Abstract
Although nearly one fifth of all human cancers have an infectious aetiology, the causes for the majority of cancers remain unexplained. Despite the enormous data output from high-throughput shotgun sequencing, viral DNA in a clinical sample typically constitutes a proportion of host DNA that is too small to be detected. Sequence variation among virus genomes complicates application of sequence-specific and highly sensitive, PCR methods. Therefore, we aimed to develop and characterize a method that permits sensitive detection of sequences despite considerable variation. We demonstrate that our low-stringency in-solution hybridization method enables detection of <100 viral copies. Furthermore, distantly related proviral sequences may be enriched by orders of magnitude, enabling discovery of hitherto unknown viral sequences by high-throughput sequencing. The sensitivity was sufficient to detect retroviral sequences in clinical samples. We used this method to conduct an investigation for novel retrovirus in samples from three cancer types. In accordance with recent studies our investigation revealed no retroviral infections in human B-cell lymphoma cells, cutaneous T-cell lymphoma or colorectal cancer biopsies. Nonetheless, our generally applicable method makes sensitive detection possible and permits sequencing of distantly related sequences from complex material.
Similar content being viewed by others
Introduction
It is estimated that almost one fifth (18.6%) of all cancers in humans have an underlying infectious aetiology1. Among these are important viral infections such as Epstein-Barr virus (EBV), hepatitis B and C virus (HBV and HCV, respectively) and most notably human papillomaviruses (HPV). Retroviruses are also involved in human cancers. Human T-lymphotropic virus type 1 (HTLV-1) causes adult T-cell leukaemia/lymphoma, whereas HTLV-2 has been associated with cases of myelopathy but its relation to cancer remains controversial2. Several animal retroviruses likewise cause lymphoma, leukaemia or other lympho-proliferative diseases in birds and mammals3. It is conceivable that undiscovered retroviruses might be involved in human cancers or lympho-proliferative diseases. The detection of such viruses is challenging.
Sequence variation is one of the main challenges in virus discovery. Extensive variation in nucleotide sequence complicates virus family classification (e.g. Picornaviridae). This applies also to most retroviruses that show considerable sequence variability. HIV-1 inter-subtype variation exceeds 35% in the variable env gene4 and >10% in the relatively conserved gag and pol genes5. Additional sequence variation between virus species is contributed by the presence of additional species-specific genes.
The proportion of viral nucleic acids in a cancer sample is usually very small as compared to host-derived genetic material. Firstly, retroviral genomes rarely exceed 10–12 kb and hence constitute a minor fraction of the genome of the infected host cell. Secondly, the infected cell type may constitute only a small fraction of the sample and thirdly, the infected cells may contain a relatively low number of viral genome copies. In Kaposi’s sarcoma lesions the Human Herpes virus 8-positive spindle cells constitute only a fraction of all atypical cells. Likewise, retrovirus genomes in humans (e.g. HTLV-1 or HIV-1) are typically present in infected individuals as single integrated proviral copies in minor fractions of nucleated cells in peripheral blood.
Sensitive detection of unknown viral sequences can be undertaken by high-throughput sequence-independent shotgun sequencing. However, because of the quantitative disproportion between viral and host genomic material, no more than a few viral sequence reads can be expected per million reads from host DNA. The proportion of viral nucleic acids can be greatly enriched by mechanical and enzymatic procedures that reduce the host genetic material6,7,8 combined with (random) amplification of the capsid-protected viral metagenome9,10. These methods are not feasible for investigation of integrated proviral DNA or episomal latent viral nucleic acids. Instead, target enrichment by hybridization (or target capture) can be performed; either in-solution or on solid-surface arrays or beads.
Target capture has been applied to diagnostics11, array analysis of virus12,13, or SNP analysis14 and used for enrichment of high-throughput sequencing libraries15,16,17. Most methods are dependent on stringent reaction conditions for discrimination between correct target and competing irrelevant sequences with varying similarity. Kane et al. established that cross hybridization may happen if nucleotide sequence similarity exceeds around 75%18, unless carefully controlled19. Matching stretches of as little as 12–15 complementary nucleotides are sufficient to mediate unspecific cross-hybridization of 50-bp oligonucleotides18,19. The risk of cross-hybridization has prompted researchers and manufacturers to maximize stringency during capture, including tightly controlled reaction conditions involving denaturing compounds (e.g. formamide) and optimized temperatures.
Where sequence variation poses a challenge, highly specific methods, such as PCR, may be applied with low stringency. For example, lowering of the annealing temperature, inclusion of promiscuously annealing nucleotides (e.g. inosine), or increased MgCl2 concentrations may decrease PCR specificity and enable amplification of variant sequences. Capture probes are longer than standard PCR primers and have been used for solid-surface microarray detection of viral sequences13. Similarly, we hypothesized that the conditions in capture enrichment may be tweaked to allow sufficient cross-hybridization to recover genomic material from unknown viruses from sequencing libraries prepared from patient sample material.
We have explored the use of in-solution target enrichment in virus discovery using DNA capture probes. We have compared shotgun Illumina library sequencing of integrated provirus with target-enriched library sequencing. Our results showed enrichment of control material with varying sequence similarity to bait, redefining the lower limit of detection and improving the sequence coverage. The developed method was used to conduct an investigation of samples from the clinically important cancer types; colon cancer, T- and B-cell lymphoma. We chose to investigate human lymphoma samples also because this kind of cancer in various animals may be caused by retrovirus infection. Our low-stringency capture method shows important potential in pathogen discovery in complex sample material containing small proportions of distantly related sequences.
Results
Probe design
From GenBank we selected genomic sequences from 118 exogenous retroviruses, which are associated with cancer in a vertebrate animal species or in humans (Supplementary Table 1). For certain viral species, sequences from several strains were collected that represent sequence variation. The collected genomic sequences represent viruses from all major branches in the retrovirus phylogeny20. The selected sequences (0.87 Mb) were used as templates for custom design and synthesis of probes. Probe lengths ranged between 60–94 nucleotides (average ± st.dev. = 73.6 ± 4.17). Mapping of the hybridization probe sequences to the selected retrovirus genome sequences revealed extensive tiling of probes resulting in full coverage except for short stretches around ambiguous positions in the reference genomes.
