Abstract
The networks of predator-prey interactions in ecological systems are remarkably complex, but nevertheless surprisingly stable in terms of long term persistence of the system as a whole. In order to understand the mechanism driving the complexity and stability of such food webs, we developed an eco-evolutionary model in which new species emerge as modifications of existing ones and dynamic ecological interactions determine which species are viable. The food-web structure thereby emerges from the dynamical interplay between speciation and trophic interactions. The proposed model is less abstract than earlier evolutionary food web models in the sense that all three evolving traits have a clear biological meaning, namely the average body mass of the individuals, the preferred prey body mass and the width of their potential prey body mass spectrum. We observed networks with a wide range of sizes and structures and high similarity to natural food webs. The model networks exhibit a continuous species turnover, but massive extinction waves that affect more than 50% of the network are not observed.
Similar content being viewed by others
Introduction
Classical models addressing the structure and stability of food webs are based on stochastic algorithms that produce structural patterns similar to empirically measured food webs1, such as the niche model2 or the cascade model3. A more recent approach is to use the empirically found allometries of body size and foraging behaviour of individual consumers to predict the links between species on a more biological basis4.
However, real food webs are not produced by a generative algorithm, but have been shaped by their evolutionary history and show an ongoing species turnover. New species in a food web occur by immigration and speciation and species vanish due to extinction. Currently, the world faces one of the largest extinction waves ever, which is thought to be caused by anthropogenic drivers such as climate change and land use5. Even without human interference or other catastrophic causes and apart from evolutionary suicide due to runaway selection6, biological extinctions occur due to intrinsic processes, i.e., the dynamic trophic and competitive interactions among species7,8. The stability of food webs in terms of resistance to extinction waves after a perturbation (such as the removal or addition of a species), thus also depends on the network structure of these interactions between the species9,10 and conversely the network structure results from species extinctions and additions. Understanding the interplay of food web structure and stability has therefore been identified as one of the most important questions in ecology11.
Over the last decade, several models were introduced that include evolutionary rules on a longer time scale, in addition to population dynamics on shorter time scales: The former enables new species to enter the system, whereas the latter determines which species are viable and which go extinct. The newly emerging species can be modelled and interpreted either as invaders from another, not explicitly considered region or as “mutants” of existing species. The emerging network structures evolve in a self-organising manner, giving rise to complex, species-rich communities even when starting from initial networks with very few species1.
A particularly simple and often cited evolutionary food web model was introduced in 2005 by Loeuille and Loreau12 and subsequently modified by several other authors13,14,15. Each species is characterised by its body mass, which is the only evolving trait. Feeding and competition interactions are determined via differences in body mass. The fact that body mass is an ecologically interpretable trait makes the results from this model easily comparable to empirical data. This major advantage has been pointed out in the review on large community-evolution models by Brännström et al.16. The evolutionary process in this model generates large networks that show an almost static behaviour, with clearly defined niches all of which are and remain occupied. Even if a newly emerging species is slightly better adapted to the resources and therefore displaces a species of similar body mass, it has the same feeding preferences and hence the same function in the food web, leading to a very low species turnover without secondary extinctions15. The network structure is robust with respect to various changes in the population dynamics rules, indicating that some simple, robust mechanism structuring these food webs is at work12,15.
Complex networks with a less rigid structure emerge in the evolutionary version of the niche model17. The model allows for the evolution of three traits instead of just one, namely the niche value, the centre and the width of the feeding range. Other authors describe a species in a more abstract way by a vector of many traits, as implemented in the matching model18,19 and in the webworld model20,21. Recently, also several individual-based models for evolving food webs were introduced22,23,24. The emergence of complex food webs in these models is highly nontrivial. Some past attempts to set up an evolutionary model lead to repeated network collapse instead of persisting complex networks25. Other attempts lead to trivial network structures, like simple food chains in the evolving niche model17 or a single trophic level in the webworld model21. In both models, adaptive foraging was required in order to obtain more complex networks.
Allhoff and Drossel15 suggested that an evolutionary food web model has to fulfil two conditions to be able to generate diverse and complex networks. First, it should allow for the evolution of more traits in addition to body mass in order to generate several possible survival strategies like for example specialists and omnivores. This idea is consistent with results from a recent empirical study by Rall et al.26, who found that predators of similar body mass differ significantly in their feeding preferences. Second, the evolution of each trait has to be restricted in order to prevent unrealistic trends, for example towards extremely small or large body masses or towards extremely broad or narrow feeding ranges. In this context, the stabilising effect of adaptive foraging in previous models could be explained by the fact that a predator can focus on its most profitable prey without losing adaptation to other prey.
In this paper, we propose a new evolutionary food web model that includes the restriction of trait evolution in a more direct way. Similarly to the evolutionary niche model17 and supported by empirical data regarding the body-mass ratios of predator-prey pairs27,28, we characterise a species by three traits with clear biological meaning: its own body mass (which determines its metabolic rates), its preferred prey body mass and the width of its potential prey body mass spectrum. The evolutionary rules in our model confine the traits within certain boundaries, without the requirement to include adaptive foraging.
