Abstract
Coralline red algae are significant components of sea bottom and up to now considered as exclusively marine species. Here we present the first coralline alga from a freshwater environment, found in the Cetina River (Adriatic Sea watershed). The alga is fully adapted to freshwater, as attested by reproductive structures, sporelings and an inability to survive brackish conditions. Morphological and molecular phylogenetic analyses reveal the species belongs to Pneophyllum and is described as P. cetinaensis sp. nov. The marine-freshwater transition most probably occurred during the last glaciation. The brackish-water ancestor was preadapted to osmotic stress and rapid changes in water salinity and temperature. The particular characteristics of the karst Cetina River, such as hard water enriched with dissolved calcium carbonate and a pH similar to the marine environment, favoured colonization of the river by a marine species. The upstream advance and dispersal is facilitated by exceptionally pronounced zoochory by freshwater gastropods. Pneophyllum cetinaensis defies the paradigm of Corallinales as an exclusively marine group.
Similar content being viewed by others
Introduction
Coralline red algae (Corallinophycidae, Rhodophyta) are important components of marine ecosystems. They are ubiquitous from tropical regions to the poles, thriving from the intertidal to the lower boundaries of the euphotic zone1,2. Impregnated with calcium carbonate, they fill a paramount role as ecosystem bio-constructors, consolidating coral reef and coralligenous structures3, developing extensive maerl/rhodolith beds4, providing three-dimensional habitats, favouring the development of other benthic organisms5 and significantly contributing to carbonate deposition in shallow marine water (MW)6,7.
Coralline algae are today a topical subject for scientists interested in global change8,9,10,11. They are frequently used in paleoenvironmental reconstruction and as a biological datalogger for the reconstruction of past sea levels12, water temperature13,14, surface ocean salinity and freshwater balance15 and, as recently proposed, for seawater pH oscillations8.
Within the red algal phylum (Rhodophyta), with ca 7,100 living species, Corallinales are the third most species-rich group, with 725 described living taxa16. Although some coralline algae can be found in brackish environments17, a truly freshwater (FW) representative has never been reported so far, either as a fossil or as a living species. Like other important marine lineages such as echinoderms, corallines have been considered restricted to marine water, never making the evolutionary step into FW.
Here we present the first record of a strictly FW coralline algae, discovered in the Cetina River (Croatia), a karst river of the Adriatic Sea watershed (Figs 1 and 2), to which it seems to be strictly endemic. We assessed that it pertains to the marine genus Pneophyllum by both morphological and molecular phylogenetic analyses. We also discuss the processes which facilitated the rare evolutionary adaptation and tremendous habitat shift of a single algal species in a geologically recent time in light of the specific abiotic and biotic characteristics of the karst Cetina River.
Study area.
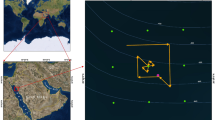
Cetina River with locations of Pneophyllum cetinaensis, including the type locality (a,b). Different aspects of the river in the canyon (c), the plain section (d) and the type locality (e). Pneophyllum cetinaensis in the type locality covers all available hard substrata, such as cobbles and pebbles (red arrow) and roots (white arrow) in shaded, shallow areas of the riverbed (f). Maps were created using Adobe In Design CS5 and Photoshop CS5 software and based on OpenStreetMap (https://www.openstreetmap.org/copyright).
Habitus of Pneophyllum cetinaensis.
Holotype (PC0145164) (a). Typically, P. cetinaensis develops as a crust on cobbles and pebbles (b,c). Extensive coverage in a shaded, shallow area in the type locality (d) where most gastropods are overgrown by P. cetinaensis, commonly with reproductive structures (arrow) (e).
Results
Species description
Pneophyllum cetinaensis Kaleb, Žuljević & V. Peña, sp.nov.
Phylum: Rhodophyta
Subclass: Corallinophycidae
Order: Corallinales
Family: Corallinaceae
Etymology: Cetinaensis from Cetina, name of the river where species is found.
Holotype: PC0145164 (Herbier Cryptogamique PC, Muséum National d´Histoire Naturelle, Paris, France, Fig. 2a)
Isotypes: PC0145165, PC0145166 and PC0145167, deposited at the National History Museum in Paris (PC). NHMS000566, NHMS000567, NHMS000568, deposited at the Natural History Museum in Split (NHMS). 600:ZAG;1:BOA, deposited at the Croatian Natural History Museum in Zagreb (CNHM). ZA39846, ZA39848, deposited at the Herbarium Croaticum at the University of Zagreb (ZA).
Date of collection: 27.08.2013.
Type locality: Otok Ljubavi (Island of Love), Cetina River, Croatia (43° 26.180’N - 16° 45.785’E).
Diagnosis
With the characteristics of Pneophyllum, it differs from other species in forming extensive and conspicuous crusts thickened and mostly multi-layered with flattened or curved branches and in having the pore canal of conceptacles simple, not surrounded by specialised cells. It also differs from any known coralline red algae in its ecology, being confined to a freshwater stream as opposed to the marine environment.
Description
Thalli are non-geniculate, attached ventrally to the substratum (cobbles, pebbles, gastropods and plant roots) (Fig. 2) by cell adhesion and forming sometimes extensive crusts up to 60 mm across or more (Fig. 2c,d). The thallus is encrusting or layered, arranged in superimposed flattened or curved fragile branches (Fig. 3c), each up to 145 μm thick. The branches are arranged horizontally and in most cases concentrically and exhibit a irregular whitish or pale pink margin (Fig. 2b). Living thallus is pink to violet with matt texture (Fig. 2). The thallus is developed from a spore germination disc with an eight-celled central element (Fig. 4).
SEM images of the holotype (H) (PC0145164) and the isotype (I) PC0145165.
