK phosphorylation at Ser485, which is believed to be an inhibitory phosphorylation site, was correlated with tau dephosphorylation28. Therefore it is possible, that AICAR and/or metformin could activate AMPK phosphorylation at Ser485 (for AMPKα1 subunit) or Ser491 (for AMPKα2 subunit) thereby inactivating the kinase and AMPK-mediated tau phosphorylation. While this phosphorylation was reported to be necessary for AMPK inhibition, it is not sufficient44. Overall, our results could in part explain the discrepancies that were reported in the literature, by showing that AMPK-induced tau phosphorylation is transient and might be different in regard to the cell lines that are used in which these drugs might differently affect non-AMPK dependent pathways.
While our in vivo studies have only assessed the impact of AMPKα2 subunit deletion on tau phosphorylation, we cannot exclude the possibility that the AMPKα1 subunit might also be involved in tau phosphorylation and further studies using AMPKα1 KO mice are warranted. It will also be interesting to determine to which extend tau pathology is modulated in double AMPKα1/α2 KO mice. Unfortunately the double AMPKα1/α2 KO is embryonic lethal45 and neuron-specific conditional KO models will be required.
In conclusion, our present study suggests that AMPK mediates tau phosphorylation in neurons and in vivo in the mouse brain. AMPK shares at least the same properties as the main tau kinases that act in AD. Indeed, evidence suggests that AMPK participates in APP processing32,46,47, synaptic dysfunction24,25,26, neuronal death48,49 and tau phosphorylation5,23 (this study). Finally, given that glucose hypometabolism and mitochondrial defects are described to occur early in the course of the disease, it is likely that AMPK, which is directly regulated by energy status, could be an early player during the development of the pathology.
Methods
Antibodies and reagents
Antibodies directed against AMPKα1/α2, ACC, pSer79-ACC were obtained from Cell Signaling Technology. Anti-AMPK α1 and anti-AMPK α2 antibodies were from R&D Systems. Anti p-Thr172AMPK antibody and p-Thr172AMPK blocking peptide were obtained from Santa Cruz Biotechnology24. Anti-actin antibody was from BD Transduction Laboratory. Anti-MAP2, anti-α-tubulin and anti-acetylated-α-tubulin antibodies were from Sigma. Mouse monoclonal antibodies PHF1 (tau pSer396/Ser404)50, CP13 (tau pSer202)51, 2E12 or RZ3 (tau pThr231)5, DA9 (total tau aa102-140)36, DA31 (total tau aa220-240)36 and MC1 (conformation dependent antibody that recognizes only tau in a pathological conformation)52 were previously described. 12E8 monoclonal antibody was obtained from Dr. Seubert (Elan Pharmaceuticals, San Francisco, CA; tau pSer262/356)53. As total tau antibody in the Western-blot experiments, we used tau Cter (homemade well characterized antibody recognizing the 11 amino-acids in the c-terminal part of tau27)54. Tau5 (total tau) and AT180 (tau pThr231) were from Invitrogen.
AICAR, metformin, PD98059, H-89, LY294002 were purchased from Tocris; roscovitin and MARK/Par1 inhibitor from Merck; Compound C was from Santa-Cruz Biotechnology and rapamycin was from Cell Signaling Technology.
Animals
All animal experiments were performed according to procedures approved by the Feinstein Institute for Medical Research Institutional Animal Care and Use Committee. PS19 tau transgenic mice expressing P301S human mutant tau protein under the direction of the mouse prion protein promoter were obtained from The Jackson Laboratory (Bar Harbor, ME, USA)37 and were crossed with the AMPKα2 knock-out mice (AMPKα2−/−)35 to obtain AMPKα2−/−:TauP301S and AMPKα2+/+:TauP301S controls. Males and females were used at 6 or 8 months of age as indicated. Following sacrifice, brains were quickly isolated and hemi-brains were either snap-frozen or immersion-fixed in 4% paraformaldehyde (PFA) for histochemical analyses.