The model most similar to our model is the one by Loeuille and Loreau12. It also has body mass as a key trait and a similar concept for setting the feeding preferences. Our model differs from the model by Loeuille and Loreau in the number of traits that characterize a species (3 instead of 1), the functional response (Beddington-deAngelis instead of linear), the competition rules (based on link overlap instead of body mass differences), the possibility of cannibalism and loops (included only in our model) and the resource dynamics. Moreover, we consider body mass ratios instead of body mass differences so that the body masses in our model spread over several orders of magnitude instead of only one. The bio-energetics of the species in our model follow well documented allometric scaling relationships29, leading to networks with realistic body-mass scaling relations that can be tested directly against empirical data.
We demonstrate the capabilities of our model by evaluating 18 common food web properties and compare them to a data set of 51 empirical food webs from a large variety of different ecosystems. We further use the well-known evolutionary model by Loeuille and Loreau12 as a benchmark to assess the quality of the predictions of our model. In principle, both models are able to produce diverse networks. However, we obtain a higher variability in the feeding preferences and survival strategies and therefore more realistic values for the corresponding network properties. Moreover, while the network structures of Loeuille and Loreau are static, species turnover and extinction avalanches occur naturally in our model.
The Model
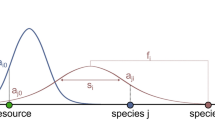
The model includes fast ecological processes (population dynamics), which determine whether a species is viable in a given environment that is created by the other species and slow evolutionary processes (speciation events), which add new species and enable the network to grow and produce a self-organised structure. A species i is characterised by its body mass, mi, the centre of its feeding range, fi and the width of its feeding range, si. These traits determine the feeding interactions in the community (see Fig. 1) and thereby the population dynamics. A summary of all model parameters and variables is given in table 1.
Model illustration using 4 species.
Species 3 (black triangle) is characterised by its body mass m3, the centre of its feeding range f3 and the width of its feeding range s3. The Gaussian function (black curve) describes its attack rate kernel N3j on potential prey species. Here, species 3 feeds on species 2 and 1 (grey triangles) with a high resp. low attack rate. Species 1 and 2 are consumers of the external resource, represented as species 0 with a body mass m0 = 1 (white triangle). Also shown is the corresponding network graph.
Population dynamics
The population dynamics follows the multi-species generalisation of the bioenergetics approach by Yodzis and Innes30,31. The rates of change of the biomass densities Bi of the populations are given by

for the external resource (species 0) and

for consumer species. G0 = R(1 − B0/K) is the logistic growth rate of the external resource, ej is the efficiency with which biomass of species j can be assimilated by its consumers, gij is the mass-specific rate with which species i consumes species j and xi is i’s mass-specific respiration rate. The mass-specific consumption rate is given by

where

is the rate of successful attacks of species i on individuals of species j, with the Gaussian feeding kernel Nij as shown in Fig. 1. The parameter hi is the handling time of species i for one unit of prey biomass and cil quantifies interference competition among predators i and l32,33,34. It depends on their similarity, as measured by the overlap  of their feeding kernels, via
of their feeding kernels, via

The normalisation of the competition with Iii was proposed by Scheffer et al.35 and accounts for the fact that the competition matrix is not symmetric. More specialised species exert a higher competition pressure than species with broad feeding ranges. The overlap Iil is similar to the niche overlap discussed by May36.
We assume that interference competition is significantly higher within a species than between different species, e.g. due to territorial or mating behaviour. To account for this, we introduce an intraspecific competition parameter cintra and set cii = cfood + cintra.
Speciation events
Each simulation starts with a single ancestor species with body mass m1 = 100 and feeding parameters f1 = 1 and s1 = 1, which is thus feeding on the external resource with its maximum attack rate. The initial biomass densities are B0 = K = 100 for the resource and B1 = m1 ⋅ ε = 2 ⋅ 10−2 for the ancestor species. The parameter ε is the extinction threshold, i.e., the minimum population density required for a population to survive. At each unit time step, species below this extinction threshold get removed from the system.
A speciation event occurs with probability ω = 0.0001 per unit time. This is so rare that the system is typically close to a fixed point before the next mutation occurs. Then, one of the currently existing species (but not the external resource) is chosen randomly as parent species i for a “mutant” species j. Thus, every species has the same probability ω/S to “mutate”, where S is the number of currently viable species. The logarithm of the mutant’s body mass, log10(mj), is chosen randomly from the interval [log10(0.5mi), log10(2mi)], meaning that the body masses of parent and mutant species differ at most by a factor of 2. The mutant’s initial biomass density is set to Bj = mj ⋅ ε and is taken from the parent species.
The mutant’s feeding traits fj and sj are independent of the parent species. The logarithm of the feeding centre, log10 fj, is drawn randomly from the interval [(log10(mj) − 3), (log10(mj) − 0.5)], meaning that the preferred prey body mass is 3 to 1000 times smaller than the consumer’s body mass and following the results from Brose et al.27. The width of the feeding range, sj, is drawn randomly from the interval [0.5,1.5]. A small value of sj corresponds to a more specialised consumer, while a large value of sj characterises a consumer with a broad feeding range and lower attack rates. A combination of large preferred prey mass fj and a wide feeding range enables a consumer to prey on species with a larger body mass than its own. This enables the emergence of cannibalism and feeding loops. The fixed intervals keep the evolving traits in reasonable ranges and prevent unrealistic trends, following the results by Allhoff and Drossel15.
