Abstract
Humans and other animals use previous experiences to make behavioural decisions, balancing the probabilities of receiving rewards or punishments with alternative actions. The dopaminergic system plays a key role in this assessment: for instance, a decrease in dopamine transmission, which is signalled by the failure of an expected reward, may elicit a distinct behavioural response. Here, we tested the effect of exogenously administered dopaminergic compounds on a cooperative vertebrate’s decision-making process, in a natural setting. We show, in the Indo-Pacific bluestreak cleaner wrasse Labroides dimidiatus, that blocking dopamine receptors in the wild induces cleaners to initiate more interactions with and to provide greater amounts of physical contact to their client fish partners. This costly form of tactile stimulation using their fins is typically used to prolong interactions and to reconcile with clients after cheating. Interestingly, client jolt rate, a correlate of cheating by cleaners, remained unaffected. Thus, in low effective dopaminergic transmission conditions cleaners may renegotiate the occurrence and duration of the interaction with a costly offer. Our results provide first evidence for a prominent role of the dopaminergic system in decision-making in the context of cooperation in fish.
Similar content being viewed by others
Introduction
Humans excel in their ability to cooperate among unrelated individuals1 but it is clear that there is also enormous variation between other species with respect to their cooperative tendencies. A recent major research focus is to understand the mechanistic basis of cooperative behaviour, particularly the cognitive and physiological processes underlying decision-making2,3,4. Vertebrate brain structures involved in social decision-making are highly conserved. Most importantly, all vertebrates have a so-called social decision-making network, which consists of the social behaviour network and the mesolimbic reward system5,6. This network appears to be highly sensitive to the dopaminergic system5,7, making dopamine a prime candidate for the modulation of cooperative behaviour.
Dopamine (DA) is a neurotransmitter involved in a variety of neurochemical and neurohormonal actions that affect and modulate animal behaviour and cognition4,6. Dopamine is involved in reward and risk assessment, behaviour reinforcement8,9 and anticipatory responses to reward-associated stimuli8 as its release signals the outcome of an action as appetitive or aversive10,11. Thus, DA is key to associative learning12. First, DA signals the delivery of an unexpected outcome (reward or punishment) which is usually preceded or paired with specific stimuli13. Later, through repeated encounters, individuals learn to associate the outcome with the preceding stimuli and the dopaminergic response progressively shifts to this earlier event-predicting stimuli rather than responding to the outcome itself11,14,15,16. This gradually enables animals to anticipate outcomes in current interactions by recalling previously learned associations, which results in appropriate decision-making17. Moreover, DA signalling suffers a depression (DA transmission decreases momentarily) whenever the event happens contrarily to the prediction and the expected outcome fails to occur18. This decrease may elicit a distinct behavioural response: for example, in humans, the omission of an expected reward can lead to emotional distress19, while in other mammals, birds and teleost fish it may induce aggressive behaviour20,21,22. Nevertheless, signalling environmental changes is key for learning and decision-making, as these allow for an evaluation of the behavioural adjustments needed in order to achieve the expected outcome once again23. As such, anticipation is crucial for deciding between different courses of action available18, as different options entail uncertain final outcomes. A prime context in which correct anticipation is crucial is cooperation between unrelated individuals that is based on investments. The classic theoretic game model to describe such cooperation is the iterated prisoner’s dilemma24. In this 2-player game, mutual cooperation yields higher payoffs than mutual defection but defecting yields a higher payoff than cooperating, independently of the partner’s action. Thus, there are incentives both to cooperate and to defect and an individual’s best decision will depend on the partner’s previous strategy25. Similar conflicting incentives exist in many other potentially cooperative interactions26. A good example is the marine cleaning mutualism involving the Indo-Pacific bluestreak cleaner wrasse Labroides dimidiatus. As summarised elsewhere27, these territorial cleaner fish remove ectoparasites from visiting ‘client’ reef fish. Interactions are best described as a repeated game; clients are estimated to visit cleaning stations typically 5–30 times per day, with maximal estimates above 100 visits28. A conflict of interest exists because cleaners prefer to eat client mucus, which constitutes cheating as it is detrimental to the client. Cheating is visible to the human observer through clients performing body jolts in response to cleaner wrasse mouth contacts27. As a consequence of cleaner wrasse food preferences, clients have to make cleaners feed against their preference to obtain a good service. How this is achieved depends on the clients’ strategic options in this repeated game. For predatory clients, the mere threat of reciprocation (trying to eat a cheating cleaner) is apparently enough to cause high service quality, while non-predatory client species either punish cleaners through aggressive chasing or leave and switch to a different cleaner for their next inspection, which constitutes the threat of departure27,29. In response, cleaners flexibly adjust their cheating frequency to a variety of parameters, which include client’s control mechanisms, the presence of bystanders, the presence of a co-inspecting cleaner partner, the client’s value as a food source and also the cleaner’s own physiological state27,30,31,32. Furthermore, cleaners can improve their service quality by providing a form of physical contact (known as tactile stimulation or massages) to clients, touching them with their pectoral and (especially) pelvic fins. Cleaners use tactile stimulation in a variety of contexts but usually when the outcome of the interaction is not certain: to build relationships with new clients, to reconcile after a cheating event, to prolong interactions with clients about to leave and as a pre-conflict management strategy with predators33,34. Clients apparently benefit from receiving tactile stimulation as it lowers baseline and acute stress levels (i.e. cortisol levels35). Thus, in marine cleaning mutualisms, two elements of behavioural negotiation are used by partners to resolve the conflict over cooperative payoffs: a) the use of threats (reciprocity or departure) and b) the use of tactile stimulation to encourage clients to stay at cleaning stations as a type of negotiation29. Overall, game theory has successfully been used to predict and explain partner control mechanisms in this system36. Regarding cleaner wrasses’ behavioural adjustments, game models should consider how physiological constraints (for example, the existence of stressed cleaners32) may limit the expression of some of these decision rules.