Tissue preparation
Brains were homogenized in an appropriate volume of homogenizing buffer, a solution of TBS (Tris Buffered Saline), pH 7.4 containing 10 mM NaF, 1 mM NaVO3, 2 mM EGTA, proteases and phosphatases inhibitors (complete Mini Roche). Protein concentrations were determined using BCA assay.
For heat stable tau, NaCl was added to a 500 μl aliquot of homogenate to a final concentration of 250 mM and β-Mercaptoethanol was added to 5% final concentration. Homogenates were mixed, heated at 100 °C for 15 minutes, mixed again by vortexing, cooled on ice for 30 minutes, vortexed again and spun down at 20,000 × g for 10 minutes at 4 °C. The supernatant, containing heat stable tau fraction was retained and used for subsequent experiments.
To obtain insoluble tau preparation38, homogenates were spun at 6,000 × g for 10 minutes at 4 °C, supernatants were collected and spun at 200,000 × g for 30 minutes at 25 °C. Pellets were resuspended in homogenizing buffer and spun again at 200,000 × g for 30 minutes at 25 °C. Finally, pellets were resuspended in an equal amount of Laemmli sample buffer and boiled for 5 minutes.
Primary neuronal cultures and treatments
Primary neurons were obtained from C57BL6 wild-type mice as described previously32. Fetuses were obtained from females that were sacrificed at 18.5 days of gestation. Forebrains were dissected in ice-cold Hank’s balanced salt solution containing 0.5% w/v D-glucose and 25 mM Hepes under a dissection microscope. Dissociation was carried out mechanically in dissection medium containing 0.01% w/v papain, 0.1% w/v dispase II and 0.01% w/v DNase and by incubation at 37 °C twice for 10 min. Cells were then spun down at 220 × g for 5 min at 4 °C, resuspended in Neurobasal medium supplemented with 2% B27, 1 mM sodium pyruvate, 100 units/ml penicillin, 100 μg/ml streptomycin, 2 mM Glutamax (Invitrogen), filtered through a 40-μm cell strainer, counted and plated on poly-L-ornithine- and laminin-coated plates at a density of about 106 cells/well. Culture medium was completely replaced after 16 h and new medium (1:3 of starting volume) was added every 3 days until the end of the culture period. Drug treatments were realized directly in the conditioned media.
Lactate dehydrogenase (LDH) release measurements
Cell toxicity was assessed by the measurement of lactate dehydrogenase release as per manufacturer’s instructions (CytoTox 96® Non-Radioactive Cytotoxicity Assay, Promega). Absorbance measurements were obtained using a SpectraMax® i3 (Molecular Devices, Sunnyvale, CA 94089, USA).
Cell fractionation into cytosolic and microtubule fractions
Cell fractionation was performed as previously described34. Briefly, primary neurons were recovered in warmed lysis buffer containing 1 mM MgCl2, 2 mM EGTA, 30% glycerol, 0.1% Triton X-100, 80 mM Pipes, pH 6.8 supplemented with proteases and phosphatases inhibitors. Homogenates were ultracentrifuged at 100,000 × g at 27 °C for 20 min, supernatants were collected as cytosolic fractions. The remaining pellets (microtubule fractions) were washed once and recovered in an equal volume to that of cytosolic fraction by sonication in RIPA buffer and homogenization at 4 °C for 1 h. Samples were mixed with loading buffer and equal volumes were loaded and analyzed by Western blotting.
Western blot (WB) analysis
Cell extracts were separated by SDS-PAGE and transferred to nitrocellulose membranes as described previously32. Membranes were probed with the antibodies listed above and analyzed by enhanced chemiluminescence detection.