Pneophyllum-type surface with lenticular epithallial cells (arrows) (H) (a); bistratose thallus with elongate initials (arrows) and one layer of epithallial cells (arrowheads) (H) (b); superimposed branches with peripheral region (p) composed of filaments perpendicular to the thallus surface (I) (c); sporangial conceptacle with small columella at the base of the chamber (arrow) (I) (d); spermatangial conceptacle showing a conical chamber (H) (e); female conceptacle showing an elliptical chamber (H) (f).
The pseudoparenchymatous thallus has a dorsiventral organisation and dimerous construction and can be bistratose or multi layered. The ventral region consists in a single layer of squarish cells (10–22 μm long × 6–15 μm in diameter) forming a filament more or less parallel to thallus surface. The peripheral region can consist only of the epithallial cell (3–5 μm × 6–9 μm) or is composed of filaments perpendicular to thallus surface, with one or more rectangular cells (6–14 μm × 7–12 μm) (Fig. 3b,c). The subepithallial initials are elongate ranging from 13 to 24 μm long and 5 to 12 μm in diameter. The thallus surface is Pneophyllum-type (SEM) with wide lenticular epithallial cells (3–6 μm long and 6–10 μm wide) (Fig. 3a).
Cells of the same filament are joined by primary pit-connections, whereas cells of adjacent filaments (other than epithallial cells and subepithallial initials) are connected laterally by cell fusions. Secondary pit-connections were not found. Trichocytes are common terminally on erect filaments.
Gametangial thalli monoecious with male and female conceptacles occurring in the same thallus. Male conceptacles are uniporate, flat or slightly protruding above surrounding thallus surface, roof 4–5 cells thick above chamber (30–43 μm), chambers conical 24–28 μm high × 83–88 μm in diameter (Fig. 3e). The floor of the conceptacle 6–8 cells below thallus surface. Pore sometimes fringed but without spout. Female conceptacle are uniporate, slightly raised or hemispherical with domed to elliptical chambers 85–93 μm high × 225–232 μm in diameter (Fig. 3f). The floor of the conceptacle 8 cells below thallus surface. Roof composed of 5–6 cells layers (42–64 μm). Pore simple, without papillae. Tetrasporangial conceptacles are uniporate, hemispherical and protruding, with elliptical chambers (60–77 μm high × 130–182 μm in diameter) and a small columella usually present at the base (Fig. 3d). The roof of the conceptacles is 3–5 layers thick (20–37 μm). The floor of the conceptacle chambers is 4–8 cells below the thallus surface. Tetrasporangia are zonate 100–110 μm × 60–64 μm. Old conceptacles never becoming buried within the thallus.
Morphologically, P. cetinaensis can readily be delimited from other Pneophyllum species known to occur in the European Atlantic and Mediterranean coasts (the generitype P. fragile, P. confervicola, P. limitatum, P. lobescens, P. subplanum, P. myriocarpum and P. zonale). Pneophyllum cetinaensis can form extensive and conspicuous crusts and is the only species with flattened or curved branches arranged in horizontally oriented layers. Moreover the thalli are thickened, mostly multi layered. Erect filaments are known to occur only in P. zonale, P. myriocarpum and in P. fragile. In P. zonale, the pore canal of tetra/bisporangial conceptales is surrounded by free, unicellular filaments while in P. myriocarpum the pore canal is surrounded by a hyaline collar. By contrast, P. fragile lacks such specialized structures in tetra/bisporangial conceptale pore canals; however, unlike P. cetinaensis, the spermatangial conceptacles are provided with a spout.
Molecular phylogenetic analyses
The phylogenetic analyses inferred from the plastid psbA gene resolved the FW coralline species P. cetinaensis within the genus Pneophyllum with strong support (89%/0.78 for ML and BI, respectively, Fig. 5). In our phylogenetic analyses, P. cetinaensis was resolved as a distinct lineage, but its exact position in the genus and the relationships with Atlantic and Mediterranean isolates was not resolved (Fig. 5, Supplementary Table S1).
Phylogenetic tree inferred from ML and BI analyses of the psbA sequences of Pneophyllum cetinaensis, Pneophyllum taxa recorded in the Atlantic Ocean and Mediterranean Sea (P. zonale, P. subplanum, P. limitatum are represented by their type collections) and genera from the orders Corallinales, Hapalidiales and Sporolithales.
Boostrap ML values >50% and posterior probabilities >0.50 from Bayesian inference are shown for each node. Members of the order Sporolithales were used as outgroup. Scale bar: 0.02 substitutions per site.
Distribution and ecology
Pneophyllum cetinaensis is, to the best of our knowledge, present only in the Cetina River (Croatia) and its tributary the Veliki Rumin. Despite intensive searching, the species was not found in the nearby Jadro and Žrnovnica rivers (15 km and 20 km from the mouth of the Cetina River, respectively; Fig. 1).
The Cetina River is a typical permanent karst river of the Adriatic Sea watershed located in a topographically complex karst landscape. It is 105 km long, with the main spring 382 m above sea level and a few short tributaries. The upper course flows across plains, the middle incises a deep canyon and the lower course runs through a valley. The canyon area has numerous waterfalls, with the 49 m-high Gubavica Waterfall being the highest. Most of the river basin’s bedrock is Cretaceous carbonate rock, mainly limestone. The riverbed ranges from rocky to a cover of cobbles, pebbles and sand. The river’s water quality is good except in small areas close to towns, where the quality is moderate with a certain degree of pollution18. The water can be characterized as hard to very hard (see Table 1 for the basic physicochemical parameters).