When testing the robustness of the model predictions with respect to the model details, we used alternative rules, where the probability for choosing a parent species is proportional to its biomass (similar to the model by Loeuille and Loreau12) or to its inverse generation time  so that the mutation rate is proportional to the reproduction rate. Furthermore, we tested Gaussian distributions of mutant body masses around the parent with a standard deviation between 0.09 and 1. We used a cutoff at two standard deviations resulting in a maximum body mass factor between parent and daughter species between 102⋅0.09 ≈ 1.5 and 102⋅1 ≈ 100. The former describes local speciation events, whereas the latter describes species invasions from not explicitly modelled regions. We also compared the results to simulations where the mutants body mass is drawn randomly from the interval [10−0.5,106]. Finally, with a similar approach, we also included heredity into the feeding parameters si and fi by combining Gaussian distributions around the parent’s traits with the above given mutation intervals.
so that the mutation rate is proportional to the reproduction rate. Furthermore, we tested Gaussian distributions of mutant body masses around the parent with a standard deviation between 0.09 and 1. We used a cutoff at two standard deviations resulting in a maximum body mass factor between parent and daughter species between 102⋅0.09 ≈ 1.5 and 102⋅1 ≈ 100. The former describes local speciation events, whereas the latter describes species invasions from not explicitly modelled regions. We also compared the results to simulations where the mutants body mass is drawn randomly from the interval [10−0.5,106]. Finally, with a similar approach, we also included heredity into the feeding parameters si and fi by combining Gaussian distributions around the parent’s traits with the above given mutation intervals.
Methods
The computer code for our simulations was written in C. We used the Runge-Kutta-Fehlberg algorithm provided by the GNU Scientific library37 for the numerical integration of the differential equations. Simulations were run for 5 ⋅ 108 time units. For comparison, the generation time of the initial ancestor species with body size m1 = 100 is of the order of  time units.
time units.
The competition parameters cfood and cintra have a strong effect on the diversity of the emerging food webs. To obtain the network variability observed in nature, we performed computer simulations with all four combinations of cfood = 0.6 or 0.8 and cintra = 1.4 or 1.8. The time series of these simulations are shown in the online supplementary material. From each simulation run, we collected 80 food webs obtained after every 5 ⋅ 106 time units from t = 108 to t = 5 ⋅ 108, resulting in a total of 320 different networks. Due to the initial build-up of the network, the first 108 time units were not taken into account.
The structure of the emerging food webs is compared both to food webs produced with the model by Loeuille and Loreau12 and to empirical food webs. For the empirical data, we re-evaluated 51 of the 65 food webs from different ecosystem types analysed by Riede et al.38 for which we had body-mass data for all species in the network (for the complete list see online supplementary material). For the model by Loeuille and Loreau, we evaluated the final network structures obtained with 75 combinations of different parameter values. Due to the static network structure, we could not obtain different networks from one evolutionary simulation. The niche width was set to  and the competition strength to α0 = 0.1, 0.2, 0.3, 0.4, 0.5, similar to the original work. To get networks of comparable size we decreased the competition range, β = 0.025, 0.05, 0.075.
and the competition strength to α0 = 0.1, 0.2, 0.3, 0.4, 0.5, similar to the original work. To get networks of comparable size we decreased the competition range, β = 0.025, 0.05, 0.075.
Both models use Gaussian feeding kernels with in principle infinite width to describe the feeding interactions, meaning that each species can prey on every other species. Thus, for analysis, very weak links have to be cut off in order to obtain meaningful network structures. In our networks, we removed all links that contribute less than 75% of the average link to the total resources of a consumer. This criterion is weaker than it might seem, because most of the links of a predator are very weak and so is the average link strength. Our cutoff measure depends on both the attack rate and the prey’s biomass density. It thereby mimics unavoidable sampling limits in empirical food-web studies. For the networks produced by the algorithm of Loeuille and Loreau we used the cutoff criterion of the original work and removed all links with an attack rate that is smaller than 15% of the respective predator’s potential maximum attack rate, disregarding the prey’s biomass density. Since the value of the cutoff criterion is to some extent arbitrary, we report its effects on the predicted network properties in the online supplementary material. There we also show results obtained for the model by Loeuille and Loreau with our cutoff rule.
Results
A typical simulation run
A typical simulation run with the competition parameters cintra = 1.4 and cfood = 0.8 is shown in Fig. 2. After an initial period of strong diversification, the system reaches a size of approximately 60 species (panel (a)) on 3 to 4 trophic levels above the resource (panel (c)). The species form clusters of similar body masses, as shown in panel (b). New predator and prey species emerge preferentially within these clusters: A prey species in a cluster experiences less predation pressure due to the saturation of the functional response of the predator and the predation input of a predator is larger if its feeding preferences match such a cluster. Therefore, we observe a trend towards strong specialisation on these clusters, resulting in the following network structure. Species in the first cluster have a body mass of approximately 101, specialise on the resource and represent most of the second trophic level. Species in the second cluster with a body mass of approximately 102 − 103 feed either on the resource (TL ≈ 2) or on the first cluster (TL ≈ 3). Species in the top cluster with a body mass greater than 103 specialise either on the first or on the second cluster and therefore have intermediate trophic positions (3 ≤ TL ≤ 4). Some species have even higher trophic positions due to cannibalism and loops.
