Abstract
CRISPR/Cas9 has been widely used in generating site-specific genetically modified animal models. Myostatin (MSTN) is a negative regulator of muscle mass, related to muscle growth and differentiation. The knockout of MSTN with the desired phenotype of double muscle has been successfully generated in mice, goats, pigs and cattle, but not in rabbits. In this study, the MSTN knockout (KO) rabbits were generated by co-injection of Cas9 mRNA and sgRNA into zygotes. The typical phenotype of double muscle with hyperplasia or hypertrophy of muscle fiber was observed in MSTN KO rabbits. Furthermore, a similar phenotype was found in the F1 generation, suggesting that the mutation of MSTN could be stably inherited in the MSTN KO rabbits. In summary, we have successfully generated MSTN KO rabbits using CRISPR/Cas9 system with high efficiency, which is a reliable and effective animal model for the study of muscle development and related diseases.
Similar content being viewed by others
Introduction
Gene editing technologies have been developed to study the function of genes, generate genetically modified animal models for biomedical research and improve animal traits in agriculture. CRISPR/Cas9 system consists of a single guide RNA (sgRNA) and a human codon-optimized Cas9 nuclease, which can induce targeted mutations by nonhomologous end joining (NHEJ). Co-injection of Cas9 mRNA and sgRNA into zygotes has been used as an efficient tool to generate gene-targeted animal models in mice1, sheep2, monkeys3 and pigs4.
Rabbits are a promising animal model for biomedical research, as they show more similarities to human beings in terms of physiology and anatomy than mice and rats and require low cost maintenance and short pregnancy period compared to pigs and monkeys5. Currently, rabbits have been extensively used as a more appropriate animal model for studying cardiovascular/metabolic and ophthalmic diseases6,7.
Myostatin (MSTN) is a member of the transforming growth factor beta (TGF-β) superfamily, which acts as a negative regulator of muscle growth8,9. It has been reported that spontaneous mutations of MSTN in cattle10 and sheep11 causes muscle hypertrophy. A double-muscled phenotype with the characteristics of increased muscle mass was also obtained in MSTN KO sheep2, pigs12 and dogs13, which encourages us to generate MSTN KO rabbits for the study of muscle development and improvement of animal traits for agriculture in the future.
Here, in order to generate MSTN KO rabbits, the in vitro transcribed mRNA encoding for Cas9 and sgRNA targeting the MSTN gene, was microinjected into the cytoplasm of rabbit pronuclear-stage embryos. We demonstrated the high efficiency of CRISPR/Cas9 system-mediated gene editing and the desired phenotype of double muscle was obtained in the MSTN KO rabbits.
Results
CRISPR/Cas9-mediated MSTN KO in rabbit zygotes
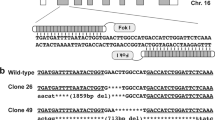
In order to disrupt the function of MSTN in rabbits, two sgRNAs targeting the CDS of the rabbit MSTN gene were designed using the CRISPR/Cas9 online design tool (http://tools.genomeengineering.org) (Fig. 1A). To determine the efficiency of CRISPR/Cas9 system in zygotes, the Cas9 mRNA and sgRNAs were microinjected into rabbit zygotes and cultured until the blastocyst stage. Then 12 blastocysts were harvested and subjected to PCR amplification and T7E1 cleavage assay. As shown in Fig. 1B,C, the mutation of MSTN was found in 10 tested blastocysts (83.3%), in which 3 blastocysts (25%) displayed biallelic mutations, suggesting that the two sgRNAs-directed CRISPR/Cas9 system was an efficient tool for disrupting the rabbit MSTN gene in zygotes.
CRISPR/Cas9-mediated gene knockout of MSTN in zygote.
(A) Schematic diagram of sgRNA targeting the rabbit MSTN gene loci. Two sgRNA sequences, sgRNA1 and sgRNA2, are marked in red and the protospacer adjacent moti (PAM) sequences are presented in green. (B) Mutation detection in blastocyst by T-cloning and Sanger sequencing. The WT sequence is shown at the top of the targeting sequence. E: embryos; WT: wild type; deletions “−”; insertion “+”. (C) Mutation detection in blastocyst by T7E1 cleavage assay. M, DL2000; 1–12 represent different blastocysts used in this study.