The natural hydrological regime of the Cetina River has changed significantly since the 1960s with the construction of hydroelectric power plants (HEPP) and artificial lakes (Fig. 1). Especially in the last 40 km (the type locality), the regime has moderated, showing less seasonal variation19. Since 1980, 90% of the water is transported via tunnels from the Prančevići artificial lake to Zakučac HEPP, near the river mouth (Fig. 1). The annual variation in water flow before building this HEPP was a minimum of 20 m3 s−1 in summer up to 200 m3 s−1 in winter. Due to water diversion, the mean annual discharge has now dropped from 100 m3 s−1 to an almost constant 10 m3 s−1 in the last 40 km of the river course19, except for short periods of intense rains. Discharge from the Prančevići artificial lake is estimated to be at least 8 m3 s−1 to satisfy the ecologically acceptable flow (biological minimum)19. At the type locality, a current speed of 20 cm s−1 was measured above the pebbled bottom and of 110 cm s−1 in moss-covered areas20.
Pneophyllum cetinaensis is present throughout almost the entire length of the Cetina River from 0 to 300 m above sea level, which is about 75 km from the river mouth (Fig. 1). The alga was not found upstream of the artificial Peruča Lake. It is probably not present in artificial lakes due to the high variation in water levels. In the estuary, it is found only in the shallowest water layer, down to around 50 cm deep, where there is no seawater influence. Pneophyllum cetinaensis develops in areas with either slow or fast water currents from 0 to 2 m deep (Fig. 2). Most commonly, it grows on the self-shaded sides of pebbles and not on substrata directly exposed to the sun. The area with the greatest algal development is the type locality, Otok ljubavi (Figs 1b,f and 2d). On the river margin (0–30 cm deep, 0–1 m from the shore), where water flow is not strong and the bed is shaded by a dense tree canopy (Fig. 1e), the alga nearly completely covers the available hard substratum (mostly cobbles, pebbles and plant roots) (Figs 1f and 2d) and can be found on most adult gastropods (Fig. 2e). More than 95% of the gastropods collected from cobbles and pebbles are Theodoxus fluviatilis fluviatilis (Linnaeus 1758). Algal crusts occurred on 96% of specimens of the latter species larger than 4 mm (n = 267), of which 40% had algae covering more than 50% of the shell surface. In gastropods smaller than 4 mm (n = 124), alga occurred on 39% of gastropods and always covering less than 50% of the shell surface.
Discussion
Biome transitions are rare macroevolutionary events with profound consequences for terrestrial21, FW22 and marine habitats23. The high diversity of FW species living today is a result of the diversification of a small number of successful ancestral lineages that invaded FW from the marine or terrestrial biomes in ancient geological times21.
In spite of the direct contact between MW and FW, invasions of marine species into FW are infrequent events and restricted to a small number of lineages. Of the 31 animal phyla found in the sea, 11 are exclusively marine and have never successfully invaded FW habitats22. The rarity of transition and consequences for diversity can be observed in almost every FW animal group. It is estimated, for instance, that just one ancestral lineage of marine sponge has resulted in today’s diversity of around 300 FW sponges24. Similarly, less than 40 MW lineages of gastropods have produced today’s total diversity of 4,000 described FW species25. Even microbes have only infrequently crossed the marine-FW boundary and most of those transitions occurred long ago in evolutionary time despite large population sizes, high genetic diversity and a good potential for long-distance dispersal26.
The diversity of FW red algae is surprisingly small compared to MW. There are only about 200 FW species accounting for 3% of red algal diversity16,27. The majority of FW red alga belong to the order Batrachospermales, which is exclusively found in FW27. Other FW species are scattered across the red algal tree of life, classified in species-poor orders mostly confined to FW environments27,28 (Fig. 6). They are found at very few locations29, often surprisingly disjunctive30 and in specific, sometimes extreme, environments31. Except for two species of the predominantly marine genus Hildenbrandia, none of the strictly FW red algae have close relatives in the marine biome, indicating that their ancestral lineages invaded FW in ancient evolutionary time.
Phylogenetic relationships of red algae.
Branches showing the currently number of genera and species estimated from AlgaeBase16. FW genera are listed under the leaf showing the major lineages among the Rhodophyta. Genera in blue include exclusively FW species.
There are only 15 species (belonging to the order Ceramiales) which have bridged the MW-FW boundary, but even then they still live in marine habitats where they exclusively reproduce27. Ceramiales does not contain a single strictly FW species. Together with FW representatives of Hildenbrandia, they can be considered as evolutionary secondary immigrants from the sea31,32, sharing common characteristics such as the absence of sexual reproduction in FW, while vegetative reproduction is present only by means of gemmae in Hildenbrandia27.
Pneophyllum cetinaensis, which we discovered to be strictly endemic to the Cetina River (Croatia) (Figs 1 and 2), is the first known FW coralline alga. It is thus a member of a widely distributed, highly species-rich and diversified order that is immensely important in the geological record and up to now considered an exclusively marine group of species33 (Fig. 6). Its marine genealogy places P. cetinaensis as a secondary FW immigrant32. The inability to live in the estuary of the Cetina River where there is at least some influence of diluted seawater, along with the development of sexual and asexual reproductive structures (conceptacles, Figs 2e,3d–f and 4), vouch for its full adaptation to FW conditions.
A scenario invoked to explain many FW invasions is the landlocking of marine species as a result of sea-level changes at different spatial and temporal scales, with the subsequent dilution of seawater. Most marine species would vanish in such new conditions and only on very rare occasions would they adapt to the new environment34. The most serious obstacles preventing invasion into FW are the regulation of osmotic pressure, ionic concentration, pH level, low temperature, constant runoff, food resources, competition and available living space26,34. All of these hurdles were surmounted by P. cetinaensis in recent geological history, probably due to the preadaptation of its brackish-water ancestor and the specific characteristics of the karst Cetina River.