Initial testing of target enrichment was performed on control material consisting of human genomic DNA (gDNA) containing 1.2 × 103 copies of HIV-1Bx08 provirus DNA per μg, matching the probes perfectly. The enrichment of target (represented by HIV-1gag) as well as loss of non-targeted autosomal DNA sequences (B2m) was measured using quantitative PCR.
Prior to enrichment, HIV-1gag was detectable in library DNA in one of two replicates with a high CT-value (~44), indicating a low number of DNA fragments containing the HIV-1gag sequence. In contrast, a comparable amount of target-enriched library DNA contained substantially higher quantities of HIV-1gag in both replicates (Fig. 1A). A modest decrease was seen for the non-enriched human B2m target after enrichment (Fig. 1B). The results encouraged us to use quantitative PCR to estimate target enrichment and de-selection of non-target DNA fragments in optimization experiments, prior to sequencing.
Target enrichment was determined by comparing Illumina sequencing data obtained before and after capture target enrichment. Prior to capture, shotgun sequencing revealed no sign of HIV-1 provirus in the library, as no reads (of total reads >27 × 106) mapped to the HIV-1Bx08 reference genome. In contrast, in two experiments we found 87 or 225 unique read pairs, respectively, mapping to the HIV-1Bx08 reference genome in capture-enriched library DNA (Table 1) in dataset of comparable size (1.5 − 2.0 × 107 unique reads). The level of detection was sufficient to achieve full coverage (>22×) of the HIV-1Bx08 genome (Fig. 1C). In the negative control libraries (non-infected donor PBMC DNA), HIV-1 reads were not detected (Table 1).
Target enrichment increases sensitivity of provirus detection
We investigated target capture of varying quantities of proviral HIV-1 in gDNA from blood donors (1 μg/reaction). It was determined that the HIV-1 targets shared 100% (HIV-1Bx08) or 86% (HIV-1CC0030) sequence similarity to the bait (Table 1).
Shotgun sequencing resulted in no read pairs mapping to the HIV-1 reference genome at quantities <9.1 × 103 HIV-1 copies per reaction. In material containing 1.4 × 105 copies of provirus, 3 to 10 unique read pairs were detected corresponding to an on-target ratio of 0.6–14.2 per million reads (Table 1). Considering the genome size of proviral HIV-1 and the applied sequencing depth, this is consistent with the expected numbers (0–1 read) mapping to HIV-1.
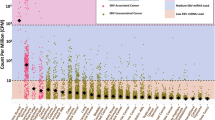
In target-enriched libraries ≥18 unique read pairs were detected that map to the reference genome at quantities down to 9.1 × 101 copies/μg DNA (Table 1). As seen in Fig. 2, there is a clear relationship between the quantity of provirus and the proportion of unique read pairs in sequenced target-enriched libraries.
Relationship between input of viral target and proportion of unique reads mapping to HIV-1 target for captured (solid) or non-captured libraries (open).
Targets and probes showed 86% (squares) or 100% (triangles) sequence similarity. Captured negative control samples are included (round). Results are expressed as on-target unique reads per million of total reads (ppm). ND: None detected.
When comparing target enrichment with shotgun sequencing we found on-target ratios after target enrichment to be 1–3 orders of magnitudes higher (Fig. 2). Furthermore, target capture resulted in increased sensitivity of provirus detection at the present sequencing depth (Table 1).
Viral target capture at low stringency conditions
The in-solution capture format permits lowering of the stringency of hybridization by decreasing the hybridization temperature or adjusting the formamide concentration. Both alternatives were tested using human gDNA containing known quantities of proviral HIV-1CC30 DNA only 86% similar to bait.
Inspired by Mason et al.21, we compared in-solution hybridization at the recommended reaction temperature with gradually decreasing temperatures. In libraries before and after capture, qPCR analysis indicated comparable enrichment of relevant target (HIV-1gag) and comparable lessening (to CT>30) of non-targeted autosomal (B2m) DNA fragments at both conditions (Fig. 3A).
Quantitative PCR analysis of sequencing libraries.
Relative enrichment of target (HIV-1CC30gag) and loss of non-target target (B2m) were measured by qPCR pre- and post-capture. Capture was performed (A) using 20% formamide and different temperatures (47 °C or touch-down), or (B) at reduced formamide concentrations (10% or 0% as indicated). All samples were performed in 2 or 4 technical replicates. ND: Not detected.
Quantitative PCR analysis also indicated that capture at 47 °C, using 20% or 10% formamide, also resulted in comparable enrichment of relevant targets. In contrast, 0% formamide reduced the enrichment of relevant target while increasing the recovery of non-target sequences (Fig. 3B).
Illumina sequencing corroborated these qPCR results. For sub-optimally matching target, or matching target we found no indications that touch-down temperatures improved on-target ratios of reads mapping to reference sequence (Fig. 4A). The reduction of formamide concentration from 20% to 10% seemed to permit an increased proportion of unique reads in the sample with 9.1 × 103 copies/reaction (Fig. 4B).
Enrichment of distantly related sequences using low stringency capture
Our overall aim was to capture distantly related exogenous retroviral sequences in complex samples from humans. Even short complementary regions may mediate hybridization between capture probes and targets18,19 within the host genome e.g. orthologous genes and human endogenous retrovirus (HERV) sequences. Consequently, we investigated the extent of such cross-hybridization in our samples, potentially resulting from unwanted capture of short complementary host genome sequences.
We divided the host genome (hg19) into two groups of 1 kb bins; those containing at least one stretch of ≥25 nucleotides with perfect identity to the sequences of the capture probes and those without. We analysed the sequence coverage in these host genome regions obtained after shotgun- or capture-enriched sequencing (Fig. 5).
Analysis of similarity between probes and host genome.
The normalized number of 1 kb-mers is shown from the human reference genome (Hg19) that contains ≥1 motif of ≥25 bp nucleotides with perfect identity to any of the capture probes (○) or without such motif(s) (•). Data from all sequencing shotgun sequencing experiments (A) and capture-enriched experiments (B) are merged.