Generation of MSTN KO rabbits by CRISPR/Cas9 system
To generate MSTN KO rabbits, a total of 158 injected zygotes were transferred into the oviducts of 4 surrogate rabbits. After a full-term gestation, the 4 surrogate mothers gave birth to 20 live pups successfully (Table 1). The T-cloning and PCR-sequence results showed that the MSTN mutation was detected in 16 pups and the indels ranged from 3 bp to 76 bp (Fig. 2A), which was also confirmed by T7E1 assay (Fig. 2B,C). In addition, fragment deletions between the two sgRNAs targeting sites were frequently observed in this study (81.3%). Furthermore, the typical phenotype of double muscle was observed in F0 MSTN KO rabbits at 4 months of age, which compared with their wild type (WT) counterparts (Fig. 2D).
Generation of MSTN KO rabbit via zygote injection.
(A) T-cloning and Sanger sequencing of 20 pups. F01-1–F01–9 represent the F0 pups from the first time of microinjection. F02-1–F02–11 represent the F0 pups from the second microinjection. The sequences of sgRNA are shown in red and the PAM sequences are presented in green. The length of deletions is noted to the right of each sequence (- deletion). WT: wild type; deletions “−”; insertion “+”. (B) T7E1 cleavage assay for the mutation detection of F01-1–F01–9. M, DL2000; 1–9 represent the pups used in this study. (C) T7E1 cleavage assay for the mutation detection of F02-1–F02–11. M, DL2000; 1–11 represent the pups used in this study. (D) Photos of MSTN−/− (F02–3), MSTN+/− (F02–6) and WT (F02–11) rabbits (4-month old) in F0. Note that an obvious double muscular phenotype was found in MSTN−/− and MSTN+/− rabbits, which compared to the WT.
Off-target effect is a major concern of the CRISPR/Cas9 system. To test whether off-target effect occurred in these genetically modified rabbits, a total of 5 potential off-target sites (POTS) for each sgRNA were predicted by the CRISPR design online tool. All POTS were PCR amplified and subjected to T7E1 cleavage and Sanger sequence. The results showed that no mutation was detected in these POTS, indicating that the Cas9/sgRNA system did not induce undesirable off-target effect in the MSTN KO rabbits (Supplementary Table S2 and Supplementary Fig. S1).
Heritability of the MSTN KO rabbits
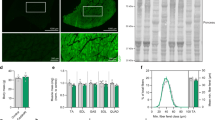
To determine whether the MSTN KO could be stably transmitted to the offspring, the female founder F01–7 was mated with male rabbit F01–5. T-cloning sequence analysis and T7E1 cleavage assay demonstrated that 7 out of 9 newborn F1 rabbits carried MSTN mutations. In these pups, the F12, F15, F16, F18, F19 were monoallelic, while F13, F17 were biallelic MSTN KO rabbits (Fig. 3A,B). To investigate whether gene mutations abolished the MSTN protein translation, western blot analysis was carried out using the protein extracted from gluteus maximus tissue of MSTN−/−, MSTN+/− and WT rabbits. As shown in Fig. 3C, the MSTN protein was significantly decreased in MSTN−/− and MSTN+/− rabbits, compared to their WT counterparts.
Heritability of the MSTN KO rabbit.
(A) T-cloning and Sanger sequencing analyses of MSTN KO rabbits in F1. F11–F19 represent the F1 MSTN KO rabbits. The sequences of sgRNA are shown in red and the PAM sequences are presented in green. WT: wild type; deletions “−”. (B) The mutation detection of F1 rabbits by T7E1 cleavage assay. M, DL2000; F11–F19 represent the offspring pups used in this study. (C) Determination of MSTN protein in skeletal muscle by Western blot. Equal amounts of protein were used and the β-actin was used as reference control.
Increased body weight and muscle mass in MSTN KO rabbits
In order to investigate the differences in muscle development in MSTN KO rabbits, the body weight of MSTN+/− and WT groups (n = 4) were recorded weekly. As shown in Fig. 4A, the body weight was not significantly different between the two groups during the first 6 weeks. However, the MSTN+/− rabbits were obviously heavier than WT rabbits after 6 weeks of age. In addition, the typical double-muscled phenotype was also found in MSTN+/− rabbits at 2 months of age (Fig. 4B).
Increased body weight and muscle mass in MSTN KO rabbits.
(A) The average body weight of MSTN+/− and WT rabbits from F1 (n = 4). (B) Photos of MSTN+/− (F1 6) and WT (F1 1) rabbits (2-month old) in F1. (C) The average weight of heart (H), tongue (T), gluteus maximus (G) and vastus lateralis (V) from MSTN+/− and WT rabbits (n = 4). Data are expressed as mean ± SEM. *p < 0.05, **p < 0.01, ***p < 0.001, Student’s t test. (D) Representative of heart (H), tongue (T), gluteus maximus (G) and vastus lateralis (V) from MSTN+/− and WT rabbits.