Network size, body masses and flow-based trophic positions60 of all species occurring during one exemplary simulation run with competition parameters cintra = 1.4 and cfood = 0.8.
Panel (a) also shows the average network size and its standard deviation for 18 simulations with identical parameters but different random numbers. Body masses and trophic positions were plotted at every 25th mutation event. Network visualisations for the time points indicated by vertical lines are shown in the online supplementary material.
The initial build-up of the network continues until the species in the top cluster are close to the extinction threshold. Once all clusters have emerged, the system shows a continuous turnover of species. We suppose the following turnover mechanism. Mutants with very few predators can occur occasionally if their body mass is between two clusters and if the other species are specialised on the clusters. If such a mutant has viable feeding parameters, it can grow a large population and displace many other species at once, potentially even causing secondary extinctions. Examples for such extinction events are visible at t ≈ 2 ⋅ 108 and t ≈ 4.3 ⋅ 108. After an extinction event, the network rearranges and temporally also species with broader feeding ranges appear, before the trend towards specialisation followed by an extinction event starts again.
Network evaluation and comparison
We compared 320 networks from our model with 51 empirical networks and 75 networks from the model by Loeuille and Loreau12, see Fig. 3. Panels (a)–(c) show the distributions of body masses of all three data sets. The observed peaks in our simulated data correspond to the body mass clusters mentioned before. The distance between the peak maxima is determined by the upper boundary of the mutation interval of the feeding centre. Single empirical food webs show a similar peak pattern (not shown). In contrast, the body mass distribution of the model by Loeuille and Loreau looks blurred, due to our choice of the niche width  . With smaller values of the feeding range s, the network structure is strongly layered and clusters of body masses that are multiples of the feeding distance d occur, where each species feeds on those in the cluster below and is prey to those in the cluster above15. We also observed that because we based the network structure on predator-prey body-mass ratios instead of body-mass differences, the resulting community-size spectra from our model follow empirical observations and theoretical predictions more closely than those from the model by Loeuille and Loreau, as shown in the online supplementary material.
. With smaller values of the feeding range s, the network structure is strongly layered and clusters of body masses that are multiples of the feeding distance d occur, where each species feeds on those in the cluster below and is prey to those in the cluster above15. We also observed that because we based the network structure on predator-prey body-mass ratios instead of body-mass differences, the resulting community-size spectra from our model follow empirical observations and theoretical predictions more closely than those from the model by Loeuille and Loreau, as shown in the online supplementary material.
Frequency distributions of body masses and short-weighted trophic level60, as well as the distributions of generality (number of prey species) and vulnerability (number of predators).
The latter two are normalised by the average number of links per species. nm: 320 networks from 4 simulations of our new model with all four combinations of cfood = 0.6 or 0.8 and cintra = 1.4 or 1.8. emp: Average over 51 empirical food webs. LL: Average over 75 simulations of the model by Loeuille and Loreau12. Note that panel (c) shows absolute body masses, since in this model all body masses are in the same order of magnitude. See Methods for more information.
Panels (d)–(f) show the distributions of trophic levels of all three data sets. Here, we use the short-weighted instead of the flow-based trophic level. This allows for better comparison with the empirical data for which the population sizes are often not available. The comparison between the two models reveals the main difference between the two different cutoff rules. A link with intermediate attack rate to a small prey population represents only a small proportion of the predator’s diet and is therefore neglected when using our cutoff threshold (75% of the average link). However, it is not recognised as a weak link with the cutoff rule by Loeuille and Loreau (15% of the maximum attack rate). On the other hand, a link with small attack rate to a big prey population (especially to the resource) is deleted in their model. Thus, trophic levels are overestimated, whereas our model with our cutoff rule results in a quite realistic distribution.
Both models have difficulties reproducing the empirical distributions of generality (number of prey species) and vulnerability (number of predators), which are much broader than the distributions produced by the models (panels (g)–(l)). For the model by Loeuille and Loreau, the distribution results from the fact that the species in the model feed only on prey with smaller body masses. The situation is similar to the cascade model3, which also constrains predators to feed only on prey with a lower rank. Consequently, both generality and vulnerability cannot be larger than twice the average number of links per species. In our new model, the distribution of the vulnerability shows two humps. The first hump contains the carnivores in the higher trophic levels that feed on herbivores or on other carnivores. They have a high generality and a small vulnerability. The second hump contains the herbivores that feed on the resource. They are prey to many other species and hence have a high vulnerability.
We ascribe the differences between the models and the empirical distributions to the fact that both models have only one resource, which means that all herbivores feed on the same resource, whereas in empirical networks herbivores can have more than one resource. Furthermore, both models ignore the within-species body-mass distribution by assigning to each species a precise value of the body mass. This also narrows down the range of body sizes a species can feed on or is vulnerable to.