After capture enrichment a higher number of regions with 25-mer probe similarity were sequenced with coverage greater than approximately 100 (Fig. 5B). Shotgun sequencing did not show the same bias (Fig. 5A). The phenomenon was most pronounced in libraries captured in 10% formamide. In contrast, touchdown temperature conditions did not increase this cross-hybridization. Our results showed that we selectively detected the regions in the host genome that contained regions (≥25 bp) identical to probes. These results corroborated the previous analyses and indicate that identity in relatively short regions is sufficient for capture enrichment of 100–1000 bp DNA fragments; particularly at low stringency conditions.
Capture of distantly related PERVs
To further explore the concept of intended cross-hybridization, we tested to what extent distantly related proviral sequences were enriched by capture. The extent of capture was investigated in human embryonic kidney (HEK293 cells) containing multiple copies per cell of proviral porcine endogenous retrovirus (PERV) DNA. The pairwise similarity of any capture probe to the PERV genomes was <91%. Approximately 2% of the capture probes showed ≥60% overall similarity to PERV genomes in regions of gag, pol and env. Predictably, we achieved highest coverage in these regions upon capture (Fig. 6).
Low stringency target enrichment of PERV-A and PERV-B genomes from infected HEK293 cell lines.
Coverage of PERV-A (A) or PERV-B (B) genomes after shotgun sequencing (dashed) or low stringency target enrichment (solid lines). The coverage of 25-mer matching probe motifs are shown in (grey □). Location is indicated of the gag, pol and env genes.
With shotgun sequencing the proportion of unique on-target reads was 159 or 516 per million reads (ppm). This proportion translates into 3× or 12.8× coverage, respectively, for proviral PERV-A or PERV-B DNA, with a sequencing depth of 65 × 106 or 138 × 106 reads/sample (Fig. 6). As would ideally be expected from shotgun sequencing, the coverage was relatively evenly distributed over the provirus genomes. The obvious finding is that proviral sequences may be detected with sufficient sequencing depth and virus load.
Moreover, target enrichment by low stringency capture increased the average on-target proportion 174 and 252-fold, respectively, yielding an average sequence coverage of 865 or 949 (Fig. 6). As predicted, coverage tended to peak in regions of the genome with relatively high similarity to the capture probes. Indeed regions with high coverage after capture also contained ≥25-mer motifs identical to capture probes (Fig. 6). All capture probes with such similarity were designed specifically for 17 non-PERV gamma retrovirus reference genomes (see Supplementary Fig. S1 online). Importantly, the reads mapping to the PERV reference sequences did not map elsewhere to annotated genomic HERV sequences (see below), reducing the possibility that reads derived from HERVs. In summary, target (proviral) sequences can be enriched by distantly related probe sequences at low stringency conditions.
As a relatively large proportion of the human genome consists of HERV sequences, enrichment of HERV sequences could be expected. Among the HERV families, the HERV-K is the biologically most active. In some contexts, members of the HERV-K family produce viral particles22. Cloned HERV-K113 proviral DNA produces particles in baculovirus-infected Sf9 cells23. Because HERV-K particles are also seen in germ cell cancer and melanoma24 a role in disease has been suspected. HERV-K113 is distantly related to the beta-retroviruses jaagsiekte sheep retrovirus (JSRV) and mouse mammary tumor virus (MMTV), hence 0.041% of the capture probes map with ≥60% similarity to three distinct regions of the HERV-K113 genome (see Supplementary Fig. 2 online). Consequently, although HERV-K113 is readily detected by shotgun sequencing of HEK293 cells, a small enrichment of HERV-K113 was seen but only in regions with similarity to probes. These results indicate the limits for cross-hybridization.
HIV-1 samples
We investigated peripheral blood from HIV-1-infected individuals (subtype A or B) to demonstrate that the sensitivity of the method is sufficient for clinically relevant samples. For both samples, HIV-1 reads were detected (0.48–0.74 ppm) after low-stringency capture, which is lower than the captured positive control samples (Fig. 2), suggesting a limited DNA viral load. No viral reads were detected in the shotgun sequencing dataset despite sequencing more than 67 × 106 reads per sample (see Supplementary table 2 online). These results demonstrate the method has sufficient sensitivity for detection of proviral DNA in typical clinical sample material.
Capture enrichment of retroviral DNA sequences from cancer samples
Because our capture method enriches for retroviral sequences that may be only distantly related, it has applications in virus discovery. A number of retrovirus species are known causes of cancer, such as lympho-proliferative diseases, in animals and humans3,25. Therefore, we conducted sequence analysis of capture-enriched libraries from B-cell lymphoma cell lines, T-cell lymphoma. As colorectal cancer is an abundant cancer type, such samples were also investigated the presence of reads showing similarity to known vertebrate viruses (Table 2 and Supplementary Table 3).
In capture-enriched libraries prepared from gDNA extracts (34.2–191.1 million trimmed reads per library), many read pairs showed apparent similarity to known acutely transforming retroviruses, such as Abelson murine leukaemia virus, Rous sarcoma virus and others. Further analysis of the location of the reads showed that these reads mapped within the viral oncogenes (e.g. v-abl or v-src). In all cases BLASTn26 analysis of reads or contiguous sequences mapping to viral oncogenes showed higher similarity to the orthologous sequences in the human genome. Thus, as expected, the probes included for oncogenic retroviruses enrich for human orthologous sequences as well. Importantly since initial automated digital subtraction against hg19 required ≥97% sequence identity, some reads with less sequence similarity were not digitally subtracted.
In addition, we conducted shotgun sequencing of DNA libraries (Table 2). As expected, the reads mapping within cellular orthologs to viral oncogenes, were orders of magnitude less abundant in these datasets. Some shotgun-sequenced gDNA libraries from fully transformed B-lymphoma cells (n = 12), biopsies containing cutaneous T-cell lymphoma (n = 6) and biopsies from patients with colorectal cancer (n = 13), contained low numbers of read pairs (often ≤5), mapping to reference genomes of viruses commonly found in humans (e.g. parvovirus B19, herpesviruses) (Table 2). Such findings were not investigated further, as they are all readily explained as random findings not related to the cancer. In one sample (CGG_5_000274), 3223 read pairs mapped across the human herpesvirus type 6A (HHV-6A) reference genome (GenBank NC_001664.2). The proportion of read pairs mapping to HHV-6A or hg19 corresponded to the relative sizes of the HHV-6A and the human diploid genome, suggesting that the sample originated from a patient with a chromosomally integrated copy of HHV-6A. Integrated HHV-6 typically results in high viral loads in all sites27, but has no known association to cancer. The results emphasize that shotgun sequencing is sufficiently sensitive to detect viral genomes only when present in unusually high proportions of complex sample material.