To determine if the increase in body weight was caused by enlarged muscle mass in MSTN KO rabbits, the average weight of heart, tongue, gluteus maximus and vastus lateralis from MSTN+/− and WT groups was determined at 2 months of age. As shown in Fig. 4C,D, no abnormal development was observed, while the average weight of individual muscles was dramatically increased in the MSTN KO rabbits, which compared to the WT counterparts.
Hyperplasia and/or hypertrophy of muscle fibers in MSTN KO rabbits
To determine whether the increase in muscle mass is due to hyperplasia and/or hypertrophy of muscle fibers, histological analysis was performed in the tongue and gluteus maximus from WT and MSTN KO rabbits (Fig. 5A). The results showed that the average size of tongue myofibers in MSTN+/− rabbits (1539.20 ± 132.52 μm2, p < 0.05) was substantially larger than in WT rabbits (1055.81 ± 257.57 μm2) (Fig. 5B). Additionally, the average myofiber size in gluteus maximus was significantly increased in the MSTN+/− (2011.01 ± 169.79 um2, p < 0.05) rabbits, compared to the WT rabbits (1318.51 ± 261.48um2). Furthermore, the average fiber density in gluteus maximus from the MSTN+/− rabbits (327.79 ± 21.12, p < 0.001) was significantly higher than in the WT rabbits (211.37 ± 19.89) (Fig. 5B).
A hyperplasia and/or hypertrophy of muscle fibers was observed in MSTN KO rabbits.
(A) H&E staining of the muscle fibers from tongue and gluteus maximus. (a) Cross and longitudinal section in tongue; (b) Cross section in gluteus maximus. (B) The statistical analysis of the fiber size and number in tongue and gluteus maximus from MSTN+/− and WT rabbits (2-month old). Average fiber size and number in tongue (a,b); and in gluteus maximus (c,d). Data are expressed as mean ± SEM. *p < 0.05, **p < 0.01, ***p < 0.001, Student’s t test.
These results therefore suggest that the enlarged tongue in the MSTN KO rabbits is due to fiber hypertrophy. Whereas, it seems that the increased muscle mass of gluteus maximus in the MSTN KO rabbits is due to both fiber hyperplasia and hypertrophy.
Discussion
In recent years, with the aim to study muscle development and increase meat production, MSTN has been modified in various species such as mice14, goats15, sheep2 and pigs12. In this study, we successfully generated MSTN KO rabbits by microinjection of Cas9/sgRNA into one-cell stage embryos, demonstrating that this system could be used to efficiently generate gene knockout rabbits. Moreover, fragment deletions were frequently detected between the two sgRNAs targeting sites in MSTN KO rabbits, suggesting that the dual sgRNA-directed CRISPR/Cas9 system could provide an efficient tool for gene knockout in the mammal genome16.
Off-target mutation has been frequently reported in Cas9-mediated gene editing system17,18,19, however, no off-target effect was detected in the MSTN KO rabbits. We believe that it might be due to the low concentration of Cas9/sgRNA, which degraded immediately after targeting the aimed gene. In addition, the strict match of seed sequences (8–12 bases close to PAM) is a critical factor for ensuring the site-specific cleavage of CRISPR/Cas9 system20. Furthermore, to avoid the off-target effect, two modified forms of Cas9, namely, D10ACas921 and FokI-dCas9 (fCas9)22 can be used for gene editing in the future.
Although the increased muscle mass makes it attractive to create MSTN KO livestock, the calving difficulty induced by enlarged body size of fetus is a big problem in these MSTN-mutant animals23. It has been demonstrated that the MSTN-deficient animals exhibit disorders similar to large offspring syndrome (LOS)8,24. In our study, although the MSTN KO rabbits showed a typical double-muscled phenotype and a dramatically increased body weight, there was no significantly difference in body size and weight at birth compared to the WT controls. In addition, the MSTN KO rabbits appears normal, healthy and without reproduction problems, indicating that MSTN KO rabbits generated by CRISPR/Cas9 system are suitable for the study of muscle development and related diseases.