Here, we aimed to investigate the relevance of the dopaminergic system for the cleaners’ service quality during cleaning interactions and how these individuals respond to changes of perception elicited by DA level shifts. Only a few studies have examined the role of the DA system on the modulation of fish behaviour, mostly on locomotor activity37, brain responses to light and hydrostatic pressure38, feeding behaviour39, coping with unpredictability40, learning and nicotine41, gene expression and neuroendocrine signalling42,43,44,45 and learning performance in a cooperative context46. Only some of the above-cited studies employed drugs aimed at the Dopamine D1 and D2 receptors, that were previously developed for mammals, which were successfully used in fish to test for putative effects on behaviour or gene expression37,46. For example, in cichlids, the effects caused by the use of a non-selective DA agonist that activates both D1 and D2 receptors on locomotor activity were blocked by the D1 antagonist (SCH-23390) but not by the D2 antagonist (metoclopramide). Also, the effects of several D1 and D2 related drugs produced distinct neuroendocrine and brain expression responses42,43,44,45. Using cleaners, Messias and colleagues46 showed that there is a direct involvement of the D1 receptor pathways on their natural ability to learn. As in these previous studies, we exogenously administered a D1 receptor agonist (D1a - SKF38393), an antagonist (D1an - SCH23390), a D2 receptor agonist (D2a - Quinpirole) and an antagonist (D2an - Metoclopramide), as well as a control (saline) to female cleaner wrasses in situ. As this mutualistic system occurs in a biological market27,47, efficient dopaminergic transmission could play a role in the modulation of cleaners’ willingness to negotiate with clients over the occurrence and duration of interactions as well as cleaners’ willingness to cooperate rather than cheating27. High increases in DA transmission via administration of agonists are connected with pathological gambling48 and excessive risk-taking49. Hence, we predict D1a and D2a to decrease cleaners’ cooperative investment levels and increase their cheating frequencies. Since D2 receptors can also be found pre-synaptically (i.e. as auto-receptors) in some areas of the brain, it is also possible that D2 stimulation leads to risk-avoidance behaviour by overstimulating the pre-synaptic receptors50. Similarly, DA receptor blockade induces risk-avoidance behaviour through an increase in sensitivity to negative stimuli49,51,52. We thus also predict that DA antagonists would cause cleaners to seek clients to clean more and provide more tactile stimulation to entice clients to stay longer, with the possibility that blocking the D2 autoreceptors might lead to abnormal DA transmission. Regarding cheating by cleaners, a perceived reduction in the probability of expected outcomes would mean a reduced ability to maintain the interaction with clients and a lower likelihood to obtain food. Such perception may, either lead to an extension of negotiation, where high rates of tactile stimulation lead to a reduction of cheating, or to just abandoning negotiation – with cleaners foraging as much and as quickly possible, which would mean immediate cheating by feeding on clients’ mucus53.
Results
The five compounds administered consisted of a D1 receptor agonist SKF38393 (D1a) and antagonist SCH23390 (D1an), a D2 receptor agonist Quinpirole (D2a) and antagonist Metoclopramide (D2an) and a saline solution as a control. Using Statistica 12 software, Kruskal-Wallis ANOVA tests were performed to detect differences between treatments in each behavioural variable and Dunn’s Post-Hoc tests were applied to compare each treatment against the control group which include a Bonferroni adjustment to account for multiple comparisons54.
Dopamine effects on the likelihood to engage in cleaning behaviour
Cleaner wrasses treated with D1an inspected a significantly higher proportion of clients when compared with the control group (Kruskal-Wallis ANOVA, H (4, N = 50) = 17.4435, p = 0.0016; Dunn’s Post-Hoc test: D1an vs Saline, p = 0.0001, Fig. 1A), whereas other treatments did not differ from the control group (Dunn’s Post-Hoc test: D1a vs Saline, p = 0.4000; D2a vs Saline, p = 0.1451; D2an vs Saline, p = 0.2764, Fig. 1A). Cleaner wrasses treated with D1an also had on average longer interactions with clients compared with the control group (Kruskal-Wallis ANOVA, H (4, N = 50) = 18.2820, p = 0.0011; Dunn’s Post-Hoc test: D1an vs Saline, p = 0.0025, Fig. 1A), whereas other treatments did not differ from the control group (Dunn’s Post-Hoc test: D1a vs Saline, p = 0.4000; D2a vs Saline, p = 0.4000; D2an vs Saline, p = 0.4000, Fig. 1B).