Capture of mRNA libraries
In all RNA libraries we found read pairs mapping to the parvovirus-like hybrid virus (PHV), reference genome (GenBank NC_022089.1). In the digitally subtracted shotgun libraries (n = 16), frequencies ranged between 1.4–655.5 ppm PHV reads. As described by Naccache et al., PHV was found to be a contaminant of the RNA purification kits28. PHV sequences were not eliminated entirely in capture-enriched libraries, although the proportions of PHV seemed to be smaller in these (3.6–7.2-fold, n = 5). Likewise, all shotgun-sequenced RNA libraries contained read pairs mapping to avian retrovirus reference genomes (e.g. 11.2–1005.6 ppm, median = 139.5, n = 16, post digital subtraction for AY350569). These are likely derived from residual avian retrovirus genomic material in the ScriptSeq reverse transcriptase enzyme. Subsequent enrichment by retrovirus-specific probes increases the frequency of these sequences by several orders of magnitude.
Analysis of DNA and RNA libraries in this study uncovered no novel viruses involved in cancer. The presence of PHV-like sequences and avian retroviral sequences in RNA kits or reagents may complicate virus discovery.
Discussion
In the present experiments we have cast a wide-ranging net to capture and enrich sequencing libraries for unknown exogenous retroviral DNA sequences in potentially very low proportions of non-target DNA. Thus, our application of target capture is fundamentally different from other applications where the target may be several megabases of abundant sequence15,16,29,30,31, or degraded ancient DNA in complex sample material15,32. For this investigation, selection of reference genomes included known exogenous virus species. As the purpose of the study was to screen cancer material for retrovirus, we accepted that the probes may tend to over-represent viral species already abundant in the sequence database. Analysis of DNA and RNA libraries in this study uncovered no new retroviruses involved in cancer.
One of the challenges to high-throughput sequencing in pathogen discovery is to detect rare targets in a vast excess of host DNA33. To our knowledge only two studies have used capture enriched high-throughput sequencing of viral targets; both for known and invariable sequences. Depledge et al. capture-enriched sequencing libraries for low titer episomal herpesvirus from a vast excess of host genetic material using RNA probes17. Likewise, integrated Merkel cell polyomavirus sequences were enriched by target capture from formalin-fixed paraffin-embedded tumour samples using PCR amplicons as bait34. We have detected different subtypes of proviral HIV-1 sequences in clinical samples upon capture. We found no indications of artefact contaminating reads in control samples, suggesting that our method permits virus discovery for low titre integrated viral targets with unknown, but related, sequences in an abundant background. This resembles capture enrichment of endogenous ancient DNA from complex material32.
A number of new viruses have been identified using different means of enrichment, including mechanical and enzymatic depletion of host genomic material in combination with subsequent randomly primed PCR9,10,35,36. Whereas these methods have been successful in detection of virions from humans and animals, proviral DNA can remain undetected by random PCR37. Integrated-, latent episomal- and virion-associated viral sequences may be enriched from libraries by capture34.
Considerable sequence variation exists for both DNA and RNA viruses. A very divergent type of human papilloma virus (HPV) was described showing only 61% similarity to other HPV types38. Among the RNA viruses, the inter-subtype genetic distances between HCV strains exceeds 30%39. Retroviruses (e.g. HIV-1) are also highly variable4,5. Therefore the sequence-specific methods, such as high-throughput multiplex qPCR40 or microarrays11, may be inadequate for discovery of novel viruses. Low stringency capture enrichment increases the possibilities of detecting viral DNA and RNA from unknown virus genotypes or species13. By enrichment of the distantly related PERV sequences, we demonstrate that the stringency, or specificity, in the experiments is low enough to allow enrichment of new virus species among the variable retroviruses. In a similar way, low stringency conditions were originally used for identification of certain HERVs using probes derived from non-human retroviruses41,42. Other studies have used touch-down temperature conditions to enrich mitochondrial DNA or chromosomal sequences from related animal species with >60% sequence similarity21,43. Similarly, sub-stringent temperatures were used to allow cross-species hybridization of short DNA fragments in historical material32,44. In our experiments the reduced formamide concentration (10%) or a 5 °C gradual reduction of temperature both relaxed the hybridization stringency which is comparable with other studies21,43.
We selectively detected genomic regions in virus- and in the host genomes that match the bait. Data analysis generated similar results using two different criteria; ≥60% overall similarity43 or perfectly matching ≥25 bp motifs. The 25 bp motif criterion more accurately explained our results (Figs 5 and 6). This is consistent with the notion that a continuous stretch of annealing DNA is advantageous to the formation of a helical conformation which is stable during hybridization and also in good agreement with results from a microarray study13.
The sensitivity of the methods used in virus discovery is rarely assessed. In capture-enriched libraries, we detected as few as 91 copies of HIV-1 proviral DNA per μg gDNA (Table 1). This corresponds to a lower detection limit of approximately <700 viral copy per 1 × 106 cells, which is comparable to the DNA load in other human retroviral infections45. Inclusion of samples from HIV-1-infected individuals substantiates that the sensitivity of the method is sufficient for relevant clinical material, although in HIV-1 elite controllers the DNA load may be only 10–80 copies per million cells46.
From the numbers of on-target reads obtained by shotgun sequencing (Table 1) we estimate that >1 × 108 reads per library would probably be required to reach the same level of sensitivity with shotgun sequencing. The level of sensitivity is comparable to studies with random-primed PCR10,37. In contrast the detection limit in microarray was >1 × 104 viral DNA copies per reaction13.