Tau enzyme-linked immunosorbent assays (ELISA)
Low tau sandwich ELISA were performed as described previously36. Briefly, ninety-six-well plates were coated with the following purified monoclonal tau antibodies: DA31 (total tau), PHF1 (pSer396/404), CP13 (pSer202) and 2E12 (pThr231) at a concentration of 6 μg/ml in coating buffer (230 mM K2HPO4, 135 mM KH2PO4, 130 mM NaCl, 2 mM EDTA, 0.05% NaN3, pH 7.2) for at least 48 h at 4 °C. Plates were washed 3 times in wash buffer and blocked for 1 h at room temperature using SuperBlock (ThermoScientific). Plates were washed 5 times and 50 μl of appropriate sample, recombinant tau or PHF-tau diluted in 20% SuperBlock in 1× TBS were added to the plate. 50 μl of the total detection antibody DA9-HRP diluted 1:50 in 20% Superblock in 1× TBS was added to the samples. Plates were then incubated overnight at 4 °C. Plates were washed 9 times and 100 μl of 1-step ULTRA TMB-ELISA (ThermoScientific) was added for 30 minutes at room temperature. Finally, 100 μl of 2 M H2SO4 was added to stop the reaction and plates were read using an Infinite m200 plate reader (Tecan) at 450 nm.
Immunofluorescence
Neurons grown on poly-D-lysin and laminin-coated glass coverslips were fixed in 4% (PFA) and permeabilized using 0.25% Triton X-100 before blocking in 1% BSA for 1 h at RT. Neurons were then incubated with primary antibodies directed against total tau (Tau5, 1:200 dilution), MAP2 (1:200 dilution), total AMPKα1/α2 (1:100 dilution) and activated AMPK (p-Thr172AMPK, 1:100 dilution) O/N at 4 °C, followed with anti-IgG secondary antibodies coupled to Alexa Fluor 488 and 568 (1:500 dilution, Invitrogen). Nuclei were visualized with DAPI. Images were acquired on a Zeiss Axio Imager Z2 microscope (Carl Zeiss,Germany) equipped with a Hamamatsu ORCA-Flash4.0 digital camera (Hamamatsu Photonics, Japan) and ApoTome.2 system (Carl Zeiss,Germany). P-Thr172AMPK blocking peptide was used as a specificity control for the p-Thr172AMPK antibody. In these conditions, p-Thr172AMPK antibodies (200 μg/ml) were pre-incubated O/N at 4 °C with a five-fold excess of blocking peptide (1 mg/ml) in PBS (Phosphate Buffered Saline) before proceeding with the immunofluorescence protocol described above.
In situ Proximity ligation assay (PLA)
To detect AMPK-tau interactions in PFA-fixed primary neurons, mouse monoclonal antibodies directed against tau (Tau5, 1:200 dilution) in combination with polyclonal rabbit antibodies directed against p-Thr172AMPK (1:100 dilution) were used for in situ proximity ligation assay. PLA was performed according to the manufacturer’s protocol (Duolink® Technology). Briefly, primary antibodies were incubated as described for immunofluorescence protocol, followed by incubation with secondary antibodies conjugated to oligonucleotides strands denoted PLUS and MINUS (PLA probes Anti-Rabbit PLUS and Anti-Mouse MINUS, Sigma). The ligation and polymerization steps were conducted at 37 °C in a humid chamber followed by detection using complementary fluorescent-labeled oligonucleotides (Duolink® In situ Detection reagents Green). Negative controls for PLA were realized using the same conditions with either one or both of the primary antibodies lacking.
Immunohistochemistry
Immunohistochemistry was performed as previously described5. Briefly, paraffin-embedded brain sections were deparaffinized by immersion in toluene and hydration through graded ethanol solutions. Antigen recovery was performed by warming the slides for 30 min at 70 °C in 10 mM citrate buffer, pH 6.0. Endogenous peroxidase activity was inhibited by incubation in 3% hydrogen peroxide for 15 min at RT. After washing in TBS containing 0.05% Triton-X100 (TBS-T), sections were blocked in 5% normal goat serum, 1 mg/ml BSA, 1 mM NaF, 0.05% Triton-X100 in TBS for 1 h at RT. Sections were then incubated in the presence of MC1 primary antibodies diluted in TBS containing 1% goat serum, 1 mg/ml BSA, 1 mM NaF and 0.05% Triton-X100 overnight at 4 °C in a humidified chamber. After washing, the sections were incubated with biotin-coupled anti-mouse secondary antibodies (1:1,000 dilution) before incubation with streptavidin-horseradish peroxidase (1:1,000 dilution, Southern Biotech) and visualization with diaminobenzidine tetrahydrochloride. Staining was performed blindly on 3 slices per animal. Images were quantified using a constant threshold on Mercator software (Explora Nova) to obtain the ratio of the surface stained on the surface of the region of interest.
