Abstract
Amphibian chytridiomycosis, an emerging infectious disease caused by the fungus Batrachochytrium dendrobatidis (Bd), has been a significant driver of amphibian declines. While globally widespread, Bd had not yet been reported from within Madagascar. We document surveys conducted across the country between 2005 and 2014, showing Bd's first record in 2010. Subsequently, Bd was detected in multiple areas, with prevalence reaching up to 100%. Detection of Bd appears to be associated with mid to high elevation sites and to have a seasonal pattern, with greater detectability during the dry season. Lineage-based PCR was performed on a subset of samples. While some did not amplify with any lineage probe, when a positive signal was observed, samples were most similar to the Global Panzootic Lineage (BdGPL). These results may suggest that Bd arrived recently, but do not exclude the existence of a previously undetected endemic Bd genotype. Representatives of all native anuran families have tested Bd-positive and exposure trials confirm infection by Bd is possible. Bd's presence could pose significant threats to Madagascar's unique “megadiverse” amphibians.
Similar content being viewed by others
Introduction
Amphibian population declines and extinctions are occurring at unprecedented rates1. Multiple anthropogenic factors including habitat destruction and alteration, introduction of alien species and over-exploitation are linked to the global declines of amphibians. Chytridiomycosis, an emerging infectious disease caused by the pathogen Batrachochytrium dendrobatidis (Bd), is also recognized as playing a significant role in the rapid declines and extinctions of amphibians around the world2,3. Bd has been detected in over 500 species worldwide (http://www.bd-maps.net/) and at least 200 species have declined as a result of chytrid infection4. Bd has decimated amphibian populations in the Neotropics, Australian Wet Tropics, the western USA, Europe and east Africa5,6,7,8. For example, in the Neotropics, approximately 67% (110 species) of the genus Atelopus disappeared across their range9 and when Bd arrived in Panama, 41% of the amphibian diversity was lost from the highland site of El Copé10. The pathogen's ability to infect numerous host species and spread rapidly through amphibian assemblages and on a global scale make it the greatest disease threat to biodiversity at the current time11. Despite the occurrence of Bd on almost every continent, there are regions of the world that are considered pathogen-free, including Papua New Guinea12 and Madagascar. In Madagascar, comprehensive surveys for Bd were conducted in 2005–2010 with no detection of the pathogen13,14,15,16, despite much of the eastern rainforest being climatically suitable for Bd17,37.
Madagascar harbours an extraordinary amphibian diversity, with more than 290 described species and well over 200 undescribed candidate species of frogs belonging to five independent radiations18,19. Except for two introduced species, all Malagasy amphibians are endemic to the island which therefore hosts a considerable proportion of the currently ca. 7300 amphibian species inhabiting the world20. Many Malagasy rainforest sites hold numerous sympatric amphibian species21 and an extraordinary density of adults and tadpoles22. Comparatively high frog diversity also occurs in apparently hostile dry habitats with temporary pools23. The most important threat to Madagascar's amphibians is deforestation, yet some species are also threatened by excessive pet trade collection and the likely effects of climate change and habitat alteration24,25,26,27. The introduction of a non-native, virulent lineage of Bd would add to the threats posed against Madagascar's unique amphibian communities.
In the past years, numerous activities to meet the challenges of amphibian conservation in Madagascar have occurred. In 2006 the first “A Conservation Strategy for the Amphibians of Madagascar” workshop was organized to develop the “Sahonagasy Action Plan”, a national action plan28, resulting later in several actions, including information management and citizen-science initiatives, reserve planning29 and in-situ breeding facilities30. In 2010, the Chytridiomycosis Working Group (CWG) was established to facilitate chytrid-related research in Madagascar and the Chytrid Emergency Cell (CEC) was created to develop specific protocols to prevent the arrival of Bd and to rapidly respond to chytridiomycosis outbreaks in Madagascar. In addition, the National Monitoring Plan (NMP) which biannually surveys for Bd across the island at eight selected sites was launched31. All these activities started under the premise that Bd was absent in Madagascar, according to the surveys published before 201413,14,15,16,32; however, recently Bd was reported on Malagasy frogs imported to the USA for the pet trade in 201233.
Here we present the first evidence for the widespread presence of Bd in wild amphibian populations from Bd surveys carried out from 2005–2014 at various sites across the country and provide preliminary information about the identity of the Bd lineage.
Results
Results are based on the analysis of 4,155 amphibians tested for the presence of Bd, 1,113 of which have been presented in previous publications13,14,15,16,32,34. Fifty-two sites across Madagascar were sampled, with the earliest sampling undertaken in 2005. Ninety-nine different sampling events were completed, with a mean sample size of 42 ± 3 SE frogs across these sampling events. For this study we combined all data available to us from 2005–2014. These data are from (i) samples of the National Monitoring Plan, (ii) samples obtained in the context of a skin microbiota study of Madagascar's amphibians and (iii) samples collected opportunistically. While the data thus do not agree with an ideal sampling design, they do cover all major biomes and elevation zones of the island, as well as both the dry and wet season, allowing for the first assessment of spatial and temporal patterns of Bd occurrence and prevalence in Madagascar (see Supplementary Table 1 for more details).