While the cost of sequencing continues to decrease, capture probe libraries remains to be a considerable expense. The cost per sample may be reduced by immortalized probe libraries47 or by multiplexing of samples48. In virus discovery application, where the target is unknown (and not known to be present), enrichment improves the lower detection level. Our comparison of the proportion of unique on-target reads pre- and post-enrichment was commonly 1–3 orders of magnitude (range 18–2948 fold), which is comparable to other studies using a single round of capture of various targets16,43,49,50. The impact on improved cost and efficiency of downstream bioinformatics analysis is not to be underestimated. As sequencing depth and data volumes are rapidly improving, the bioinformatics is more tractable with enriched sequencing data that is orders of magnitude less voluminous than untargeted sequencing. More importantly, pre-enriched sequencing also helps with matching reads to targets where borderline hits can be more unambiguously defined. Benefits of this would be seen especially in detection of unknown/novel viruses where reads would be imperfectly aligned to known viral material.
Seven different viruses are currently known to cause cancer in humans. HTLV-1 is the aetiology of adult T-cell leukaemia25 and other retroviruses are involved in various animal lympho-proliferative diseases3. Therefore, we included human lymphoma types in our investigation. We also investigated colon cancer samples for traces of retroviral infection as evidenced by proviral DNA and, to a lesser extent, viral RNA. Here we show for the first time the results of screening for unknown expressed or non-expressed-, integrated-, or latent episomal viral DNA in human cancer using massive parallel sequencing. The method complements other methods like sequence-independent PCR on material previously enriched for virions (e.g.10) and array detection12,13.
Our capture-enriched or shotgun libraries revealed evidence of other known viral infections but not for retroviral infections in human cancer samples. These results are in good agreement with recent analyses of shotgun RNA sequencing data from thousands of samples from various cancer types, but not including lymphoma samples51,52.
The probes mediated enrichment for host (human) genome sequences encoding cellular orthologs to retroviral oncogenes (e.g. c-abl and c-src). The bias was most pronounced in libraries captured at 10% formamide, whereas touchdown temperature conditions, in our experiments, were ineffective in improving cross-hybridization. Enrichment of cellular ortholog sequences is an acceptable by-product and servers as an internal control, in the search for new pathogens. A minor concern may be that an abundant cellular ortholog may impair sensitive detection of a retroviral counterpart if rare.
Although intended cross hybridization may not result in full coverage of a viral pathogen, even a very limited coverage may allow subsequent targeted research53. The probability of identifying new viruses in cancer is likely to be lower than in samples from patients with acute febrile illness. The selection of probes determines the broadness of the analysis. For this study, probes covered genome sequences from 118 retroviruses associated with cancer. Our results show that distantly related sequences are captured (incl. virus species in control material, reagent contaminants or cellular orthologs). Likewise, we believe that unknown but distantly related viral sequences would have been detected if present in concentrations higher than one viral copy per 1000 host cells.
Our method has several advantages; firstly, it allows detection of episomal or integrated DNA or RNA54, secondly, it offers sensitivity superior to microarray technology13 and comparable to that of random PCR10 and thirdly, it is compatible with standard protocols for Illumina sequencing platforms and requires only standard laboratory equipment54. Finally, it offers a flexible format of probes (i.e. target size, tiling) that can be individually adapted to different applications. Applications of the low stringency target-enriched sequencing are numerous within several fields of research in which sequences are unknown or variable. Targeted enrichment reduces the cost of sequencing and consequently enables increased number of samples and/or sequencing depth. The method may be an alternative to multiplex PCR or long-range PCR to study gene families or inter-species genetic markers of (distantly) related species. In pathogen discovery enrichment with probes similar to any group of pathogens (e.g. virus, bacteria or fungi) can provide a first handle on the unknown related species. Furthermore, it may be possible to improve enrichment of endogenous DNA cross-species in ancient DNA studies32,49, or within selected taxa in environmental DNA studies55.
In this study we demonstrate that retrovirus are sensitively detected by high-throughput sequencing after enrichment with hybridisation probes, based on distantly related retroviral sequences. In accordance with recent studies on different cancers, our investigation of human B-cell lymphoma cells, cutaneous T-cell lymphoma or colorectal cancer biopsies revealed no retroviral infections associated with cancer.
Methods
Ethics statement
The following two ethical boards reviewed the protocol for the present study: The Regional Committee on Health Research Ethics (Case No. H-2-2012-FSP2) and the National Committee on Health Research Ethics (Case No. 1304226). Both review boards waivered the requirement for informed consent, in accordance with national legislation (Sundhedsloven), as the study design included only samples anonymized at collection (at Department of Surgery, Herlev Hospital, Department of Dermatology Bispebjerg hospital or Department of Haematology, Aalborg University Hospital). Sample material was obtained from cancerous tissue already removed during treatment of patients, by JLL, RG or KD, respectively. Dataset depleted for reads mapping to human genome are deposited at Sequence Read Archive.
Cells and patient samples
Human gDNA containing proviral HIV-1 DNA of isolates Bx08 (GenBank AY713411) and CC0030 (GenBank FJ694791) were generated by standard virus propagation in Phytohemagglutinin-P-stimulated donor peripheral blood mononuclear cells (PBMCs) from blood donors. Frozen whole blood from HIV-1 patients were obtained from Statens Serum Institut.
Human embryonic kidney cell lines (HEK293) infected with porcine endogenous retroviruses (PERV)-A (AJ133817) or PERV-B (AJ133818) were kindly provided by Yasuhiro Takeuchi.
Cryopreserved fully transformed B-cell lymphoma cell lines (RPMI-8226, KMS-12-BM, KMS-12-BM, KMS-12-PE, MOLP-8, MOLP-2, SU-DHL-5, SU-DHL-4, U266, U698M, OCI-Ly8, OCI-Ly7_M and OCI-Ly3_M) were obtained from Aalborg Hospital, Department of Haematology. Cutaneous T-cell lymphoma biopsies were obtained from Bispebjerg Hospital, Department of Dermatology. Colon cancer needle biopsies were obtained from Herlev Hospital, Department of Surgery. Biopsies were obtained on fresh tissue immediately after surgical resection, by microsurgical dissection in the operating room. The resected bowel was closed at both ends and a needle biopsy of the tumor was taken through the serosal side of the bowel at the tumour site. In this way it was possible to obtain a tumour biopsy not contaminated by bowel content. A control biopsy was obtained simultaneously from the same site and it underwent microscopy to ensure, that it contained tumour tissue and not only e.g. necrosis.