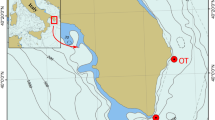
About 120,000 years ago, global sea levels began descending from their maximum level, which was slightly higher than present-day levels35, to a minimum 20,000 years ago, which corresponds to the last glacial maximum when sea levels were about 120 m below present-day levels36. This was not a continuous drawdown, as there were many reversals in the descending trend37. During the last glacial maximum, the Cetina River had to cross four depressions (today at 60 to 90 m below sea level) before reaching the Adriatic Sea38 (Fig. 7). In periods when the depressions were part of the Cetina River estuary and global sea level was descending or ascending, the depressions were probably inhabited by brackish species.
The Adriatic coast in the area of the Cetina River mouth.
Recent situation (a) and the Cetina River about 20,000 years ago (b) with sea level at −115 m. The Cetina River ran through four paleolakes (I to IV), filling the depressions before reaching the Adriatic Sea. Under a scenario of sea-level oscillations, these paleo-lakes repeatedly became part of a paleo-estuary, most likely facilitating adaptation to FW conditions and peripatric speciation of the ancestor of Pneophyllum cetinaensis. Grey lines indicate the present-day shoreline. Maps were created using Adobe In Design CS5 and Photoshop CS5 software and based on OpenStreetMap (https://www.openstreetmap.org/copyright). Cetina paleo-course is drawn based on the data from38.
Pneophyllum spp. were typical of these brackish inhabitants, as they are today in the Adriatic lagoons, estuaries and deltas where they flourish as inconspicuous epiphytes on seagrass39. As an inhabitant of a paleo-estuary, the ancestor of P. cetinaensis was preadapted to osmotic stress and rapid changes in water salinity and ambient temperature that rapidly oscillated beyond the thresholds of typical shallow-marine habitats. A small enhancement in osmotic regulation40, together with the common r-strategist and opportunistic nature of Pneophyllum spp., would result in offspring with higher fitness, equipped for full FW colonization. Specimens may have survived on the shallowest estuary bottom (mainly occupied by FW) and subsequently spread upstream. However, survival and spreading of Pneophyllum could not happen in just any type of river; rather, the specific characteristics of the karst Cetina River may have largely determined the favourable outcome.
For proper growth and cell wall calcification, Pneophyllum, like other coralline algae, requires calcium carbonate and magnesium, which are sufficiently present in seawater, but not in every type of river. As most of the Cetina River catchment lies on carbonate rocks, mainly limestone19, its water is hard and enriched in dissolved calcium carbonate and magnesium ions (Table 1), essential for the development of corallines. Moreover, high levels of ions in the Cetina River indicated by high conductivity (Table 1), made osmoregulation easier for marine species. Difficulties in osmoregulation during the transition from MW to soft water in compared with hard water water have been observed in the brackish water flatworm41, while alleviation of osmotic stress thanks to high concentrations of ions has been suggested as important for the establishment of a FW population of the primarily marine red alga Polysiphonia subtilissima Montagne42.
The calcified tissue of corallines cannot develop in an acidic environment43, such as in soft-water rivers or acid tropical rivers with dissolved fulvic and humic substances44. In particular a significant reduction in epiphytic coralline algal cover with increasing acidification due to natural CO2 vents has been reported by45. Therefore, the hard water of karst, carbonate rivers with pH values similar to the marine environment, as is the case of the Cetina River (median pH of 8.12), are the only potential ones for coralline algae invasion.
Unlike most FW invaders, P. cetinaensis did not encounter competitors and predators in the Cetina River21. Cobbles and pebbles in the Cetina River bed are mostly uncolonized by other macroalgae and mosses, which was probably also the case during the early invasion of P. cetinaensis. This substratum therefore provided a favourable, vacant habitat for the alga to occupy. Calcified cell walls give coralline algae excellent protection from herbivory, which promoted the diversification of specialized grazers in the sea46. The absence of specialized grazers favoured the establishment of P. cetinaensis in the Cetina River, where common river herbivores, mainly gastropods, amphipods and insects47 cannot feed on calcified crusts. However, by feeding on epiphytes overgrowing P. cetinaensis, those herbivores probably perform the same beneficiary function as marine herbivores: cleaning coralline algal surfaces of fast-growing epiphytic species48.
Our observation suggests that FW gastropods, especially T. fluviatilis fluviatilis, have one additional, peculiar function in the biology of P. cetinaensis. This alga, like other red algae, does not have a vagile life stage and is constantly facing washout in the river stream. As most adult gastropods (more than 95% in the type locality) are overgrown by crusts of P. cetinaensis, commonly with developed reproductive organs (Fig. 2e), they serve as the main dispersal vector of P. cetinaensis through the river. In areas with no algal encrustation on pebbles, such as the slightly eutrophic part of the river in the vicinity of the town of Trilj (Fig. 1a), the algae can be found on gastropods, indicating an affinity of spores to attach onto gastropod shells and/or gastropod mobility. Most gastropod species can actively move upstream from 0.3 to 1.0 km per year49. Over periods of tens to hundreds of years, benthic molluscs could have dispersed P. cetinaensis over a distance of 75 km along the river, even surmounting waterfalls (up to 50 m high). Such pronounced malacochory is also benefited by one more peculiarity of karst rivers: a high predominance of gastropods among benthic macroinvertebrates50. In the area of the type locality (Fig. 1), gastropods represent around 40% of the total number of benthic macroinvertebrates, with a maximum density of almost 4,500 specimens m−2 47.
The fact that other karst rivers close to the Cetina River share similar FW gastropod fauna50, but lack P. cetinaensis despite the river’s proximity and pronounced transportation by gastropods, supports our hypothesis of a geologically recent biome transition and estimated onset of the Cetina River invasion within the last 120,000 years.