By analysing the 320 networks from the 4 simulations separately (see Fig. 4), we found two trends concerning the network size (panel (a)): First, the stronger the intraspecific competition cintra, the smaller are the population sizes and the more populations can survive on the same amount of energy provided by the resource. Second, the stronger the competition for food cfood is, the sooner species can displace others resulting in rather small networks with fast evolutionary species turnover.
Network properties of four realisations with different values of the competition parameters.
w/w: Weak competition, cintra = 1.4/cfood = 0.6. w/s: Weak intraspecific competition and strong competition for food, cintra = 1.4 / cfood = 0.8. s/w: Strong intraspecific competition and weak competition for food, cintra = 1.6 / cfood = 0.6. s/s: Strong competition, cintra = 1.6 / cfood = 0.8. emp: Average over 51 empirical food webs. LL: Average over 75 simulations of the model by Loeuille and Loreau12. See Methods for more information. Details on the calculation of these network characteristics can be found in the online supplementary material.
Both models are able to produce networks of realistic sizes, but tend to overestimate the number of links per species (panel (b)) and hence the connectance (panel (c)). The effect is much larger in the model by Loeuille and Loreau due to their original cutoff rule. This also explains the high fraction of omnivores and the low fraction of top and herbivorous species (panels (d)–(f)), as well as the high values of the number of chains and the clustering coefficient (panel (o) and (p)) and the small value of the characteristic path length (panel (r)). In the online supplementary material, we show that the model by Loeuille and Loreau provides more realistic predictions when using our cutoff rule.
Both models fail to reproduce the maximum similarity (panel (q)), due to the same reasons that also lead to the narrow distributions of generality and vulnerability. For the remaining panels, the model by Loeuille and Loreau performs worse than our model regardless which cutoff rule is used. For example, the short-weighted trophic levels (panel (j)–(l)) are not only overestimated due to the cutoff rule, but also reflect the regular network structure. As mentioned above, these networks are layer-like structures, where each cluster represents one trophic level. Since all clusters accommodate a similar number of species instead of heaving more species on lower levels like in our model, the mean trophic level is overestimated. Moreover, the model does not include cannibalism (panel (m)) and loops (panel (n)), for which our model provides good predictions.
Due to the evolution of three instead of one trait, we obtain more diverse network structures than Loeuille and Loreau. We observe a higher standard deviation of the generality, the vulnerability and the linkedness (panel (g)–(i)), reflecting different feeding preferences and survival strategies.
Robustness of the results against variations of the evolutionary rules
In order to ensure that our findings are no artefacts of the specific choice of evolutionary rules, we tested the robustness of our results against the changes outlined at the end of the model section. We found that making mutation probabilities dependent on biomass or body mass influences the time dependency of the network development but leaves the averaged network properties, like the total network size, the distribution of body masses and the fraction of species or biomasses per trophic level, mostly unchanged. Also the trend towards strong specialisation with subsequent extinctions still occurs in these variants.
When changing the degree to which the parent’s body mass is inherited by the mutant, the main effect was that species turnover became slower with stronger inheritance. In this case it is less likely that mutants with body masses between two clusters occur, which have few predators and cause extinction avalanches. The probability for such mutants increases with a decreasing degree of inheritance, which is consistent with our oberservation that the body mass clusters appear to be blurred in case of a very low degree of inheritance. The same is true for randomly chosen body masses. However, we still obtain large, complex networks.
If the parent species i and the mutant j have similar feeding centres, fi ≈ fj, the initial build-up of different trophic levels and their recovery after an extinction avalanche is also slowed down. With very strong inheritance of the feeding centre, all species will focus on the resource and no mutant emerges with a feeding centre matching the first body mass cluster, leading to trivial structures with only one trophic level. If parent and mutant have a similar degree of specialisation, si ≈ sj, all species exert and experience a similar competition pressure. Thus, instead of one species displacing another, both populations stay small and hence more populations per trophic level can survive. However, small or intermediate degrees of inheritance in the feeding traits leave the network characteristics again mostly unchanged. The situation is different, when either the feeding range or the feeding centre is chosen from an interval around the parent’s trait without any body mass dependent constraint. In consistency with the predictions of Allhoff and Drossel15, these variants lead to unrealistic trends and trivial instead of complex network structures.
Discussion
We introduced a new evolutionary food web model where the feeding links are based on body mass and where species differ by body mass, feeding centre and feeding range. By iterating population dynamics and speciation events for a sufficiently long time, we obtained complex networks, which show a high degree of commonality with empirical food webs. The new model is able to produce more realistic and more diverse network structures than the model by Loeuille and Loreau12.
Both models use a very similar approach of Gaussian feeding kernels to determine the interactions between the species, which by construction leads to perfectly interval networks. Following the results of Stouffer et al.39, we assume this to be a reasonable approximation. In contrast to the model by Loeuille and Loreau, the new model allows for cannibalism and loops, since the feeding range can extend to body masses larger than that of the predator. The species in our model can have different feeding preferences and survival strategies, due to the larger number of evolving traits in our model. This leads to a higher variability in network characteristics such as linkedness, generality and vulnerability, even though natural variability is still larger, which we ascribe to the facts that our model has only one basal resource and no body-size structure within species. We showed that an appropriate choice of the cutoff rule for weak links is essential for obtaining realistic results for connectance and trophic structure.