Probe design
From GenBank genome sequences were compiled from a total of 118 retroviruses associated with cancer in humans or other vertebrate species (Supplementary Table 1). In instances where several genotypes existed, different variants were included, to represent the existing sequence variation (e.g. ovine nasal tumour virus or HTLV). No identical sequences were included. In 27 cases partial genome sequences were included when no near-full length genomes were available. One near-full length HIV-1 genome (AY713411) was included for control experiments. SeqCap EZ hybridization probes (n = 729,243) were designed and synthesized by Roche NimbleGen (Madison USA).
Library building
Genomic DNA was extracted from cells, biopsies, or blood according to instructions in QIAamp DNA Mini kit (Qiagen). DNA libraries were prepared from 1 μg of DNA according to the Illumina Truseq DNA protocol (PE-940-2001) or an in-house protocol using the NEBnext E6070 (New England Biolabs) reagents56. RNA was extracted from B-lymphoma cells and colon cancer biopsies using mRNA direct Dynabeads (Lifetechnologies) or High Pure Viral RNA kit (Roche), respectively. For lymphoma samples this included ribosomal RNA depletion using RiboZero (Epicentre cat. No. SCL24G) and subsequent purification, using RNeasy MINelute colunms (Qiagen). All RNA libraries were prepared using ScriptSeq Complete Gold Kit (Epicentre) according to manufacturer’s protocol.
Capture
Target enrichment (capture) reactions were performed with 1 μg of library as described in the SeqCap EZ library SR protocol (Roche NimbleGen). In some hybridization reactions the volume of Hybridization component A (formamide) was replaced with water to reduce final formamide concentrations. In other reactions the hybridization temperature was gradually decreased from 47 °C to 42 °C (1 °C/12 hours).
Quantitative PCR
The SeqCap EZ library kit (NimbleGen) includes internal PCR controls to monitor capture efficiency. In addition, standard TaqMan qPCR assays were used to monitor enrichment of targets (HIV-1Bx08 gag) and deselection of non-targeted gDNA sequences, Beta-2-microglobulin (B2m). Roche LC480 probes master (cat no. 04 707 494001) or Roche LC480 SYBR Green I (cat no. 04707516001) reagents were used in TaqMan or SYBR green qPCRs respectively, according to manufacturer’s instructions using comparable concentrations of template DNA (range 4–28 ng/rxn). All qPCRs were performed on a Roche LC480 instrument. Primers and probes are listed in Supplementary Table 4.
Quantification of HIV-1 provirus in control gDNA was performed at the accredited laboratory at Statens Serum Institut, Department of Virology, according to a procedure published elsewhere57.
Sequencing and data analysis
Pools of Illumina libraries were created using unique sequencing indices. Sequencing was performed on HiSeq 2000 instruments at BGI Europe or the Danish National High-Throughput DNA Sequencing Centre56. All sequencing was performed as 100 bp paired-end runs except the two HIV-1 samples, which were 100 bp single-end.
Basic sequence editing, mapping and alignments were performed in Geneious Pro software (ver 5.6.3).
Sequence analysis of control samples
Paired-end sequencing reads were cleaned for adapter sequence using AdapterRemoval58 which removes adapter sequences and merges overlapping mate sequences. The resulting read pairs and singletons were mapped to the human genome (hg19) and selected viral genomes using BWA59. Multiple read pairs/singletons mapping with the same start and end coordinates were considered clonal PCR products and were discarded from further analysis (keeping one representative). Throughout the manuscript the remaining reads are referred to as ‘unique’. Host genome-depleted data from HIV-1-infected individuals were included in BLASTn analysis against reference HIV-1 genomes. Hits exceeding 90% of the read length were counted.
Host genome capture analysis
All possible 25-mers from the capture probe sequences were mapped against the human genome. The human genome was divided into non-overlapping 1 kbp windows and windows overlapping with at least one mapping 25-mer were recorded. This resulted in 50,284 windows with matches to the probe 25-mers and 3,045,381 windows without matches. The average coverage was then compared between the two categories of windows.
Digital subtraction and in silico identification of virus
Digital subtraction of human-like read pairs was performed in two steps using the alignment tools BWA59 using aln default parameters and BLASTn. All read pairs where at least one of the two reads mapped to hg19 were discarded using default alignment parameters. Reads were considered mapped by BWA if they were not assigned SAM flag 460 or if the BLAST alignment was >90% id, 90+ bp and with an e-value <10−3. Dataset depleted for reads mapping to human genome are available at Sequence Read Archive (http://www.ncbi.nlm.nih.gov/Traces/sra/) (PRJNA238023). The remaining reads were mapped by BLAST using identical settings to a database consisting of 1333 NCBI RefSeq vertebrate viruses and their 52970 NCBI Genome Neighbors downloaded from the NCBI Viral Genomes Resource. Positive hits were defined as read pairs where both reads mapped within a distance of 1000 bp in a forward-reverse manner.
Additional Information
How to cite this article: Vinner, L. et al. Investigation of Human Cancers for Retrovirus by Low-Stringency Target Enrichment and High-Throughput Sequencing. Sci. Rep. 5, 13201; doi: 10.1038/srep13201 (2015).
References
Hausen Zur, H. The search for infectious causes of human cancers: Where and why. Virology 392, 1–10 (2009).
Araujo, A. & Hall, W. W. Human T-lymphotropic virus type II and neurological disease. Ann. Neurol. 56, 10–19 (2004).
Coffin, J. M. Retroviruses. (CSHL Press, 1997).
Lynch, R. M., Shen, T., Gnanakaran, S. & Derdeyn, C. A. Appreciating HIV type 1 diversity: subtype differences in Env. AIDS Res. Hum. Retroviruses 25, 237–248 (2009).
Robertson, D. L. et al. HIV-1 nomenclature proposal. Human Retroviruses and AIDS (1999).
Law, J. et al. Identification of Hepatotropic Viruses from Plasma Using Deep Sequencing: A Next Generation Diagnostic Tool. PLoS ONE 8, e60595 (2013).
Yozwiak, N. L. et al. Virus Identification in Unknown Tropical Febrile Illness Cases Using Deep Sequencing. PLoS Negl. Trop. Dis. 6, e1485 (2012).