Pneophyllum is a widely distributed genus encompassing 18 species currently accepted taxonomically16. Seven species have been reported from the European Atlantic and Mediterranean coasts. All these taxa are represented in our molecular analyses by DNA sequences from type material (P. lobescens, P. limitatum and P. subplanum) or from historical collections available for molecular studies. The exception is P. zonale (a species described from the Atlantic French coast growing on a small piece of glass51), for which all attempts to obtain molecular data from the isolectotype failed. The use of type specimens as taxonomical references confirms that the novel species described herein has not been formally described among European Pneophyllum; furthermore, it highlights the presence of additional cryptic species within the genus. It is obvious that further systematic research is required to assess the diversity within the genus Pneophyllum, for which current taxonomical features for species determination might be insufficient.
The molecular data places P. cetinaensis in the Pneophyllum clade and also negates close phylogenetic relation to any other recognized European species (Fig. 5). The results suggest many intriguing questions for future studies: what happened with the marine/brackish water ancestor of P. cetinaensis? Does the ancestral species still have marine/brackish water descendants or have they vanished?
Taking into account our present insufficient knowledge of the diversity of the Pneophyllum clade and that P. cetinaensis does not have close relatives among described marine Pneophyllum species, we can suppose that a marine/brackish species closely related to P. cetinaensis might still be found in the Adriatic/Mediterranean Area.
Pneophyllum cetinaensis defies the paradigm of coralline algae being exclusively marine species. The fact that coralline algae can exist in a FW habitat will probably open new discussions and produce significant impacts in different fields. Standard textbook concepts consider coralline algae as paleoenvironmental indicators, stating that they are “commonly adapted to normal marine salinities”52, although it is acknowledged that they can tolerate brackish to hypersaline conditions53.
Changes in sea levels throughout the Pleistocene resulted in several well-documented peripatric populations of marine species isolated in marine lakes, which serve as a suitable subject for the research of evolutionary processes54. Pneophyllum cetinaensis also has a large potential to become a model organism to study evolution through peripatric speciation. Furthermore, as a species that crossed the border between the marine and freshwater biomes, it is of particular interest for studying the ecophysiological mechanisms and underlying genomic characteristics of the transition. The Cetina River is in a karst region of the Balkan Peninsula that is unique due to numerous endemic species in FW, sea, land and especially in caves. During the Pleistocene glaciation, this area (along with the Iberian Peninsula and the Apennines) was a major refugium for European species as it remained largely unaffected by glaciers. Consequently, many species survived glaciations and due to the karst geology, they remained isolated and evolved as endemic species55. The Adriatic basin has more than 40 endemic FW fish species, many endemic to only one river (sometimes very short). In addition, there is also a notable quantity of species that would never be expected to occupy the habitat in which they are found. Examples of such unexpected species in the Balkan area are the only known cave sponge Eunapius subterraneus, the rare stygobitic cave leech Croatobranchus mestrovi, the unique FW cave-dwelling tube worm Marifugia cavatica, the only underground bivalve in the world Congeria kusceri and the only cave-dwelling chordate species found in Europe Proteus anguinus56. Pneophyllum cetinaensis, a unique FW coralline alga, takes its place among these species, confirming that the Balkan Peninsula is a hot spot for endemism and peculiar species, many probably yet to be discovered. Although finding a new species today is not unusual, the discovery of a FW coralline alga is quite surprising.
Methods
Field observations and sampling
Following our initial observation of Pneophyllum cetinaensis in 2013, we inspected the Cetina River at numerous locations from the river mouth to the river spring, including tributaries (Fig. 1). The aim was to collect data on distribution, as well as biotic and abiotic elements that might serve to characterize the biology and ecology of the species. Samples were collected for morphological and molecular analyses on the type locality (Fig. 1, Supplementary Table S1). Data on physical and chemical parameters were obtained from Hrvatske vode (the legal entity for water management in Croatia). The dataset includes measurements from the start of 2009 till the end of 2013 at two gauging stations covering the lower (Radmanove mlinice station) and upper (Cetina station) river courses (Fig. 1). Sampling was basically made on monthly interval. Radmanove mlinice station is in the type locality of P. cetinaensis.
We studied the frequency of algal development on gastropods by sampling numerous snail specimens from randomly collected cobbles and pebbles in the type locality (Island of Love) (Fig. 1). The presence and abundance of algal crusts was assessed under stereo-microscope taking into account gastropod species, size (larger or smaller than 4 mm) and coverage by the alga (under or above 50% of the shell surface). Two nearby karst rivers, the Jadro and Žrnovnica, were checked thoroughly for possible alga occurrences (Fig. 1).
Morphological analyses
Specimens were air dried and stored in silica gel. Fragments were mounted on aluminium stubs and coated with gold/palladium (with S150 Sputter Coater, Edwards, Crawley, UK) prior to viewing with a LEICA Steroscan 430i (Cambridge, UK) at 20 kV. For the study of the reproductive cycle, fresh samples were collected and stored in 10 L dark plastic containers and transported to the laboratory within 12 hours, along with FW stored in several 25 L containers. The culture was set up in a thermoconstant room at 14 °C; five marked microscopy slides were placed at the bottom of the aquarium for the settlement of spores.
Molecular study
We studied 11 specimens of P. cetinaensis, recent collections of Pneophyllum taxa in the Mediterranean and Atlantic Europe, as well as type species (P. fragile, P. lobescens, P. limitatum, P. subplanum, P. zonale) and other important historical collections of Pneophyllum species collected over the last two centuries and deposited at the Natural History Museum (BM), the Muséum National d´Histoire Naturelle (PC) and at the Norwegian University of Science and Technology (TRH) (see Supplementary Table S1 online). Except for P. cetinaensis, the rest of the collections were collected in marine areas, intertidally or subtidally, growing as epiliths on stones or glass but also as epiphytes on seaweeds and seagrasses (see Supplementary Table S1 online).