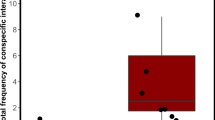
The effect of D1 agonist (D1a) and antagonist (D1an), D2 agonist (D2a) and antagonist (D2an) on cleaners’ likelihood to engage in cleaning behaviour measured by (a) the proportion of clients inspected (calculated as the total number of clients inspected/total number of visits) and (b) the mean interaction duration (total time of interaction/total number of interactions); cleaners’ cooperative investment measured by: (c) the proportion of interactions with tactile stimulation events (frequency of clients inspected where tactile stimulation occurred/total number of interactions), (d) the proportion of time cleaners spent providing tactile stimulation (total tactile stimulation duration/total interaction duration); and (e) cleaner wrasse cheating levels measured by the frequency of client jolts per 100 seconds of inspection. Medians and interquartile ranges are shown. Significant values are shown above bars: *<0.05; **<0.01; ***<0.001 and refer to Dunn’s Post-Hoc tests affecting each dopamine treatment against the reference (saline) group, for a total sample size of 10 individuals.
Dopamine effects on tactile stimulation of clients
Cleaner wrasses treated with either D1an or D2an had a higher proportion of interactions in which they provided tactile stimulation to their clients compared to the control group (Kruskal-Wallis ANOVA, H (4, N = 50) = 23.47111, p = 0.0001; Dunn’s Post-Hoc test: D1an vs Saline, p = 0.0004; D2an vs Saline, p = 0.0111, Fig. 1C) whereas other treatments did not differ from the control group (Dunn’s Post-Hoc test: D1a vs Saline, p = 0.0856; D2a vs Saline, p = 0.4000, Fig. 1C). Cleaner wrasse treated with D1an spent a greater proportion of inspection time providing tactile stimulation to their clients (Kruskal-Wallis ANOVA, H (4, N = 50) = 11.1371, p = 0.0251; Dunn’s Post-Hoc test: D1an vs Saline, p = 0.0082, Fig. 1D), whereas other treatments did not differ from the control group (Dunn’s Post-Hoc test: D1a vs Saline, p = 0.4000; D2a vs Saline, p = 0.4000; D2an vs Saline, p = 0.4000, Fig. 1D).
Dopamine effects on cleaner wrasses cheating levels
Client jolt frequency in all dopamine treatments did not differ from the control group (Kruskal-Wallis ANOVA, H (4, N = 50) = 4.76234, p = 0.3126, Fig. 1E).
Discussion
We provide experimental evidence that the dopaminergic system is linked to the decisions of cleaner wrasses during interactions with client reef fish. Decreasing DA transmission was effective, particularly with respect to the D1 type receptor, while increasing effective dopamine transmission did not yield measurable effects. This latter result agrees with previous studies suggesting that increases in dopaminergic transmission are not as relevant to decision making based on previously made associations as it is during the learning process13. Decreases in DA transmission signal an outcome that is worse than predicted11, which in the case of cleaners would mean a reduced likelihood to obtain food during a cleaning interaction or higher chances of being punished (by being chased or the client leaving). In game-theoretical terms, physiological evidence suggests that dopamine affects an individual’s perception of the payoffs associated with each potential action. The key question was then how cleaners would respond to changes in payoff perception in a repeated game with respect to their effort to make the interaction happen or to prolong it and how this would affect their level of cooperative foraging. Our results show that blocking DA receptors made cleaners seek more interactions and increase tactile stimulation to clients (both duration and frequency), i.e. the behaviours that cleaners can use to negotiate the occurrence and duration of interactions with clients27. In contrast, levels of cooperative foraging and hence cheating rates remained unaffected by our experimental manipulations. In other words, reductions in normal DA tone lead cleaners to renegotiate the occurrence and duration of the interaction using a ‘costly offer’ instead of reducing overall cheating rates. The lack of change in cheating rates was surprising, given that theoretical considerations would predict either an increase or a decrease51,53. A new model that captures the specifics of cleaning interactions may help to establish whether our measured effects of dopamine on cleaners would be optimal under natural conditions. Such a model could also investigate how far changes in the perception of repetitiveness probability would produce similar effects or not. Moreover, future research should test for the role of dopaminergic pathways on cleaner wrasse decision-making under particular conditions in which the temptation to cheat is higher, for example in laboratory “eating against preference” contextual tasks (alone or in pairs).