Animals with MSTN mutations are characterized by increased muscle mass, which results from a combination of increased muscle fiber size and number8. Previous studies have demonstrated that the enlarged muscle is attributed to muscle fiber hyperplasia rather than hypertrophy in MSTN−/− pigs12 and Belgian Blue cattle25. While both fiber hypertrophy and hyperplasia led to the increased muscle mass in MSTN KO mice9. In our study, both enlarged fiber size and increased fiber cell number were found in gluteus maximus, while the fiber hypertrophy was mainly responsible for the enlarged tongue in MSTN+/− rabbits. Therefore we believe that the increased muscle phenotype in the MSTN KO rabbits is attributed to hyperplasia and/or hypertrophy of muscle fibers. Besides, the MSTN-regulated muscle proliferation and differentiation are also affected by expression levels of MSTN mRNA, the health state and the individual developmental stage of the organism.
In conclusion, the MSTN KO rabbits were efficiently generated by microinjection Cas9/sgRNA mixture into the pronuclear-stage embryos and the typical double-muscled phenotype was obtained in the founder and offspring MSTN KO rabbits. These MSTN KO rabbits can be a promising tool for studying muscle development and improving economically important traits in livestock.
Materials and Methods
Ethics statement
All experiments involving animals were conducted according to the guidelines for the animal care and use of laboratory animals established by the Animal Care Center and Use Committee of Jilin University. All experimental protocols were approved by the Ethics Committee of Jilin University. New Zealand rabbits were housed under standard laboratory conditions of a 12-h light and 12-h dark cycle in individual cages and were fed twice a day with commercial rabbit basic diet (Animal center of Jilin University) and water ad libitum. The sexually mature rabbits were used for embryo collection and embryo transfer recipients.
Vector construction and in vitro transcription
The 3× FLAG-NLS-SpCas9-NLS vector (Addgene ID 48137) was linearized with NotI and in vitro transcribed to mRNA using the mMessage mMachine SP6 Kit (Ambion). Then the product was purified with the RNeasy Mini Kit (Qiagen) according to the manufacturer’s instructions.
To prepare the vector of in vitro transcription of sgRNA, the pair of complementary 20-nt DNA oligos were annealed to generate a double-stranded DNA fragment and then sub-cloned into the BbsI-linearized pUC57-T7-gRNA vector (Addgene ID 51306). The sequence of two sgRNAs is shown in Supplementary Table S1. The PCR products for in vitro transcription of sgRNA were amplified using T7 primers (T7-F: 5′-GAAATTAATACGACTCACTATA-3′ and T7-R: 5′-AAAAAAAGCACCGA CTCGGTGCCAC-3′). Then in vitro transcription was performed using T7 RNA Synthesis Kit (Ambion) and the synthesized mRNA was purified using miRNeasy Mini Kit (Qiagen) according to the manufacturer’s instructions. The concentration and quality of the synthesized mRNA were determined by Nandrop 2000 and agarose gel electrophoresis, respectively.
Zygote injection with Cas9/sgRNA
Zygotes were collected through surgical oviduct flushing from donors after superovulation treatment and natural mating, as described previously5. Rabbit embryos at the pronuclear stage (around 18–20 h post-mated) were transferred into oocyte manipulation medium, containing 9.5 g TCM-199, 0.05 g NaHCO3 (Sigma, S4019), 0.75 g Hepes (Sigma, H3784), 0.05 g penicillin, 0.06 g streptomycin, 1.755 g NaCl, 3.0 g BSA and 1 L Milli Q H2O. A mixture of in vitro transcribed sgRNA (40 ng/μL) and Cas9 mRNA (180 ng/μL) was injected into the cytoplasm of pronuclear stage embryos. The injected embryos were transferred to embryo culture medium for 30–60 min culture at 38.5 °C, 5% carbon dioxide and humidity conditions. Then approximately 30–50 injected embryos were transferred into the oviduct of the recipient mother.
Genotyping of MSTN mutation in embryos and pups
To test the mutation patterns, the injected embryos developed to blastocyst stage were collected and the genomic DNA was extracted with embryo lysis containing 1% NP40 at 50 °C for 20 minutes and 90 °C for 5 minutes in BIO-RAD PCR machine. The genomic DNA from ear punch tissues of newborn pups was isolated using the TIANamp Genomic DNA Kit (TIANGEN, Beijing, China). Then the target site was PCR amplified using the primers as follows: F 5′GGAGCAAGAGCCAATCATAGA 3′ and R 5′ TGAGGCTGTGAAGGCATAAG 3′. The PCR products were subjected to T7EI assay and T-cloning sequence.