The increased number of evolving traits compared to the model by Loeuille and Loreau has also a large effect on the evolutionary trends. The networks show an ongoing species turnover and are subject to constant restructuring. The species in our model form body mass clusters and the evolutionary process is characterised by a trend towards increased specialisation on these clusters. Similar specialisation trends have also been observed in other studies15,17. We assume the following explanation for the continuous species turnover. The evolved specialists gradually replace less efficient species with broader feeding ranges that cover also the gaps between the body mass clusters. Those broad ranged species have the role of keystone species that stabilise the networks against the occurrence of large extinction avalanches40,41. In the absence of control by such predators, new mutants (or invaders) can find niches between two clusters with very little predation pressure, where they can grow to high abundance and cause extinction avalanches propagating from lower to higher trophic levels. After such extinction events, the empty niches can be reoccupied also by species with broader feeding ranges, before the speciation process starts again.
This corresponds to the results of Binzer et al.8, who identified specialised species on high trophic levels to be prone to secondary extinctions and to the results of Rossberg42, who suggested a very similar turnover mechanism for the results of his model. In consistence with the described mechanism, also Mellard and Ballantyne43 reported that co-evolution of species does not necessarily lead to high levels of resilience for the ecosystem as a whole. However, such a turnover mechanism is missing in the model by Loeuille and Loreau. There, a displaced species is always replaced by a new species of a very similar body mass. And since the body mass is the only evolving trait, the new species has automatically the same predators and the same prey, excluding the possibility of secondary extinctions or major changes in the network structure15. The same is true for the model version by Brannström et al., which led to evolutionary equilibria, where no more mutants are able to invade the system14. Ingram et al. reported that also their model extension with evolving feeding ranges, but with fixed predator-prey body mass ratios, tends to reach dynamically stable configurations with little structural change13.
However, real ecosystems do show extinction events of different sizes and their distribution evaluated over geological times resembles a power law44. For this reason, it has been suggested that ecosystems show self-organised criticality (SOC)45, which means that the intrinsic dynamics of the systems is responsible for the power-law size distribution of extinctions. However, the question remains open due to sparse and ambiguous data46,47. Some previous evolutionary food web models, for example the evolutionary niche model17, exhibit SOC, whereas other models like the webworld model20 or the model by Loeuille and Loreau12 do not. The size distribution of extinction avalanches in our model is a power law with an exponent around 4 (not shown). Because of its steepness, this power law covers only approximately one decade, meaning that extinction events of more than 10 species are extremely rare. This is not the type of SOC required to explain the large extinction events in earth history, where up to 90 percent of all species went extinct. Regarding the time span a species is present in the system, our model is consistent with paleobiological data concerning the fact that higher trophic level species stay in the system for a shorter time span than lower level species46, although it should be mentioned that the exact distribution of these time spans in our model depends on the relation between a species’ body mass and its mutation probability.
The evolutionary rules implemented in our model are simplified and to some extent artificial. To make sure that our results do not depend on these simplified rules, we tested several variations concerning the mutation and inheritance rules. Our general finding is that minor changes in the evolutionary algorithm have only minor effects on the results. The overall mechanism with a trend towards specialisation followed by an extinction event as explained above is robust to changes in the evolutionary rules. Also the time averaged network structures remain mostly unchanged. However, the typical time period for a specialisation-extinction cycle can change with extinction events being triggered sooner or later.
The fact that our networks show realistic patterns concerning many common food web properties suggests that our model provides a valuable tool to discuss urgent topics in ecological research. For example, the allometric equations are extendable by temperature terms (e.g.48,49,50,51). This approach would allow to model how warming might change evolution and extinction waves, in order to discuss current global change questions.
Another idea would be to address habitat loss and habitat fragmentation as a prominent example of an external driver of extinction events52,53. Recently, various approaches have been made to study the influence of the spatial environment on the food web composition and stability. If space has the structure of discrete habitats, these food webs can be interpreted as “networks of networks”54,55. However, most of the studies on such metacommunities so far focus on spatial aspects under the assumption that the species composition is static, although it has been emphasised that combining the spatial and the evolutionary perspective is essential for a better understanding of ecosystems56,57,58. Recently, Allhoff et al. studied a spatial version of the model by Loeuille and Loreau59. However, their findings were associated with the applied competition rules and the remarkable stability of the original model, highlighting the assumption that a more dynamic species turnover as in our new model would lead to a better understanding of the interplay between evolving food web structure and spatial structure.
Additional Information
How to cite this article: Allhoff, K.T. et al. Evolutionary food web model based on body masses gives realistic networks with permanent species turnover. Sci. Rep. 5, 10955; doi: 10.1038/srep10955 (2015).
References
Drossel, B. & McKane, A. J. Handbook of Graphs and Networks: From the Genome to the Internet, Ch. 10, 218–247 (Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, 2005).
Williams, R. J. & Martinez, N. D. Simple rules yield complex food webs. Nature 404, 180–182 (2000).