Statistical analysis
Group comparisons were made using one-way ANOVA with Bonferroni’s post-hoc test using GraphPad Prism (Prism 5.0d, GraphPad Software Inc, La Jolla, CA, USA). All data are reported as mean ± SD or SEM and a value of p < 0.05 was considered statistically significant.
Additional Information
How to cite this article: Domise, M. et al. AMP-activated protein kinase modulates tau phosphorylation and tau pathology in vivo. Sci. Rep. 6, 26758; doi: 10.1038/srep26758 (2016).
References
Culmsee, C., Monnig, J., Kemp, B. E. & Mattson, M. P. AMP-activated protein kinase is highly expressed in neurons in the developing rat brain and promotes neuronal survival following glucose deprivation. J Mol Neurosci 17, 45–58, doi: 10.1385/JMN:17:1:45 (2001).
Turnley, A. M. et al. Cellular distribution and developmental expression of AMP-activated protein kinase isoforms in mouse central nervous system. J Neurochem 72, 1707–1716 (1999).
Ju, T. C. et al. Nuclear translocation of AMPK-alpha1 potentiates striatal neurodegeneration in Huntington’s disease. J Cell Biol 194, 209–227, doi: 10.1083/jcb.201105010 (2011).
Jiang, P. et al. Adenosine monophosphate-activated protein kinase overactivation leads to accumulation of alpha-synuclein oligomers and decrease of neurites. Neurobiol Aging 34, 1504–1515, doi: 10.1016/j.neurobiolaging.2012.11.001 (2013).
Vingtdeux, V., Davies, P., Dickson, D. W. & Marambaud, P. AMPK is abnormally activated in tangle- and pre-tangle-bearing neurons in Alzheimer’s disease and other tauopathies. Acta Neuropathol 121, 337–349, doi: 10.1007/s00401-010-0759-x (2011).
Li, J., Zeng, Z., Viollet, B., Ronnett, G. V. & McCullough, L. D. Neuroprotective effects of adenosine monophosphate-activated protein kinase inhibition and gene deletion in stroke. Stroke 38, 2992–2999, doi: 10.1161/STROKEAHA.107.490904 (2007).
Caceres, A. & Kosik, K. S. Inhibition of neurite polarity by tau antisense oligonucleotides in primary cerebellar neurons. Nature 343, 461–463, doi: 10.1038/343461a0 (1990).
Sato-Harada, R., Okabe, S., Umeyama, T., Kanai, Y. & Hirokawa, N. Microtubule-associated proteins regulate microtubule function as the track for intracellular membrane organelle transports. Cell Struct Funct 21, 283–295 (1996).
Reddy, P. H. Abnormal tau, mitochondrial dysfunction, impaired axonal transport of mitochondria and synaptic deprivation in Alzheimer’s disease. Brain Res 1415, 136–148, doi: 10.1016/j.brainres.2011.07.052 (2011).
Fischer, D. et al. Conformational changes specific for pseudophosphorylation at serine 262 selectively impair binding of tau to microtubules. Biochemistry 48, 10047–10055, doi: 10.1021/bi901090m (2009).
Schindowski, K. et al. Alzheimer’s disease-like tau neuropathology leads to memory deficits and loss of functional synapses in a novel mutated tau transgenic mouse without any motor deficits. Am J Pathol 169, 599–616, doi: 10.2353/ajpath.2006.060002 (2006).
Regan, P. et al. Tau phosphorylation at serine 396 residue is required for hippocampal LTD. J Neurosci 35, 4804–4812, doi: 10.1523/JNEUROSCI.2842-14.2015 (2015).