The T7EI assay was performed as described previously26. Briefly, PCR products were purified with TIANgel Midi Purification Kit (TIANGEN, Beijing, China) and were denatured and annealed in NEBuffer 2 (NEB) using a thermocycler. Hybridized PCR products were digested with T7 endonuclease 1 (NEB, M0302L) for 30 minutes at 37 °C and subjected to 2% agarose gel electrophoresis.
Off-target analysis
The POTS of the two sgRNAs were predicted using the CRISPR design tool (http://tools.genomeengineering.org). The top 5 POTS were selected for each sgRNA according to ranking scores. The PCR primers used in this study are shown in Supplementary Table S2. The PCR products were analyzed by T7EI assay and then cloned into the pGM-T (Tiangen, Beijing, China) for Sanger sequence.
Western blotting and histology analysis
Samples from gluteus maximus tissue in MSTN KO and WT rabbits (euthanized at 2 months of age) of F1 generation were homogenized and lysed in RIPA buffer supplemented with 2.5 μL/mL protease inhibitor cocktail (Roche) on ice for 30 min. The protein concentrations were determined by Bradford method (Bio-Rad). 35 μg of protein sample was subjected to a 5% stacking/12% separating SDS-polyacrylamide gel. The antibodies used in this study include: anti-MSTN polyclonal antibody (Abcam) and goat anti-rabbit IgG conjugated with horseradish peroxidase (HRP) (Santa Cruz, USA). The β-actin antibody (Santa Cruz) was used as an internal control.
The tissues of tongue and gluteus maximus from MSTN KO and WT rabbits (euthanized at 2 months old) of F1 generation were fixed with 4% paraformaldehyde, then embedded in paraffin wax and slide sectioned. The sections were stained with hematoxylin and eosin (H&E) and analyzed by microscope (Nikon ts100).
Body weight, carcass dissection and sample collection
Four MSTN+/− rabbits of F1 and four WT counterparts controls were bred under the same conditions and used in this study. All rabbits were weaned at 30 days of age and housed in individual cages of the same litter. The body weight was recorded weekly from 1–8 weeks. At 2 months of age, the rabbits were anesthetized with isofluorane, following the injection of beuthanasia (1 mL/4 kg). Then the carcasses were dissected and the heart, tongue, gluteus maximus and vastus lateralis of the MSTN KO and WT groups were weighed and recorded separately.
Statistical analysis
Data was statistically analyzed by Graphpad prism software (T test) and a p value < 0.05 was considered statistically significant. *p < 0.05, **p < 0.01, ***p < 0.001.
Additional Information
How to cite this article: Lv, Q. et al. Efficient Generation of Myostatin Gene Mutated Rabbit by CRISPR/Cas9. Sci. Rep. 6, 25029; doi: 10.1038/srep25029 (2016).
References
Horii, T. et al. Validation of microinjection methods for generating knockout mice by CRISPR/Cas-mediated genome engineering. Sci. Rep. 4, 4513, doi: 10.1038/srep04513 (2014).
Crispo, M. et al. Efficient generation of myostatin knock-out sheep using CRISPR/Cas9 technology and microinjection into zygotes. Plos one 10, e0136690 (2015).
Kang, Y. et al. CRISPR/Cas9-mediated Dax1 knockout in the monkey recapitulates human AHC-HH. Hum. Mol. Genet ddv425, doi: 10.1093/hmg/ddv425 (2015).
Wang, Y. et al. Efficient generation of gene-modified pigs via injection of zygote with Cas9/sgRNA. Sci. Rep. 5, 8256, doi: 10.1038/srep08256 (2015).
Wang, Y. et al. Generation of knockout rabbits using transcription activator-like effector nucleases. Cell. Regen. 3, 1–9 (2014).
Wang, Y. et al. Human apolipoprotein A-II protects against diet-induced atherosclerosis in transgenic rabbits. Arterioscl. Throm. Vas. 33, 224–231 (2013).
Kondo, M. et al. Generation of a transgenic rabbit model of retinal degeneration. Invest. Ophth. Vis. Sci. 50, 1371 (2009).
Lee, S.-J. Regulation of muscle mass by myostatin. Annu. Rev. Cell Dev. Biol. 20, 61–86 (2004).
McPherron, A. C., Lawler, A. M. & Lee, S.-J. Regulation of skeletal muscle mass in mice by a new TGF-p superfamily member. Nature 387, 83–90 (1997).