Cohen, J. & Newman, C. A stochastic theory of community food webs: I. models and aggregated data. Proceedings of the Royal society of London. Series B. Biological sciences 224, 421–448 (1985).
Petchey, O. L., Beckerman, A. P., Riede, J. O. & Warren, P. H. Size, foraging and food web structure. Proceedings of the National Academy of Sciences 105, 4191–4196 (2008).
Barnosky, A. D. et al. Has the earth’s sixth mass extinction already arrived? Nature 471, 51–57 (2011).
Parvinen, K. Evolutionary suicide. Acta Biotheoretica 53, 241–264 (2005).
Riede, J. O. et al. Size-based food web characteristics govern the response to species extinctions. Basic and Applied Ecology 12, 581–589 (2011).
Binzer, A. et al. The susceptibility of species to extinctions in model communities. Basic and Applied Ecology 12, 590–599 (2011).
May, R. M. Will a large complex system be stable? Nature 238, 413–414 (1972).
Otto, S. B., Rall, B. C. & Brose, U. Allometric degree distributions facilitate food-web stability. Nature 450, 1226–1229 (2007).
May, R. Unanswered questions in ecology. Philos. Trans. R. Soc. London B Biol. Sci. 354, 1951–1959 (1999).
Loeuille, N. & Loreau, M. Evolutionary emergence of size-structured food webs. PNAS 102, 5761–5766 (2005).
Ingram, T., Harmon, L. J. & Shurin, J. B. Niche evolution, trophic structure and species turnover in model food webs. The American Naturalist 174, 56–67 (2009).
Brännström, Å., Loeuille, N., Loreau, M. & Dieckmann, U. Emergence and maintenance of biodiversity in an evolutionary food-web model. Theoretical Ecology 4, 467–478 (2011).
Allhoff, K. T. & Drossel, B. When do evolutionarty food web models generate complex structures? Journal of Theoretical Biology 334, 122–129 (2013).
Brännström, Å. et al. Modelling the ecology and evolution of communities: a review of past achievements, current efforts and future promises. Evolutionary Ecology Research 14, 601–625 (2012).
Guill, C. & Drossel, B. Emergence of complexity in evolving niche-model food webs. Journal of Theoretical Biology 251, 108–120 (2008).
Rossberg, A., Matsuda, H., Amemiya, T. & Itoh, K. Food webs: Experts consuming families of experts. Journal of Theoretical Biology 241, 552–563 (2006).
Rossberg, A., Ishii, R., Amemiya, T. & Itoh, K. The top-down mechanism for body-mass-abundance scaling. Ecology 89, 567–580 (2008).
Drossel, B., Higgs, P. G. & McKane, A. J. The influence of predator-prey population dynamics on the long-term evolution of food web structure. Journal of Theoretical Biology 208, 91–107 (2001).
Drossel, B., McKane, A. J. & Quince, C. The impact of nonlinear functional responses on the long-term evolution of food web structure. J. Theor. Biol. 229, 539–548 (2004).
Bell, G. The evolution of trophic structure. Heredity 99, 494–505 (2007).
Yamaguchi, W., Kondoh, M. & Kawata, M. Effects of evolutionary changes in prey use on the relationship between food web complexity and stability. Popul. Ecol. 53, 59–72 (2011).
Takahashi, D., Brännström, Å., Mazzucco, R., Yamauchi, A. & Dieckmann, U. Abrupt community transitions and cyclic evolutionary dynamics in complex food webs. Journal of Theoretical Biology 337, 181–189 (2013).
Takahashi, D., Brännström, Å., Mazzucco, R., Yamauchi, A. & Dieckmann, U. Cyclic transitions in simulated food-web evolution. J. Plant. Interact. 6, 181–182 (2011).
Rall, B. C., Kalinkat, G., Ott, D., Vucic-Pestic, O. & Brose, U. Taxonomic versus allometric constraints on non-linear interaction strengths. Oikos 120, 483–492 (2011).
Brose, U. et al. Consumer-resource body-size relationships in natural food webs. Ecology 87, 2411–2417 (2006).
Riede, J. O. et al. Stepping in elton’s footprints: a general scaling model for body masses and trophic levels across ecosystems. Ecology Letters 14, 169–178 (2011).
Brown, J. H., Gillooly, J. F., Allen, A. P., Savage, V. M. & West, G. B. Toward a metabolic theory of ecology. Ecology 85, 1771–1789 (2004).
Yodzis, P. & Innes, S. Body size and consumer-resource dynamics. The American Naturalist 139, 1151–1175 (1992).
Brose, U., Williams, R. J. & Martinez, N. D. Allometric scaling enhances stability in complex food webs. Ecol. Lett. 9, 1228–1236 (2006).
Beddington, J. R. Mutual interference between parasites or predators and its effect on searching efficiency. Journal of Animal Ecology 44, 331–340 (1975).
DeAngelis, D. L., Goldstein, R. A. & O’Neill, R. V. A model for tropic interaction. Ecology 56, 881–892 (1975).
Skalski, G. T. & Gilliam, J. F. Functional responses with predator interference: viable alternatives to the holling type ii model. Ecology 82, 3083–3092 (2001).