Ahmed, T. et al. Rescue of impaired late-phase long-term depression in a tau transgenic mouse model. Neurobiol Aging 36, 730–739, doi: 10.1016/j.neurobiolaging.2014.09.015 (2015).
Lovestone, S. et al. Alzheimer’s disease-like phosphorylation of the microtubule-associated protein tau by glycogen synthase kinase-3 in transfected mammalian cells. Curr Biol 4, 1077–1086 (1994).
Godemann, R., Biernat, J., Mandelkow, E. & Mandelkow, E. M. Phosphorylation of tau protein by recombinant GSK-3beta: pronounced phosphorylation at select Ser/Thr-Pro motifs but no phosphorylation at Ser262 in the repeat domain. FEBS Lett 454, 157–164 (1999).
Bretteville, A. et al. Hypothermia-induced hyperphosphorylation: a new model to study tau kinase inhibitors. Sci Rep 2, 480, doi: 10.1038/srep00480 (2012).
Baumann, K., Mandelkow, E. M., Biernat, J., Piwnica-Worms, H. & Mandelkow, E. Abnormal Alzheimer-like phosphorylation of tau-protein by cyclin-dependent kinases cdk2 and cdk5. FEBS Lett 336, 417–424 (1993).
Patrick, G. N. et al. Conversion of p35 to p25 deregulates Cdk5 activity and promotes neurodegeneration. Nature 402, 615–622, doi: 10.1038/45159 (1999).
Jicha, G. A. et al. cAMP-dependent protein kinase phosphorylations on tau in Alzheimer’s disease. J Neurosci 19, 7486–7494 (1999).
Drewes, G. et al. Mitogen activated protein (MAP) kinase transforms tau protein into an Alzheimer-like state. EMBO J 11, 2131–2138 (1992).
Drewes, G., Ebneth, A., Preuss, U., Mandelkow, E. M. & Mandelkow, E. MARK, a novel family of protein kinases that phosphorylate microtubule-associated proteins and trigger microtubule disruption. Cell 89, 297–308 (1997).
Yoshida, H. & Goedert, M. Phosphorylation of microtubule-associated protein tau by AMPK-related kinases. J Neurochem 120, 165–176, doi: 10.1111/j.1471-4159.2011.07523.x (2012).
Thornton, C., Bright, N. J., Sastre, M., Muckett, P. J. & Carling, D. AMP-activated protein kinase (AMPK) is a tau kinase, activated in response to amyloid beta-peptide exposure. Biochem J 434, 503–512, doi: 10.1042/BJ20101485 (2011).
Ma, T. et al. Inhibition of AMP-activated protein kinase signaling alleviates impairments in hippocampal synaptic plasticity induced by amyloid beta. J Neurosci 34, 12230–12238, doi: 10.1523/JNEUROSCI.1694-14.2014 (2014).
Mairet-Coello, G. et al. The CAMKK2-AMPK kinase pathway mediates the synaptotoxic effects of Abeta oligomers through Tau phosphorylation. Neuron 78, 94–108, doi: 10.1016/j.neuron.2013.02.003 (2013).
Potter, W. B. et al. Metabolic regulation of neuronal plasticity by the energy sensor AMPK. PLoS One 5, e8996, doi: 10.1371/journal.pone.0008996 (2010).
Greco, S. J., Sarkar, S., Johnston, J. M. & Tezapsidis, N. Leptin regulates tau phosphorylation and amyloid through AMPK in neuronal cells. Biochem Biophys Res Commun 380, 98–104, doi: 10.1016/j.bbrc.2009.01.041 (2009).
Kim, B., Figueroa-Romero, C., Pacut, C., Backus, C. & Feldman, E. L. Insulin resistance prevents AMPK-induced tau dephosphorylation through Akt-mediated increase in AMPKSer485 phosphorylation. J Biol Chem, doi: 10.1074/jbc.M115.636852 (2015).
Mosconi, L. Brain glucose metabolism in the early and specific diagnosis of Alzheimer’s disease. FDG-PET studies in MCI and AD. Eur J Nucl Med Mol Imaging 32, 486–510, doi: 10.1007/s00259-005-1762-7 (2005).