DNA extraction, PCR and PCR product sequencing. Specimen surfaces without epiphytes were selected under a stereomicroscope and ground with a 2 mm drill bit for DNA extraction. Genomic DNA was extracted using a NucleoSpin® 96 Tissue kit (Macherey-Nagel, GmbH and Co. KG, Germany) following the manufacturer’s protocol. For P. cetinaensis, type specimens and historical collections, we employed the QIAamp® DNA Micro Kit (Qiagen S.A.S., France) following the manufacturer’s protocol for tissues. The plastid gene encoding the D1 protein of photosystem II (psbA) was amplified in one reaction using the pairs of primers psbA-F1/psbA-R2 or psbA-F1/psbA600R57 following the thermal profile58. The PCR reaction mixture followed59, except for the amplification of type specimens and historical collections for which the DNA template was not diluted. PCR products were purified and sequenced by Eurofins (Eurofins Scientific, France). Voucher specimens for P. cetinaensis and recent collections of Pneophyllum were deposited in the Muséum National d´Histoire Naturelle (PC), Natural History Museum Split (NHMS), Herbarium Croaticum - University of Zagreb (ZA) and the Croatian Natural History Museum (CNHM)60. Sequences were submitted to the Barcode of Life Data Systems (project “NGCOR”, BOLD, http://www.boldsystems.org and GenBank (accession numbers listed in Supplementary Table S1 online). For the molecular analyses, publicly available sequences of Pneophyllum were included, as well as sequences from other genera of the orders Corallinales, Hapalidiales and Sporolithales (see Supplementary Table S1 online).
Molecular analyses. Models of sequence evolution were estimated using the Akaike Information Criterion (AIC) and the Bayesian Information Criterion (BIC) obtained in jModeltest 2.1.361. Maximum Likelihood analysis for the psbA alignment was performed under a generalized time-reversible with gamma+invariant sites heterogeneity model (GTR+G+I) and the bootstrap consisted of 1,000 replicates. The psbA alignment comprised 44 haplotype sequences ranging from 376 to 851 bp, with 294 variable sites. The alignment did not include either the holotype fragment of P. fragile or the isolectotype of P. zonale, for which DNA sequences could not be obtained.
Additional Information
How to cite this article: Žuljević, A. et al. First freshwater coralline alga and the role of local features in a major biome transition. Sci. Rep. 6, 19642; doi: 10.1038/srep19642 (2016).
References
Johansen, H. W. Coralline Algae, a First Synthesis. (CRC Press, Boca Raton, Florida, 1981).
Littler, M. M., Littler, D. S., Blair, S. M. & Norris, J. N. Deepest known plant life discovered on an uncharted seamount. Science 227, 57–59, 10.1126/science.227.4682.57 (1985).
Goreau, T. F. Calcium carbonate deposition by coralline algae and corals in relation to their roles as reef-builders. Ann. NY Acad. Sci. 109, 127–167, 10.1111/j.1749-6632.1963.tb13465.x (1963).
Foster, M. S. Rhodoliths: Between rocks and soft places. J. Phycol. 37, 659–667, 10.1046/j.1529-8817.2001.00195.x (2001).
Steneck, R. S. The ecology of coralline algal crusts: convergent patterns and adaptative strategies. Annu. Rev. Ecol. Syst. 17, 273–303, 10.1146/annurev.es.17.110186.001421 (1986).
Bosence, D. & Wilson, J. Maerl growth, carbonate production rates and accumulation rates in the northeast Atlantic. Aquat. Conserv. Mar. Freshwater Ecosyst. 13, S21–S31, 10.1002/aqc.565 (2003).
Basso, D. Carbonate production by calcareous red algae and global change. Geodiversitas 34, 13–33, 10.5252/g2012n1a2 (2012).
Cusack, M., Kamenos, N. A., Rollion-Bard, C. & Tricot, G. Red coralline algae assessed as marine pH proxies using B-11 MAS NMR. Sci. Rep. 5, 10.1038/srep08175 (2014).
Rahman, M. A. & Halfar, J. First evidence of chitin in calcified coralline algae: new insights into the calcification process of Clathromorphum compactum. Sci. Rep. 4, 10.1038/srep06162 (2014).
Teichert, S. Hollow rhodoliths increase Svalbard’s shelf biodiversity. Sci. Rep. 4, 10.1038/srep06972 (2014).
Kuffner, I. B., Andersson, A. J., Jokiel, P. L., Rodgers, K. S. & Mackenzie, F. T. Decreased abundance of crustose coralline algae due to ocean acidification. Nature Geosci. 1, 114–117, 10.1038/ngeo100 (2008).
Braga, J. C. & Aguirre, J. Coralline algae indicate Pleistocene evolution from deep, open platform to outer barrier reef environments in the northern Great Barrier Reef margin. Coral Reefs 23, 547–558, 10.1007/s00338-004-0414-x (2004).
Kamenos, N. A., Cusack, M. & Moore, P. G. Coralline algae are global palaeothermometers with bi-weekly resolution. Geochim. Cosmochim. Acta 72, 771–779, 10.1016/j.gca.2007.11.019 (2008).
Halfar, J. et al. Coralline algal growth-increment widths archive North Atlantic climate variability. Palaeogeogr. Palaeoclimatol. Palaeoecol. 302, 71–80, 10.1016/j.palaeo.2010.04.009 (2011).
Hetzinger, S. et al. Coralline algal Barium as indicator for 20th century northwestern North Atlantic surface ocean freshwater variability. Sci. Rep. 3, 10.1038/srep01761 (2013).
Guiry, M. D. & Guiry, M. D. AlgaeBase. World-wide electronic publication, National University of Ireland, Galway. < http://www.algaebase.org> (2015), Accessed 22/04/2015.