Jensen, R. H. et al. Target-dependent enrichment of virions determines the reduction of high-throughput sequencing in virus discovery. PLoS ONE 10, e0122636 (2015).
Greninger, A. L. et al. The complete genome of klassevirus - a novel picornavirus in pediatric stool. Virol J 6, 82, 10.1186/1743-422X-6-82 (2009).
Uhlenhaut, C., Cohen, J. I., Fedorko, D., Nanda, S. & Krause, P. R. Use of a universal virus detection assay to identify human metapneumovirus in a hematopoietic stem cell transplant recipient with pneumonia of unknown origin. J. Clin. Virol. 44, 337–339 (2009).
Erlandsson, L., Rosenstierne, M. W., McLoughlin, K., Jaing, C. & Fomsgaard, A. The Microbial Detection Array Combined with Random Phi29-Amplification Used as a Diagnostic Tool for Virus Detection in Clinical Samples. PLoS ONE 6, e22631 (2011).
Wang, D. et al. Microarray-based detection and genotyping of viral pathogens. Proc. Natl. Acad. Sci. USA. 99, 15687–15692 (2002).
Palacios, G. et al. Panmicrobial oligonucleotide array for diagnosis of infectious diseases. Emerging Infect. Dis. 13, 73–81 (2007).
LaFramboise, T. Single nucleotide polymorphism arrays: a decade of biological, computational and technological advances. Nucleic Acids Res 37, 4181–4193 (2009).
Maricic, T., Whitten, M. & Pääbo, S. Multiplexed DNA Sequence Capture of Mitochondrial Genomes Using PCR Products. PLoS ONE 5, e14004 (2010).
Hodges, E. et al. Genome-wide in situ exon capture for selective resequencing. Nat Genet 39, 1522–1527 (2007).
Depledge, D. P. et al. Specific Capture and Whole-Genome Sequencing of Viruses from Clinical Samples. PLoS ONE 6, e27805 (2011).
Kane, M. D. et al. Assessment of the sensitivity and specificity of oligonucleotide (50mer) microarrays. Nucleic Acids Res. 28, 4552–4557 (2000).
Garhyan, J. J., Gharaibeh, R. Z. R., McGee, S. S. & Gibas, C. J. C. The illusion of specific capture: surface and solution studies of suboptimal oligonucleotide hybridization. BMC Res Notes 6, 72, 10.1186/1756-0500-6-72 (2012).
Weiss, R. A. Retrovirology 3, 67, 10.1186/1742-4690-3-67 (2006).
Mason, V. C., Li, G., Helgen, K. M. & Murphy, W. J. Efficient cross-species capture hybridization and next-generation sequencing of mitochondrial genomes from noninvasively sampled museum specimens. Genome Res. 21, 1695–1704 (2011).
Löwer, R., Löwer, J. & Kurth, R. The viruses in all of us: characteristics and biological significance of human endogenous retrovirus sequences. Proc. Natl. Acad. Sci. USA. 93, 5177–5184 (1996).
Boller, K. et al. Human endogenous retrovirus HERV-K113 is capable of producing intact viral particles. J. Gen. Virol. 89, 567–572 (2008).
Muster, T. et al. An endogenous retrovirus derived from human melanoma cells. Cancer Res. 63, 8735–8741 (2003).
Iwanaga, M., Watanabe, T. & Yamaguchi, K. Adult T-cell leukemia: a review of epidemiological evidence. Front. Microbiol. 3, 1–13 (2012).
Altschul, S. F., Gish, W., Miller, W., Myers, E. W. & Lipman, D. J. Basic local alignment search tool. J. Mol. Biol. 215, 403–410 (1990).
Hall, C. B. et al. Chromosomal Integration of Human Herpesvirus 6 Is the Major Mode of Congenital Human Herpesvirus 6 Infection. PEDIATRICS 122, 513–520 (2008).
Naccache, S. N. et al. The Perils of Pathogen Discovery: Origin of a Novel Parvovirus-Like Hybrid Genome Traced to Nucleic Acid Extraction Spin Columns. J. Virol. 87, 11966–11977 (2013).
Melnikov, A. et al. Hybrid selection for sequencing pathogen genomes from clinical samples. Genome Biol. 12, R73, 10.1186/gb-2011-12-8-r73 (2011).
Gnirke, A. et al. Solution hybrid selection with ultra-long oligonucleotides for massively parallel targeted sequencing. Nat. Biotechnol. 27, 182–189 (2009).
Bainbridge, M. N. et al. Whole exome capture in solution with 3 Gbp of data. Genome. Biol. 11, R62, 10.1186/gb-2010-11-6-r62 (2010).
Dabney, J. et al. Complete mitochondrial genome sequence of a Middle Pleistocene cave bear reconstructed from ultrashort DNA fragments. Proc. Natl. Acad. Sci. USA. 110, 15758–15763 (2013).
Barzon, L. et al. Next-generation sequencing technologies in diagnostic virology. J. Clin. Virol. 58, 346–50 (2013).
Duncavage, E. J. et al. Hybrid Capture and Next-Generation Sequencing Identify Viral Integration Sites from Formalin-Fixed, Paraffin-Embedded Tissue. J. Mol. Diagn. 13, 325–333 (2011).
Xu, B. et al. Metagenomic Analysis of Fever, Thrombocytopenia and Leukopenia Syndrome (FTLS) in Henan Province, China: Discovery of a New Bunyavirus. PLoS Pathogens 7, e1002369 (2011).
Yu, G. et al. Discovery of a Novel Polyomavirus in Acute Diarrheal Samples from Children. PLoS ONE 7, e49449 (2012).
McClenahan, S. D., Uhlenhaut, C. & Krause, P. R. Optimization of virus detection in cells using massively parallel sequencing. Biologicals 1–8; 10.1016/j.biologicals.2013.11.002 (2013).
Mokili, J. L. et al. Identification of a Novel Human Papillomavirus by Metagenomic Analysis of Samples from Patients with Febrile Respiratory Illness. PLoS ONE 8, e58404 (2013).
Kuiken, C., Yusim, K., Boykin, L. & Richardson, R. The Los Alamos hepatitis C sequence database. Bioinformatics 21, 379–384 (2005).