The blockade of dopamine receptors (both D1 and D2) appeared to cause cleaners to behave as if clients were permanently in dispute about the value of being serviced (increasing sensitivity to negative stimuli). In this case, the use of tactile stimulation agrees with previous findings, which revealed that cleaners provide tactile stimulation to manipulate client decisions, to appease predators, to build up relationships and to reconcile after a conflict33,34. Tactile stimulation is a costly form of negotiation as it is incompatible with foraging. Thus, changes in the dopaminergic system appear to be responsible for the modulation of cleaner wrasses’ perception (i.e. anticipation) concerning the expected reward and the predicted costs/risks involved in any given decision when dealing with clients (i.e. the client is viewed as more likely to leave). To the best of our knowledge, the present study represents the first examination of the effect of exogenous dopaminergic compounds’ administration in a cooperative decision-making process tested in a natural setting. The results fit laboratory studies on the role of dopamine in the modulation of risky based decision-making in humans55,56 and rats51. For instance, individuals with lower levels of dopamine in the prefrontal cortex tend to risk less in a gambling task57. Another example of effects of depletion of dopaminergic transmission involves untreated patients with Parkinson’s disease which are more sensitive to negative or uncertain outcomes (risk aversion)55,56.
Blockade of dopamine D2 receptors caused an increase of tactile stimulation events but not the amount of time spent providing it, while D1 blockade produced a stronger impairment on cleaner wrasses’ overall behaviour (which also included the frequency and time spent providing physical contact to clientele). Indeed, there are general differences in the affinity of DA to each of these receptors (as shown for mammals): for instance, D2-like receptors have a 10-to-100 fold greater affinity than the D1-like family, with the D1a being reported to have the lowest affinity for DA58,59. Moreover, because the D1-like receptors are solely found postsynaptically, while the D2-like receptors are the predominant type of autoreceptor being found both pre- and postsynaptically58, it is expected that the pathways involved in the function of both receptor types are likely distinct. D2 receptors are able to induce a negative feedback regulation that may inhibit DA neuron firing, synthesis and release60, functioning as a control mechanism and D1 receptors, in contrast, play a direct stimulatory role. Also, some studies have referred to opposing but sometimes complementary roles played functionally by D1 and D2 receptors. This is in agreement with our results which suggest that both receptors regulate these behavioural trade-offs, yet with different magnitudes, but mostly that these receptors manage the perception of feedback received by the focal animal during interactions, with D1 receptors having the main modulatory role60. Another potential explanation would arise from a putative unexpected effect of the D2 blockade on D1 receptors, which is less likely to occur due to differences in affinity between both receptor types and the selectivity of the compounds used37,46.
The absence of an effect when D1 and D2 selective agonists were administrated was not entirely surprising, as abnormal increases in DA transmission should have a higher impact on behaviour when new associations are being established and be less effective in pre-existing ones12,13. We did, however, predict both compounds to produce changes in behaviour and alterations in their strategies, as these have been found elsewhere to cause profound changes in the ability to adjust behaviour51. The lack of effects on D1a manipulation could also be caused by an abnormal activity of D2 auto-receptors inhibiting DA release50. Another possible explanation is that cleaners may already have high endogenous dopamine levels and thus the addition of an exogenous smaller dosage did not contribute to any further changes in behaviour. However, in human Parkinson’s disease patients, the side effects of significant and long-term increases in dopamine levels are relevant and may include, for instance, excessive risk taking behaviour (pathological gambling)51,61. Hence, it would be possible for other dosages to be more effective, something that is amenable for further testing in the future.
In this study, we provide first mechanistic evidence regarding the prominence of the dopaminergic system modulation in the context of cooperation (in fish); specifically to the mediation of cleaner wrasses’ negotiation skills and avoidance of negative consequences during potential conflicting interactions. Future studies should also focus on finding out how natural baseline changes in dopamine transmission may influence variation in individual contributions to cooperative behaviour and impulsivity regarding risky choices. While it is important that such studies continue to focus on cleaner wrasses, it is equally relevant to examine other vertebrate model systems, as it has also been demonstrated for humans and other animals51,57,61 particularly in the context of the highly conserved vertebrate social decision-making network5,6,7.