Falace, A., Curiel, D. & Sfriso, A. Study of the macrophyte assemblages and application of phytobenthic indices to assess the Ecological Status of the Marano-Grado Lagoon (Italy). Mar. Ecol. Evol. Persp. 30, 480–494, 10.1111/j.1439-0485.2009.00300.x (2009).
Ćuk, R. et al. Saprobic status of running waters in Croatia based on benthic macroinvertebrates. Nat. Croat. 19, 389–406 (2010).
Bonacci, O. & Roje-Bonacci, T. The influence of hydroelectrical development on the flow regime of the karstic river Cetina. Hydrol. Processes 17, 1–15, 10.1002/hyp.1190 (2003).
Božak, K. Ecological conditionality of the gastropod communities (Mollusca, Gastropoda) in the Cetina River Master thesis, Faculty of Science, University of Zagreb, (2010).
Vermeij, G. J. & Dudley, R. Why are there so few evolutionary transitions between aquatic and terrestrial ecosystems? Biol. J. Linn. Soc. 70, 541–554 (2000).
Balian, E. V., Segers, H., Leveque, C. & Martens, K. The freshwater animal diversity assessment: An overview of the results. Hydrobiologia 595, 627–637, 10.1007/s10750-007-9246-3 (2008).
Carrete Vega, G. & Wiens, J. J. Why are there so few fish in the sea? Proc. R. Soc. B. 279, 2323–2329, 10.1098/rspb.2012.0075 (2012).
Itskovich, V. B. et al. Monophyletic origin of freshwater sponges in ancient lakes based on partial structures of COXI gene. Hydrobiologia 568, 155–159, 10.1007/s10750-006-0320-z (2006).
Strong, E. E., Gargominy, O., Ponder, W. F. & Bouchet, P. Global diversity of gastropods (Gastropoda; Mollusca) in freshwater. Hydrobiologia 595, 149–166, 10.1007/s10750-007-9012-6 (2008).
Logares, R. et al. Infrequent marine-freshwater transitions in the microbial world. Trends Microb. 17, 414–422, 10.1016/j.tim.2009.05.010 (2009).
Kumano, S. Freshwater Red Algae of the World. 375 (Biopress Ltd., Bristol, 2002).
Muller, K. M., Sherwood, A. R., Pueschel, C. M., Gutell, R. R. & Sheath, R. G. A proposal for a new red algal order, the Thoreales. J. Phycol. 38, 807–820, 10.1046/j.1529-8817.2002.01055.x (2002).
Carlile, A. L. & Sherwood, A. R. Phylogenetic affinities and distribution of the Hawaiian freshwater red algae (Rhodophyta). Phycologia 52, 309–319, 10.2216/12-097.1 (2013).
Wehr, J. D., Stancheva, R., Truhn, K. & Sheath, R. G. Discovery of the rare freshwater brown alga Pleurocladia lacustris (Ectocarpales, Phaeophyceae) in California streams. West. N. Am. Naturalist 73, 148–157, 10.3398/064.073.0204 (2013).
Kwandrans, J. & Eloranta, P. Diversity of freshwater red algae in Europe. Oceanol. Hydrobiol. Stud. 39, 161–169, 10.2478/v10009-010-0015-7 (2010).
Skuja, H. Comments on fresh-water rhodophyceae. Bot. Rev. 4, 665–676 (1938).
McCoy, S. J. & Kamenos, N. A. Coralline algae (Rhodophyta) in a changing world: integrating ecological, physiological and geochemical responses to global change. J. Phycol. 51, 6–24, 10.1111/jpy.12262 (2015).
Lee, C. E. & Bell, M. A. Causes and consequences of recent freshwater invasions by saltwater animals. Trends Ecol. Evol. 14, 284–288 (1999).
Blanchon, P., Eisenhauer, A., Fietzke, J. & Liebetrau, V. Rapid sea-level rise and reef back-stepping at the close of the last interglacial highstand. Nature 458, 881–U886, 10.1038/nature07933 (2009).
Yokoyama, Y., Lambeck, K., De Deckker, P., Johnston, P. & Fifield, I. K. Timing of the Last Glacial Maximum from observed sea-level minima (vol 406, pg 713, 2000). Nature 412, 99–U19, 10.1038/35083629 (2001).
Rohling, E. J. et al. Sea-level and deep-sea-temperature variability over the past 5.3 million years. Nature 508, 477–482, 10.1038/nature13230 (2014).
Sikora, M., Mihanović, H. & Vilibić, I. Paleo-coastline of the Central Eastern Adriatic Sea and Paleo-Channels of the Cetina and Neretva rivers during the last glacial maximum. Acta Adriat. 55, 3–18 (2014).
Bressan, G. & Babbini, L. Corallinales del mar Mediterraneo: guida alla determinazione. S.I.B.M. 10, 1–237 (2003).
Dittami, S. M. et al. Towards deciphering dynamic changes and evolutionary mechanisms involved in the adaptation to low salinities in Ectocarpus (brown algae). Plant J. 71, 366–377, 10.1111/j.1365-313X.2012.04982.x (2012).
Potts, W. T. W. & Parry, G. Osmotic and Ionic Regulation in Animals. (Pergamon, London; Macmillan, New York, 1964).
Lam, D. W., Eugenia Garcia-Fernandez, M., Aboal, M. & Vis, M. L. Polysiphonia subtilissima (Ceramiales, Rhodophyta) from freshwater habitats in North America and Europe is confirmed as conspecific with marine collections. Phycologia 52, 156–160, 10.2216/12-085.1 (2013).
McCoy, S. J. & Ragazzola, F. Skeletal trade-offs in coralline algae in response to ocean acidification. Nature Clim. Change 4, 719–723, 10.1038/nclimate2273 (2014).