Scheffer, M. & van Nes, E. H. Self-organized similarity, the evolutionary emergence of groups of similar species. Proceedings of the National Academy of Sciences 103, 6230–6235 (2006).
May, R. M. On the theory of niche overlap. Theoretical Population Biology 5, 297–332 (1974).
Galassi, M. et al. GNU Scientific Library Reference Manual (Network Theory Ltd, 2009).
Riede, J. O. et al. Scaling of food-web properties with diversity and complexity across ecosystems. Advances In Ecological Research 42, 139–170 (2010).
Stouffer, D. B., Camacho, J. & Amaral, L. A. N. A robust measure of food web intervality. Proceedings of the National Academy of Sciences 103, 19015–19020 (2006).
Power, M. E. et al. Challenges in the quest for keystones. Bioscience 46, 609–620 (1996).
Eklöf, A. & Ebenmann, B. Species loss and secondary extinctions in simple and complex model communities. Journal of Animal Ecology 75, 239–246 (2006).
Rossberg, A. G. Food webs and biodiversity: foundations, models, data, Ch. 22, 287–309 (John Wiley & Sons, 2013).
Mellard, J. P. & Ballantyne IV, F. Conflict between dynamical and evolutionary stability in simple ecosystems. Theoretical Ecology 7, 273–288 (2014).
Raup, D. Biological extinction in earth history. Science 231, 1528–1533 (1986).
Sneppen, K., Bak, P., Flyvbjerg, H. & Jensen, M. H. Evolution as a self-organized critical phenomenon. Proceedings of the National Academy of Sciences 92, 5209–5213 (1995).
Newman, M. E. & Palmer, R. G. Modeling extinction (Oxford University Press, 2003).
Drossel, B. Biological evolution and statistical physics. Advances in Physics 50, 209–295 (2001).
Vasseur, D. A. & McCann, K. S. A mechanistic approach for modeling temperature-dependent consumer-resource dynamics. The American Naturalist 166, 184–198 (2005).
Binzer, A., Guill, C., Brose, U. & Rall, B. C. The dynamics of food chains under climate change and nutrient enrichment. Philosophical Transactions of the Royal Society B: Biological Sciences 367, 2935–2944 (2012).
Norberg, J., Urban, M. C., Vellend, M., Klausmeier, C. A. & Loeuille, N. Eco-evolutionary responses of biodiversity to climate change. Nature Climate Change 2, 747–751 (2012).
Stegen, J. C., Ferriere, R. & Enquist, B. J. Evolving ecological networks and the emergence of biodiversity patterns across temperature gradients. Proceedings of the Royal Society of London B: Biological Sciences 279, 1051–1060 (2012).
Hagen, M. et al. Biodiversity, species interactions and ecological networks in a fragmented world. Advances in Ecological Research 46, 89–210 (2012).
Gonzalez, A., Rayfield, B. & Lindo, Z. The disentangled bank: How loss of habitat fragments and disassembles ecological networks. American Journal of Botany 98, 503–516 (2011).
Amarasekare, P. Spatial dynamics of foodwebs. Annual Review of Ecology, Evolution and Systematics 39, 479–500 (2008).
Leibold, M. A. et al. The metacommunity concept: a framework for multi-scale community ecology. Ecology letters 7, 601–613 (2004).
Logue, J. B., Mouquet, N., Peter, H. & Hillebrand, H. Empirical approaches to metacommunities: a review and comparison with theory. Trends in Ecology & Evolution 26, 482–491 (2011).
Urban, M. C. et al. The evolutionary ecology of metacommunities. Trends in Ecology & Evolution 23, 311–317 (2008).
Loeuille, N. & Leibold, M. Ecological consequences of evolution in plant defenses in a metacommunity. Theoretical population biology 74, 34–45 (2008).
Allhoff, K. T., Weiel, E. M., Rogge, T. & Drossel, B. On the interplay of speciation and dispersal: An evolutionary food web model in space. Journal of Theoretical Biology 366, 46–56 (2014).
Williams, R. J. & Martinez, N. D. Limits to trophic levels and omnivory in complex food webs: Theory and data. The American Naturalist 163, 458–468 (2004).
Acknowledgements
This work was supported by the DFG under contract number Dr300/12-1 and 13-1. C.G. was supported by the Leopoldina Fellowship Program under contract number LPDS 2012-07. We thank Markus Schiffhauer and Jannis Weigend, who analysed several model variants concerning the mutation and heredity rules in the context of their bachelor theses. Moreover, we thank Christoph Digel and Jens Riede for providing empirical data and Sebastian Plitzko and Bernd Blasius for very helpful discussions.
Author information
Authors and Affiliations
Contributions
All authors designed the model. K.T.A. performed the simulations. K.T.A. and C.G. analysed the results. K.T.A. wrote the manuscript with minor contributions from the other authors. All authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Allhoff, K., Ritterskamp, D., Rall, B. et al. Evolutionary food web model based on body masses gives realistic networks with permanent species turnover. Sci Rep 5, 10955 (2015). https://doi.org/10.1038/srep10955
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep10955
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.