Materials and Methods
Field methods
Field experiments took place on reefs around Lizard Island (Lizard Island Research Station, Australia, 14˚40’S, 145˚28’E) with 50 female cleaner wrasses tested between late August and September 2012. Because their larvae settle onto the reef three weeks after hatching62 and at Lizard Island this occurs in November and December, their spawning likely occurs between October and December. We thus assumed all sampled females would not have their behaviour affected by this variable. Furthermore, all reefs sampled were fringing reefs, ensuring that all individuals come from ecologically similar contexts. Cleaners were selected haphazardly across reefs, while cleaning stations varied in depth (between 2 and 10 m). All individuals were captured using a barrier net and hand net, measured to the nearest mm (TL – Total Length: ranged from 6.0 cm to 8.1 cm) and their body weight was then estimated using a length-weight regression (Soares MC, unpublished data). Each focal cleaner was administered, via intramuscular injection, with one of five compounds: saline solution for control (0.9% NaCl); a selective D1 agonist SKF-38393 (D047 – Sigma); selective D1 antagonist SCH-23390 (D054 - Sigma); selective D2 (and D3) agonist Quinpirole (Q102 - Sigma); selective D2 antagonist Metoclopramide (M0763 – Sigma). Because this study was done exclusively in field conditions with limitations of time and number of fish used (collecting permit allowance), compound dosages applied were based on previous studies: 5 μg/gbw (gram of estimated body weight) of SKF-3839363,64,65, 0.5 μg/gbw of SCH-2339052,66, 2μg/gbw of Quinpirole67, 5μg/gbw of Metoclopramide37,68. Injection volumes were always 15 μl per gbw. This process never exceeded 3 min. SKF-38393 is a selective D1 and (and partial D5) agonist that can simulate dopamine activity63 and can disrupt collective behaviour, such as shoaling69. SCH-23390 is a high-affinity selective D1 antagonist with negligible effects on D2 receptors and slight effects on 5-HT2A/C receptors70. However, effects on the serotonin systems may be dismissed, since 5-HT2A receptors have not been found on fish yet71 and the dosage needed to produce effects on 5-HT2C is 10-fold higher than the dosage needed for D1 blockade72. Quinpirole is a selective D2 agonist73 widely used in a variety of scientific studies related to D2 receptor manipulation. Metoclopramide, commonly known for its anti-emetic effect via the chemoreceptor trigger zone, is a selective D2 antagonist, acting as a dopamine inhibitor37,74. Although it also has slight effects on the serotonin system, the target receptors have not yet been discovered in the teleost fish brain71. Since reward-driven behaviour and decision-making faculties are controlled by central mechanisms, all compounds chosen were reported to be capable of crossing the blood-brain barrier, to ensure the results attained take place in (but not exclusively on) central systems75,76,77,78. The order of the treatments was randomized for each dive and all treatments used different cleaner wrasse. After administering an individual it was released and then observed and videotaped for the next 45 min using a Sony Cyber-Shot DSC-W570 camera in a waterproof housing, always from a distance of 2–3 m.
Behavioural analysis
The following measurements were noted for each interaction filmed on video: a) client species visiting the cleaning station; b) who initiated the interaction: clients were scored as the ones initiating an interaction if they posed before the cleaner touched them. Otherwise, the cleaner was scored as the one initiating; c) duration (in seconds) of inspection towards each client; d) the number and duration of tactile stimulation events provided to each client; and e) number of jolts performed by clients.
Statistical Analysis
Measures of cleaner wrasse behaviour towards clients were divided into three categories: a) measures of likelihood to engage in cleaning behaviour (motivation to interact); b) measures of interactive investment (provision of tactile stimulation); and c) a measure of cleaner wrasse cheating levels (client jolt rate). The likelihood to engage with clientele was measured by: 1) the proportion of clients inspected (calculated as the total number of clients inspected/total number of visits) and 2) the mean duration of inspection (total time of interaction/total number of interactions). Measures of investment were calculated as: 1) the proportion of interactions in which tactile stimulation was used by cleaners (frequency of clients inspected where tactile stimulation occurred/total number of interactions) and 2) the proportion of time cleaners spent providing tactile stimulation to clients (total tactile stimulation duration/total interaction duration). Finally, the measure of cleaners’ cheating levels was calculated using the frequency of jolts per 100 seconds of inspection. Data were analysed using non-parametric tests because the assumptions for parametric testing were not met. Kruskal-Wallis ANOVA tests were performed to detect differences between treatments for each behavioural variable and Dunn’s Post-Hoc tests were applied using Statistica 12 software, which already include a Bonferroni adjustment to account for multiple comparisons to compare each treatment against the control group54. The p-value obtained was then further adjusted to account for comparisons against a control group, as suggested by the Statistica software54.
Ethical Note
Ethical clearance to work at Lizard Island Research Station (Australian Museum), which involved animal manipulation, was obtained from The University of Queensland Animal Ethics Committee. The use of animals and data collection complied with the laws of Australia.
Data accessibility
The datasets supporting this article have been uploaded as part of the electronic supplementary material.
Additional Information
How to cite this article: Messias, J. P. M. et al. Dopamine disruption increases negotiation for cooperative interactions in a fish. Sci. Rep. 6, 20817; doi: 10.1038/srep20817 (2016).
References
Fehr, E. & Rockenbach, B. Human altruism: economic, neural and evolutionary perspectives. Curr. Opin. Neurobiol. 14, 784–790 (2004).
Brosnan, S. F., Salwiczek, L. & Bshary, R. The interplay of cognition and cooperation. Philos. Trans. R. Soc. London. 365, 2699–2710 (2010).
Melis, A. P. & Semmann, D. How is human cooperation different? Philos. Trans. R. Soc. Lond. B. Biol. Sci. 365, 2663–2674 (2010).
Soares, M. C. et al. Hormonal mechanisms of cooperative behaviour. Proc. R. Soc. Lond. B 365, 2737–2750 (2010).
O’Connell, L. A. & Hofmann, H. A. The vertebrate mesolimbic reward system and social behavior network: a comparative synthesis. J. Comp. Neurol. 519, 3599–3639 (2011).
Goodson, J. L. The vertebrate social behavior network: evolutionary themes and variations. Horm. Behav. 48, 11–22 (2005).