Fries, R., Hanset, R. & Georges, M. A deletion in the bovine myostatin gene causes the double-muscled phenotype in cattle. Nat. Genet. 17, 71 (1997).
Clop, A. et al. A mutation creating a potential illegitimate microRNA target site in the myostatin gene affects muscularity in sheep. Nat. Genet. 38, 813–818 (2006).
Qian, L. et al. Targeted mutations in myostatin by zinc-finger nucleases result in double-muscled phenotype in Meishan pigs. Sci. Rep. 5, 14435, doi: 10.1038/srep14435 (2015).
Zou, Q. et al. Generation of gene-target dogs using CRISPR/Cas9 system. J. Mol. Cell. Biol. 7, 580–583 (2015).
Zhou, F., Sun, R., Chen, H., Fei, J. & Lu, D. Myostatin Gene Mutated Mice Induced with TALE Nucleases. Anim. Biotechnol. 26, 169–179 (2015).
Ni, W. et al. Efficient gene knockout in goats using CRISPR/Cas9 system. Plos one 9, e106718 (2014).
Zhou, J. et al. Dual sgRNAs facilitate CRISPR/Cas9 mediated mouse genome targeting. FEBS. J. 281, 1717–1725 (2014).
Pattanayak, V. et al. High-throughput profiling of off-target DNA cleavage reveals RNA-programmed Cas9 nuclease specificity. Nat. Biotechnol. 31, 839–843 (2013).
Hsu, P. D. et al. DNA targeting specificity of RNA-guided Cas9 nucleases. Nat. Biotechnol. 31, 827–832 (2013).
Fu, Y. et al. High-frequency off-target mutagenesis induced by CRISPR-Cas nucleases in human cells. Nat. Biotechnol. 31, 822–826 (2013).
Wu, X. et al. Genome-wide binding of the CRISPR endonuclease Cas9 in mammalian cells. Nat. Biotechnol. 32, 670–676 (2014).
Ran, F. A. et al. Double nicking by RNA-guided CRISPR Cas9 for enhanced genome editing specificity. Cell 154, 1380–1389 (2013).
Guilinger, J. P., Thompson, D. B. & Liu, D. R. Fusion of catalytically inactive Cas9 to FokI nuclease improves the specificity of genome modification. Nat. Biotechnol. 32, 577–582 (2014).
Arthur, P. Double muscling in cattle: a review. Crop. Pasture. Sci. 46, 1493–1515 (1995).
Mosher, D. S. et al. A mutation in the myostatin gene increases muscle mass and enhances racing performance in heterozygote dogs. Plos Genet 3, e79 (2007).
Wegner, J. et al. Growth-and breed-related changes of muscle fiber characteristics in cattle. J. Anim. Sci. 78, 1485–1496 (2000).
Guschin, D. Y. et al. A rapid and general assay for monitoring endogenous gene modification. Methods Mol. Biol. 649, 247–256 (2010).
Acknowledgements
We thank Peiran Hu and Xue Chen at the Embryo Engineering Center for the critical technical assistance. This work was financially supported by the National Transgenic Project of China (2014ZX0800602B) and National Natural Science Foundation of China (Grant No. 31201080 and 31272394).
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: Q.L., Z.L. and L.L. Performed the experiments: Q.L., L.Y., J.D. and M.C. Analyzed the data: Q.L., L.Y., Z.L. and L.L. Contributed reagents/materials/anaylsis tools: Y.W. and J.Z. Wrote the paper: Q.L. and L.L. All authors have read and approved the final manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Lv, Q., Yuan, L., Deng, J. et al. Efficient Generation of Myostatin Gene Mutated Rabbit by CRISPR/Cas9. Sci Rep 6, 25029 (2016). https://doi.org/10.1038/srep25029
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep25029
This article is cited by
-
A comprehensive review on the risks assessment and treatment options for Sarcopenia in people with diabetes
Journal of Diabetes & Metabolic Disorders (2023)
-
Functional replacement of myostatin with GDF-11 in the germline of mice
Skeletal Muscle (2022)
-
Inhibition of myostatin and related signaling pathways for the treatment of muscle atrophy in motor neuron diseases
Cellular and Molecular Life Sciences (2022)
-
Exploring the impacts of different fasting and refeeding regimes on Nile tilapia (Oreochromis niloticus L.): growth performance, histopathological study, and expression levels of some muscle growth-related genes
Fish Physiology and Biochemistry (2022)
-
Esophageal striated muscle hypertrophy and muscle fiber type transformation in MSTN knockout pigs
Transgenic Research (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.