Ferreira, I. L., Resende, R., Ferreiro, E., Rego, A. C. & Pereira, C. F. Multiple defects in energy metabolism in Alzheimer’s disease. Curr Drug Targets 11, 1193–1206 (2010).
Cabezas-Opazo, F. A. et al. Mitochondrial Dysfunction Contributes to the Pathogenesis of Alzheimer’s Disease. Oxid Med Cell Longev 2015, 509654, doi: 10.1155/2015/509654 (2015).
Vingtdeux, V. et al. AMP-activated protein kinase signaling activation by resveratrol modulates amyloid-beta peptide metabolism. J Biol Chem 285, 9100–9113, doi: 10.1074/jbc.M109.060061 (2010).
Hardie, D. G. Neither LKB1 nor AMPK are the direct targets of metformin. Gastroenterology 131, 973; author reply 974–975, doi: 10.1053/j.gastro.2006.07.032 (2006).
Hamdane, M. et al. Mitotic-like tau phosphorylation by p25-Cdk5 kinase complex. J Biol Chem 278, 34026–34034, doi: 10.1074/jbc.M302872200 (2003).
Viollet, B. et al. The AMP-activated protein kinase alpha2 catalytic subunit controls whole-body insulin sensitivity. J Clin Invest 111, 91–98, doi: 10.1172/JCI16567 (2003).
Acker, C. M., Forest, S. K., Zinkowski, R., Davies, P. & d’Abramo, C. Sensitive quantitative assays for tau and phospho-tau in transgenic mouse models. Neurobiol Aging 34, 338–350, doi: 10.1016/j.neurobiolaging.2012.05.010 (2013).
Yoshiyama, Y. et al. Synapse loss and microglial activation precede tangles in a P301S tauopathy mouse model. Neuron 53, 337–351, doi: 10.1016/j.neuron.2007.01.010 (2007).
Forest, S. K., Acker, C. M., d’Abramo, C. & Davies, P. Methods for measuring tau pathology in transgenic mouse models. J Alzheimers Dis 33, 463–471, doi: 10.3233/JAD-2012-121354 (2013).
Bretteville, A. et al. Two-dimensional electrophoresis of tau mutants reveals specific phosphorylation pattern likely linked to early tau conformational changes. PLoS One 4, e4843, doi: 10.1371/journal.pone.0004843 (2009).
Lin, Y. T. et al. The binding and phosphorylation of Thr231 is critical for Tau’s hyperphosphorylation and functional regulation by glycogen synthase kinase 3beta. J Neurochem 103, 802–813, doi: 10.1111/j.1471-4159.2007.04792.x (2007).
Galas, M. C. et al. The peptidylprolyl cis/trans-isomerase Pin1 modulates stress-induced dephosphorylation of Tau in neurons. Implication in a pathological mechanism related to Alzheimer disease. J Biol Chem 281, 19296–19304, doi: 10.1074/jbc.M601849200 (2006).
Hamdane, M. et al. Pin1 allows for differential Tau dephosphorylation in neuronal cells. Mol Cell Neurosci 32, 155–160, doi: 10.1016/j.mcn.2006.03.006 (2006).
Kickstein, E. et al. Biguanide metformin acts on tau phosphorylation via mTOR/protein phosphatase 2 A (PP2A) signaling. Proc Natl Acad Sci USA 107, 21830–21835, doi: 10.1073/pnas.0912793107 (2010).
Hurley, R. L. et al. Regulation of AMP-activated protein kinase by multisite phosphorylation in response to agents that elevate cellular cAMP. J Biol Chem 281, 36662–36672, doi: 10.1074/jbc.M606676200 (2006).
Viollet, B. et al. Physiological role of AMP-activated protein kinase (AMPK): insights from knockout mouse models. Biochem Soc Trans 31, 216–219, doi: 10.1042/ (2003).