O’Connell, L. A. & Hofmann, H. A. Evolution of a vertebrate social decision-making network. Science. 336, 1154–1157 (2012).
Heimovics, S. A., Cornil, C. A., Ball, G. F. & Riters, L. V. D1-like dopamine receptor density in nuclei involved in social behavior correlates with song in a context-dependent fashion in male European starlings. J. Neurosci. 159, 962–973 (2009).
Berridge, K. C. & Robinson, T. E. What is the role of dopamine in reward: hedonic impact, reward learning, or incentive salience? Brain Res. 28, 309–369 (1998).
Salamone, J. D. & Correa, M. The mysterious motivational functions of mesolimbic dopamine. Neuron 76, 470–485 (2012).
Schultz, W. Predictive reward signal of dopamine neurons. J. Neurophysiol. 80, 1–27 (1998).
DeWitt, E. E. J. Neuroeconomics: a formal test of dopamine’s role in reinforcement learning. Curr. Biol. 24, 321–324 (2014).
Schultz, W. Behavioral theories and the neurophysiology of reward. Annu. Rev. Psychol. 57, 87–115 (2006).
Schultz, W. et al. In Model. Inf. Process. Basal Ganglia ( Houk, J. C., Davis, J. L. & Beiser, D. G. ) 233–248 (Cambridge, MA: MIT Press, 1995).
O’Doherty, J. P. Reward representations and reward-related learning in the human brain: insights from neuroimaging. Curr. Opin. Neurobiol. 14, 769–776 (2004).
Young, L. J. & Wang, Z. The neurobiology of pair bonding. Nat. Neurosci. 7, 1048–1054 (2004).
Schultz, W. Getting formal with dopamine and reward. Neuron 36, 241–263 (2002).
Schultz, W., Dayan, P. & Montague, P. R. A neural substrate of prediction and reward. Science. 275, 1593–1599 (1997).
Abler, B., Walter, H. & Erk, S. Neural correlates of frustration. Neuroreport 16, 669–672 (2005).
Zimmerman, P. H. & Koene, P. The effect of frustrative nonreward on vocalisations and behaviour in the laying hen, Gallus gallus domesticus. Behav. Processes 44, 73–79 (1998).
Almeida, R. M. M. & Miczek, K. A. Aggression escalated by social instigation or by discontinuation of reinforcement (‘frustration’) in mice: inhibition by anpirtoline: a 5-HT1B receptor agonist. Neuropsychopharmacology 27, 171–181 (2002).
Vindas, M. A. et al. Frustrative reward omission increases aggressive behaviour of inferior fighters. Proc. R. Soc. B 281 (2014).
Steinberg, E. E. et al. A causal link between prediction errors, dopamine neurons and learning. Nat. Neurosci. 16, 966–973 (2013).
Luce, R. D. & Raiffa, H. Games and decisions: Introduction and critical survey. J. Am. Stat. Assoc. 53, 758–761 (1958).
Axelrod, R. & Hamilton, W. D. The evolution of cooperation. Science. 211, 1390–1396 (1981).
Bshary, R. & Bronstein, J. L. A general scheme to predict partner control mechanisms in pairwise cooperative interactions between unrelated individuals. Ethology 117, 271–283 (2011).
Bshary, R. & Côté, I. M. In Fish Behav. ( Magnhagen, C., Braithwaite, V. A., Forsgren, E. & Kapoor, B. G. ) 563–592 (Science Publishers, 2008). 10.1201/b10757-20
Grutter, A. S. Relationship between cleaning rates and ectoparasite loads in coral reef fishes. Mar. Ecol. Prog. Ser. 118, 51–58 (1995).
Cant, M. A. The role of threats in animal cooperation. Proc. R. Soc. B 278, 170–178 (2014).
Pinto, A., Oates, J., Grutter, A. S. & Bshary, R. Cleaner wrasses Labroides dimidiatus are more cooperative in the presence of an audience. Curr. Biol. 21, 1140–1144 (2011).
Raihani, N. J., Grutter, A. S. & Bshary, R. Female cleaner fish cooperate more with unfamiliar males. Proc. R. Soc. Lond. B 279, 2479–2486 (2012).
Soares, M. C., Cardoso, S. C., Grutter, A. S., Oliveira, R. F. & Bshary, R. Cortisol mediates cleaner wrasse switch from cooperation to cheating and tactical deception. Horm. Behav. 66, 346–350 (2014).
Bshary, R. & Würth, M. Cleaner fish Labroides dimidiatus manipulate client reef fish by providing tactile stimulation. Proc. R. Soc. Lond. B 268, 1495–1501 (2001).
Grutter, A. S. Cleaner fish use tactile dancing behavior as a preconflict management strategy. Curr. Biol. 14, 1080–1083 (2004).
Soares, M. C., Oliveira, R. F., Ros, A. F. H., Grutter, A. S. & Bshary, R. Tactile stimulation lowers stress in fish. Nat. Commun. 2, 1–5 (2011).