Kiss, M. M. M. et al. High-throughput quantitative polymerase chain reaction in picoliter droplets. Anal. Chem. 80, 8975–8981 (2008).
Repaske, R., O’Neill, R. R., Steele, P. E. & Martin, M. A. Characterization and partial nucleotide sequence of endogenous type C retrovirus segments in human chromosomal DNA. Proc. Natl. Acad. Sci. USA. 80, 678–682 (1983).
O’Connell, C., O’Brien, S., Nash, W. G. & Cohen, M. ERV3, a full-length human endogenous provirus: chromosomal localization and evolutionary relationships. Virology 138, 225–235 (1984).
Li, C., Hofreiter, M., Straube, N., Corrigan, S. & Naylor, G. J. P. Capturing protein-coding genes across highly divergent species. BioTechniques 54, 321–326 (2013).
Meyer, M. et al. A mitochondrial genome sequence of a hominin from Sima de los Huesos. Nature 505, 403–6 (2014).
Gibellini, D. et al. HIV-1 DNA load analysis in peripheral blood lymphocytes and monocytes from naïve and HAART-treated individuals. J. Infect. 56, 219–225 (2008).
Hatano, H. et al. Evidence for Persistent Low-Level Viremia in Individuals Who Control Human Immunodeficiency Virus in the Absence of Antiretroviral Therapy. J. Virol. 83, 329–335 (2008).
Querfurth, R., Fischer, A., Schweiger, M., Lehrach, H. & Mertes, F. Creation and application of immortalized bait libraries for targeted enrichment and next-generation sequencing. BioTechniques 52; 375–80 (2012).
Rohland, N. & Reich, D. Cost-effective, high-throughput DNA sequencing libraries for multiplexed target capture. Genome Res. 22, 939–946 (2012).
Carpenter, M. L. et al. Pulling out the 1%: Whole-Genome Capture for the Targeted Enrichment of Ancient DNA Sequencing Libraries. Am. J. Hum. Genet. 93, 852–64 (2013).
Ávila-Arcos, M. C. et al. Application and comparison of large-scale solution-based DNA capture-enrichment methods on ancient DNA. Sci. Rep. 1, 74, 10.1038/srep00074 (2011).
Tang, K.-W., Alaei-Mahabadi, B., Samuelsson, T., Lindh, M. & Larsson, E. The landscape of viral expression and host gene fusion and adaptation in human cancer. Nat. Comms 4, 1–9 (2013).
Khoury, J. D. et al. Landscape of DNA Virus Associations across Human Malignant Cancers: Analysis of 3,775 Cases Using RNA-Seq. J. Virol. 87, 8916–8926 (2013).
Alquezar-Planas, D. E. et al. Discovery of a divergent HPIV4 from respiratory secretions using second and third generation metagenomic sequencing. Sci. Rep. 3, 2468, 10.1038/srep02468 (2013).
Smith, M. et al. An in-solution hybridisation method for the isolation of pathogen DNA from human DNA-rich clinical samples for analysis by NGS. Open Genomics J. 5, 18–29 (2012).
Ilves, K. L. & López-Fernández, H. A targeted next-generation sequencing toolkit for exon-based cichlid phylogenomics. Mol. Ecol. Resour. 14, 802–11 (2014).
Seguin-Orlando, A. et al. Ligation Bias in Illumina Next-Generation DNA Libraries: Implications for Sequencing Ancient Genomes. PLoS ONE 8, e78575 (2013).
Edelmann, A. et al. Improvement of an ultrasensitive human immunodeficiency virus type 1 real-time reverse transcriptase-polymerase chain reaction targeting the long terminal repeat region. Transfusion 50, 685–692 (2010).
Lindgreen, S. AdapterRemoval: easy cleaning of next-generation sequencing reads. BMC Res. Notes 5, 337, 10.1186/1756-0500-5-337 (2012).
Li, H. & Durbin, R. Fast and accurate long-read alignment with Burrows-Wheeler transform. Bioinformatics 26, 589–595 (2010).
Li, H. et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
Acknowledgements
The authors would like to acknowledge Yasuhiro Takeuchi for HEK293/PERV cells; Danish National High throughput Sequencing Centre for sequencing; BGI Europe for sequencing; Sarah Natalie Vitcetz for technical assistance; Ellinor Lindberg for qPCR and Lea Brandt and Sanne Skov Jensen for PBMCs; Kristoffer Rapacki, John D. Sørensen, Kirstine Belling, Simon Rasmussen and the other members of the NGS analysis pipeline at CBS for the scientific discussion and the technical support provided. The work was supported by the Innovation Fund Denmark (The GenomeDenmark platform, grant no. 019-2011-2).
Author information
Authors and Affiliations
Contributions
Conceived concept and designed and experiments L.V.I., L.P.N., A.J.H. and E.W. Performed the experiments: L.V.I. Analysed the data: J.F.N., T.M. and L.V.I. Contributed reagents/materials/analysis tools: J.R., J.L.L., R.G., K.D., J.I., J.F., D.F. and E.W. Conceived and designed computational research: J.F.N., T.S.P., R.G. and S.B. Conducted computational research: T.M. and J.F.N. Analysed the computational data: J.F.N., T.M. and L.V.I. Prepared the manuscript: L.V.I. Critical revision and approval of manuscript: All.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Vinner, L., Mourier, T., Friis-Nielsen, J. et al. Investigation of Human Cancers for Retrovirus by Low-Stringency Target Enrichment and High-Throughput Sequencing. Sci Rep 5, 13201 (2015). https://doi.org/10.1038/srep13201
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep13201
This article is cited by
-
Target region sequencing and applications in plants
Journal of Crop Science and Biotechnology (2021)
-
Evidence for a causal role by human papillomaviruses in prostate cancer – a systematic review
Infectious Agents and Cancer (2020)
-
Enteric Virome and Carcinogenesis in the Gut
Digestive Diseases and Sciences (2020)
-
Can Bovine Leukemia Virus Be Related to Human Breast Cancer? A Review of the Evidence
Journal of Mammary Gland Biology and Neoplasia (2018)
-
Mouse mammary tumor-like virus (MMTV) is present in human breast tissue before development of virally associated breast cancer
Infectious Agents and Cancer (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.