Won, J. S., Im, Y. B., Kim, J., Singh, A. K. & Singh, I. Involvement of AMP-activated-protein-kinase (AMPK) in neuronal amyloidogenesis. Biochem Biophys Res Commun 399, 487–491, doi: 10.1016/j.bbrc.2010.07.081 (2010).
Yang, T. T., Shih, Y. S., Chen, Y. W., Kuo, Y. M. & Lee, C. W. Glucose regulates amyloid beta production via AMPK. J Neural Transm, doi: 10.1007/s00702-015-1413-5 (2015).
Weisova, P. et al. Role of 5′-adenosine monophosphate-activated protein kinase in cell survival and death responses in neurons. Antioxid Redox Signal 14, 1863–1876, doi: 10.1089/ars.2010.3544 (2011).
Manwani, B. & McCullough, L. D. Function of the master energy regulator adenosine monophosphate-activated protein kinase in stroke. J Neurosci Res 91, 1018–1029, doi: 10.1002/jnr.23207 (2013).
Greenberg, S. G., Davies, P., Schein, J. D. & Binder, L. I. Hydrofluoric acid-treated tau PHF proteins display the same biochemical properties as normal tau. J Biol Chem 267, 564–569 (1992).
Duff, K. et al. Characterization of pathology in transgenic mice over-expressing human genomic and cDNA tau transgenes. Neurobiol Dis 7, 87–98, doi: 10.1006/nbdi.1999.0279 (2000).
Jicha, G. A., Bowser, R., Kazam, I. G. & Davies, P. Alz-50 and MC-1, a new monoclonal antibody raised to paired helical filaments, recognize conformational epitopes on recombinant tau. J Neurosci Res 48, 128–132 (1997).
Seubert, P. et al. Detection of phosphorylated Ser262 in fetal tau, adult tau and paired helical filament tau. J Biol Chem 270, 18917–18922 (1995).
Sergeant, N. et al. Dysregulation of human brain microtubule-associated tau mRNA maturation in myotonic dystrophy type 1. Hum Mol Genet 10, 2143–2155 (2001).
Acknowledgements
We thank Dr. Peter Seubert (Elan Pharmaceuticals, San Francisco, CA) for providing the 12E8 antibody. We are grateful to Raphaëlle Caillierez for her technical assistance. This work was supported by the French Fondation pour la cooperation Scientifique – Plan Alzheimer 2008–2012 (Senior Innovative Grant 2013) to VV, by the National Institutes of Health Grant R01AG042508 to PM and in part through the Labex DISTALZ (Development of Innovative Strategies for a Transdisciplinary Approach to Alzheimer’s Disease). MD holds a doctoral scholarship from Lille 2 University.
Author information
Authors and Affiliations
Contributions
P.M. and V.V. designed research; M.D., S.D., C.M., H.Z., P.C. and V.V. performed research; L.B., B.V. and P.D. contributed unpublished reagents/analytic tools; P.M. and V.V. analyzed data; P.M. and V.V. wrote the paper. All authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Domise, M., Didier, S., Marinangeli, C. et al. AMP-activated protein kinase modulates tau phosphorylation and tau pathology in vivo. Sci Rep 6, 26758 (2016). https://doi.org/10.1038/srep26758
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep26758
This article is cited by
-
Palmitic Acid Induces Posttranslational Modifications of Tau Protein in Alzheimer’s Disease–Related Epitopes and Increases Intraneuronal Tau Levels
Molecular Neurobiology (2024)
-
Cyanidin-3-O-glucoside protects the brain and improves cognitive function in APPswe/PS1ΔE9 transgenic mice model
Journal of Neuroinflammation (2023)
-
Role of Tau in Various Tauopathies, Treatment Approaches, and Emerging Role of Nanotechnology in Neurodegenerative Disorders
Molecular Neurobiology (2023)
-
Aβ42 oligomers trigger synaptic loss through CAMKK2-AMPK-dependent effectors coordinating mitochondrial fission and mitophagy
Nature Communications (2022)
-
Equilibrative nucleoside transporter 1 inhibition rescues energy dysfunction and pathology in a model of tauopathy
Acta Neuropathologica Communications (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.