Bshary, R. & Oliveira, R. F. Cooperation in animals: toward a game theory within the framework of social competence. Curr. Opin. Behav. Sci. 3, 31–37 (2015).
Mok, E. Y. M. & Munro, A. D. Effects of dopaminergic drugs on locomotor activity in teleost fish of the genus Oreochromis (Cichlidae): involvement of the telencephalon. Physiol. Behav. 64, 227–234 (1998).
Takemura, A., Uchimura, M. & Shibata, Y. Dopaminergic activity in the brain of a tropical wrasse in response to changes in light and hydrostatic pressure. Gen. Comp. Endocrinol. 166, 513–519 (2010).
Leal, E. et al. Effects of dopaminergic system activation on feeding behavior and growth performance of the sea bass (Dicentrarchus labrax): a self-feeding approach. Horm. Behav. 64, 113–121 (2013).
Vindas, M. A. et al. Coping with unpredictability: dopaminergic and neurotrophic responses to omission of expected reward in Atlantic salmon (Salmo salar L.). PLoS One 9, e85543 (2014).
Eddins, D., Petro, A., Williams, P., Cerutti, D. T. & Levin, E. D. Nicotine effects on learning in zebrafish: the role of dopaminergic systems. Psychopharmacology (Berl). 202, 103–109 (2009).
Popesku, J. T. et al. The goldfish (Carassius auratus) as a model for neuroendocrine signaling. Mol. Cell. Endocrinol. 293, 43–56 (2008).
Popesku, J. T., Mennigen, J. A., Chang, J. P. & Trudeau, V. L. Dopamine D1 receptor blockage potentiates AMPA-stimulated luteinising hormone release in the goldfish. J. Neuroendocrinol. 23, 302–309 (2011).
Popesku, J. T., Navarro-Martín, L. & Trudeau, V. L. Evidence for alternative splicing of a dopamine D2 receptor in a teleost. Physiol. Biochem. Zool. 84, 135–146 (2011).
Popesku, J. T., Martyniuk, C. J. & Trudeau, V. L. Meta-type analysis of dopaminergic effects on gene expression in the neuroendocrine brain of female goldfish. Front. Endocrinol. (Lausanne). 3, 130 (2012).
Messias, J.P.M., Santos, T.P., Pinto, M. & Soares, M.C. Stimulation of dopamine D1 receptor improves learning capacity in cooperating cleaner fish. Proc. R. Soc. B. 20152272 (2016). 10.1098/rspb.2015.2272
Noë, R. In Econ. Nat. ( Nöe, R., Van Hoof, J. & Hammerstein, P. ) 93–118 (Cambridge University Press, 2001). 10.1017/CBO9780511752421.008
Quickfall, J. & Suchowersky, O. Pathological gambling associated with dopamine agonist use in restless legs syndrome. Park. Relat. Disord. 13, 535–536 (2007).
St. Onge, J. R., Chiu, Y. C. & Floresco, S. B. Differential effects of dopaminergic manipulations on risky choice. Psychopharmacol. 211, 209–221 (2010).
Bello, E. P. et al. Cocaine supersensitivity and enhanced motivation for reward in mice lacking dopamine D2 receptors. Nat. Neurosci. 14, 1033–1038 (2012).
St. Onge, J. R. & Floresco, S. B. Dopaminergic modulation of risk-based decision making. Neuropsychopharmacol. 34, 681–697 (2009).
St. Onge, J. R., Abhari, H. & Floresco, S. B. Dissociable contributions by prefrontal d1 and d2 receptors to risk-based decision making. J. Neurosci. 31, 8625–8633 (2011).
Binmore, K. Bargaining in biology? J. Evol. Biol. 23, 1351–1363 (2010).
Dell Inc. STATISTICA (data analysis software system). (2015). at < www.statsoft.com>
Frank, M. J., Seeberger, L. C. & O’reilly, R. C. By carrot or by stick: cognitive reinforcement learning in parkinsonism. Science. 306, 1940–1943 (2004).
Mimura, M., Oeda, R. & Kawamura, M. Impaired decision-making in Parkinson’s disease. Parkinsonism Relat. Disord. 12, 169–175 (2006).
Roussos, P., Giakoumaki, S. G., Pavlakis, S. & Bitsios, P. Planning, decision-making and the COMT rs4818 polymorphism in healthy males. Neuropsychologia 46, 757–763 (2008).
Beaulieu, J.-M. & Gainetdinov, R. R. The physiology, signaling and pharmacology of dopamine receptors. Pharmacol. Rev. 63, 182–217 (2011).
Tritsch, N. X. & Sabatini, B. L. Dopaminergic modulation of synaptic transmission in cortex and striatum. Neuron 76, 33–50 (2012).
Chow, J. The role of d1 dopamine receptors on incentive salience attribution. Kaleidoscope 10, 1–7 (2011).
Gallagher, D. A., O’Sullivan, S. S., Evans, A. H., Lees, A. J. & Schrag, A. Pathological gambling in Parkinson’s disease: risk factors and differences from dopamine dysregulation. An analysis of published case series. Mov. Disord. 22, 1757–1763 (2007).