Horbe, A. M. C. & da Silva Santos, A. G. Chemical Composition of Black-Watered Rivers in the Western Amazon Region (Brazil). J. Braz. Chem. Soc. 20, 1119–1126 (2009).
Martin, S., Rodolfo-Metalpa, R., Ransome, E., Rowley, S., Buia, M. C., Gattuso, J. P. & Hall-Spencer, J. Effects of naturally acidified seawater on seagrass calcareous epibionts. Biology Letters 4, 689–692, 10.1098/rsbl.2008.0412 (2008).
Steneck, R. S. Escalating herbivory and resulting adaptive trends in calcareous algal crusts. Paleobiology 9, 44–61 (1983).
Vučković, I. et al. Composition and structure of benthic macroinvertebrate communities in the Mediterranean karst river the Cetina and its tributary the Ruda, Croatia. Nat. Croat. 18, 49–82 (2009).
Steneck, R. S. in 8th International Coral Reef Symposium Proceedings of the 8th International Coral Reef Symposium Vol. 1 (eds H. A. Lessios & I. G. Macintyre ) 695–700 (Smithsonian Tropical Research Institute, Panama, 1997).
Kappes, H. & Haase, P. Slow, but steady: dispersal of freshwater molluscs. Aquat. Sci. 74, 1–14, 10.1007/s00027-011-0187-6 (2012).
Rađa, B. & Puljas, S. Do Karst Rivers “deserve” their own biotic index? A ten years study on macrozoobenthos in Croatia. Int. J. Speleol. 39, 137–147 (2010).
Chamberlain, Y. M. Studies in the Corallinaceae with special reference to Fosliella and Pneophyllum in the British Isles. Bull. br. Mus. nat. Hist. Bot. 11, 291–463 (1983).
Flügel, E. Microfacies of Carbonate Rocks. Analysis, Interpretation and Application. (Springer-Verlag Berlin Heidelberg, 2004).
Braga, J. C. & Riding, R. in Encyclopedia of Geology (eds R. C. Selley, L. R. M. Cocks, & I. R. Plimer ) 428–436 (Elsevier, 2004).
Dawson, M. N. & Hamner, W. M. Rapid evolutionary radiation of marine zooplankton in peripheral environments. P.N.A.S. 102, 9235–9240, 10.1073/pnas.0503635102 (2005).
Zanella, D., Mihaljevic, Z., Mrakovcic, M. & Caleta, M. Ecology and diet of the endemic Telestes ukliva (Cyprinidae) in the Cetina River system, Croatia. Cybium 33, 97–105 (2009).
Kučinić, M. et al. An overview of the cave and interstitial biota of Croatia. Nat. Croat. 11, UDK 575.856:551.442(497.5) (2002).
Yoon, H. S., Hackett, J. D. & Bhattacharya, D. A single origin of the peridinin- and fucoxanthin-containing plastids in dinoflagellates through tertiary endosymbiosis. P.N.A.S. 99, 11724–11729, 10.1073/pnas.172234799 (2002).
Bittner, L. Phylogénie des Corallinales (Rhodophyta) et analyse de leur diversité génétique dans le Pacifique Sud Doctoral thesis, Museum National d´Histoire Naturelle, Paris, (2009).
Peña, V. et al. An integrative systematic approach to species diversity and distribution in the genus Mesophyllum (Corallinales, Rhodophyta) in Atlantic and Mediterranean Europe. Eur. J. Phycol. 50, 20–36, 10.1080/09670262.2014.981294 (2015).
Thiers, B. Index Herbariorum: A global directory of public herbaria and associated staff. New York Botanical Garden’s Virtual Herbarium. < http://sweetgum.nybg.org/ih/> (2015), Accessed 02/05/2015.
Darriba, D., Taboada, G. L., Doallo, R. & Posada, D. jModelTest 2: more models, new heuristics and parallel computing. Nat. Methods. 9, 772–772 (2012).
Acknowledgements
AŽ, IC, MD and BA acknowledge funding support from the Croatian Ministry of Science. AF and SK acknowledge research support by M.I.U.R. (Italian Ministry for Education, Universities and Research) through a P.R.I.N. 2011 Project (Coastal bioconstructions: structure, function and management). VP acknowledges support by the postdoctoral program I2C (Xunta de Galicia) and projects from Action Transversale du Muséum National d’Histoire Naturelle (“Taxonomie moléculaire: DNA Barcode et gestion durable des collections”, “Biodiversité actuelle et fossile. Crises, stress, restaurations et panchronisme: le message systématique” and “Emergences”). We are grateful to Jo Wilbraham (BM), Tommy Presto and Kristian Hassel (TRH) for the loan of type material. We gratefully acknowledge provision of some environmental data from Hrvatske vode - Legal entity for water management in Croatia. We are grateful to C. Lauring and I. Čatić for editing the English text. AŽ thanks to T. Domazet Lošo (IRB Zagreb) and M. Kučinić (PMF Zagreb) for fruitful discussions and useful suggestions.
Author information
Authors and Affiliations
Contributions
This work is conceived by A.Ž. and S.K.; S.K., A.F. and F.V. undertook the morphological analysis; V.P. and L. Le G. contributed to phylogenetic analyses and the study of type material; A.Ž., M.D., I.C. and B.A. contributed to field research; A.Ž., M.D., I.C., B.A., S.K., A.F. contributed to spore cultivation; A.Ž., S.K., V.P. and A.F. wrote the manuscript with input from all coauthors. O.De C. and J.C.B. revised the final version. All authors contributed equally in discussing ideas and data interpretation.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Žuljević, A., Kaleb, S., Peña, V. et al. First freshwater coralline alga and the role of local features in a major biome transition. Sci Rep 6, 19642 (2016). https://doi.org/10.1038/srep19642
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep19642
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.