Brothers, E. B., Williams, D. M. & Sale, P. F. Length of larval life in twelve families of fishes at ‘One Tree Lagoon’, Great Barrier Reef, Australia. Mar. Biol. 76, 319–324 (1983).
Dong, C. J. & McReynolds, J. S. The relationship between light, dopamine release and horizontal cell coupling in the mudpuppy retina. J. Physiol. 440, 291–309 (1991).
Mattingly, B. A., Rowlett, J. K. & Lovell, G. Effects of daily SKF 38393, quinpirole and SCH 23390 treatments on locomotor activity and subsequent sensitivity to apomorphine. Psychopharmacol. 110, 320–326 (1993).
Cooper, S. J. & Al-Naser, H. A. Dopaminergic control of food choice: contrasting effects of SKF 38393 and quinpirole on high-palatability food preference in the rat. Neuropharmacol. 50, 953–963 (2006).
Rodríguez-Arias, M., Pinazo, J., Miñarro, J. & Stinus, L. Effects of SCH 23390, raclopride and haloperidol on morphine withdrawal-induced aggression in male mice. Pharmacol. Biochem. Behav. 64, 123–130 (1999).
Popesku, J. T., Martyniuk, C. J., Denslow, N. D. & Trudeau, V. L. Rapid dopaminergic modulation of the fish hypothalamic transcriptome and proteome. PLoS One 5, 1–10 (2010).
Brzuska, E., Kouril, J., Adamek, J., Stupka, Z. & Bekh, V. The application of [DTle6, ProNHEt9]mGnRH (Lecirelin) with the dopaminergic inhibitor metoclopramide to stimulate ovulation in African catfish (Clarias gariepinus). Czech J. Anim. Sci. 49, 297–306 (2004).
Echevarria, D. J., Hammack, C. M., Pratt, D. W. & Hosemann, J. D. A novel behavioral test battery to assess global drug effects using the zebrafish. Int. J. Comp. Psychol. 21, 19–34 (2009).
Hyttel, J., Arnt, J. & Berghe, M. In Clin. Pharmacol. ( Dahl, S. & Gram, L. ) 7, 109–122 (Springer Berlin Heidelberg, 1989).
Prasad, P., Ogawa, S. & Parhar, I. S. Role of serotonin in fish reproduction. Front. Neurosci. 9, 1–9 (2015).
Bourne, J. A. SCH 23390: the first selective dopamine D1-like receptor antagonist. CNS Drug Rev. 7, 399–414 (2001).
Levant, B., Grigoriadis, D. E. & DeSouza, E. B. [3H]quinpirole binding to putative D2 and D3 dopamine receptors in rat brain and pituitary gland: A quantitative autoradiographic study. J. Pharmacol. Exp. Ther. 264, 991–1001 (1993).
Albibi, R. & McCallum, R. W. Metoclopramide: Pharmacology and clinical application. Ann. Intern. Med. 98, 86–95 (1983).
Schlenker, E. H. In Hamsters the D1 Receptor Antagonist SCH 23390 Depresses Ventilation during Hypoxia. Brain Res. 1187, 146–153 (2008).
Kamien, J. B. & Woolverton, W. L. The D1 dopamine agonist SKF 38393 functions as a discriminative stimulus in rats. Psychopharmacology (Berl). 87, 368–370 (1985).
Kostrzewa, R. M., Brus, R., Rykaczewska, M. & Plech, A. Low-dose quinpirole ontogenically sensitizes to quinpirole-induced yawning in rats. Pharmacol. Biochem. Behav. 44, 487–489 (1993).
Jolliet, P. et al. Evidence of lowest brain penetration of an antiemetic drug, metopimazine, compared to domperidone, metoclopramide and chlorpromazine, using an in vitro model of the blood-brain barrier. Pharmacol. Res. 56, 11–17 (2007).
Acknowledgements
We thank the Directors and staff of Lizard Island Research Station for their support and friendship. We also thank the three anonymous referees for valuable comments in earlier versions of this manuscript. This study was supported by the Portuguese Foundation for Science and Technology-FCT (grant PTDC/MAR/105276/2008 awarded to MCS) and by the Swiss Science Foundation (SNF grant, awarded to RB). MCS was supported by the Project “Genomics and Evolutionary Biology”, co-financed by North Portugal Regional Operational Programme 2007/2013 (ON.2 – O Novo Norte), under the National Strategic Reference Framework (NSRF), through the European Regional Development Fund (ERDF).
Author information
Authors and Affiliations
Contributions
M.C.S. and J.P.M.M. designed the study. J.P.M.M. and J.R.P. collected the data. M.C.S. and J.P.M.M. analysed the data. M.C.S., J.P.M.M., R.B. and A.S.G. wrote the paper. All authors discussed results and commented on the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Messias, J., Paula, J., Grutter, A. et al. Dopamine disruption increases negotiation for cooperative interactions in a fish. Sci Rep 6, 20817 (2016). https://doi.org/10.1038/srep20817
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep20817
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.