Bd sampling
In 2005–2008, 892 amphibians were sampled in Ambohitantely, An'Ala, Andasibe, Andringitra, Ankarafantsika, Ankaratra, Antananarivo, Isalo, Manakara, Manombo, Masoala, Montagne d'Ambre and Ranomafana and all samples tested negative for Bd13,14,15.
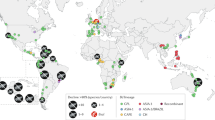
The first record of Bd was documented in December 2010 in the Makay Massif (Figure 1 and Table 1). Bd was detected in a site locally known as Andranovinily, one of five sampled sites in the Makay Massif. Three individuals out of 37 frogs tested positive, with Bd intensities ranging from 0.157–0.273 genome equivalents (GE) (Supplementary Table 1, Figure 1). All positives were from samples of Mantidactylus sp. Ca1419 (Table 2). In the same year, surveys conducted in Toamasina and Ankaratra did not detect Bd16,32.
Map of all sites sampled for Bd.
Circles represent sites of surveys conducted between 2005–2014 and red colouring highlights Bd-positive sites. Location name, site name, the month-year of detection and prevalence are provided for each location with Bd-positive occurrences. The names of all remaining sites can be found in Supplementary Table 1. ND indicates that prevalence could not be determined due to pooling of collected samples for detection analysis. The base map was obtained from www.worldofmaps.net. Points on the map were generated using QGis 2.0 (Quantum GIS Development Team, 2013) and afterwards edited on Adobe PhotoShop CS6 (Adobe, 2012).
In 2011, Bd was again detected in Makay. It was found in one sample of Ptychadena mascareniensis collected in Beroroha (Figure 1, Table 2) and had a Bd intensity of 0.239 GE (Supplementary Table 1). No Bd was detected in the 83 samples collected from the site Andranovinily (positive in 2010), nor was it detected at the three other Makay sites visited (Figure 1). Similarly, surveys in other areas, including Ankarafantsika, Toamasina, Antoetra, Andasibe and Mandena, did not yield any Bd-positive samples (Figure 1).
In 2012, Bd was detected across multiple locations: samples from Ankarafantsika (March), Ankaratra (August) and Antoetra (October) tested positive (Figure 1 and Table 1). The samples were pooled together by site for qPCR analysis to reduce cost and decrease the time needed for sample analysis and therefore it was not possible to determine prevalence of Bd at these locations. At Ankaratra and Antoetra, the positive samples were from Mantidactylus pauliani and M. sp. Ca48 respectively (Table 2). No information on the species are available from the samples collected in Ankarafantsika. The Bd intensities of the pooled samples were 11.0, 21.0 and 2.0 GE for Ankaratra, Antoetra and Ankarafantsika respectively (Supplementary Table 1). All samples from Andasibe, Ankaratra (May), Antoetra (March), Masoala, Menabe and Toamasina tested negative for Bd (Table 1).
In 2013, there was increasing sampling effort with 10 different locations surveyed (Figure 2), of which four were surveyed multiple times throughout the year. Samples from Ankaratra and Ranomafana tested positive for Bd (Figure 1 and Table 1). More specifically, the Ankaratra region was sampled four times during 2013. In February and June, all Ankaratra samples tested negative for Bd, however, subsequently in August, numerous samples tested positive for Bd. The prevalence at the Ambohimirandrana and Tavolotara sites within Ankaratra were 100% and the Bd intensities ranged from 37.3–167.8 GE and 47.3–95.2 GE respectively (Figure 1, Supplementary Table 1). Five different species tested positive for Bd, including Boophis ankaratra, B. goudoti, B. williamsi, Mantidactylus pauliani and M. sp. Ca1919 (Table 2). At Ambatolampy, a ricefield site at the base of the Ankaratra Massif, the prevalence of Bd was 63% and the Bd intensity ranged from 15.9–97.6 GE (Figure 1, Supplementary Table 1). The Bd-positive samples were collected from Heterixalus betsileo and Ptychadena mascareniensis (Table 2). In December, one sample collected from Mantidactylus sp. Ca19 at Ambohimirandrana tested positive for Bd. The Bd intensity of this sample was 0.75 GE (Supplementary Table 1). All other Ankaratra sites tested negative for Bd in December. At Ranomafana in August, frogs sampled at Vatoharanana tested positive for Bd. The prevalence of Bd was 50% and the Bd intensities ranged from 16.6–145.8 GE (Figure 1, Supplementary Table 1). All samples collected at Andasibe, Ankarafantsika, Makay, Mandena, Masoala (multiple sites), Menabe, Toamasina and Torotorofotsy tested negative for Bd (Table 1).
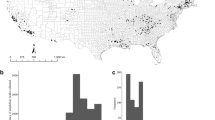
Sampling effort between 2006–2014 for each bioclimatic region of Madagascar.
A “P” indicates Bd-positive occurrences and the size of the black-outlined boxes represents the number of Bd-positive sample events. A map with corresponding colours is included to present the locations of the bioclimatic regions in Madagascar. Only sampling events with PCR-based Bd screening are included. The inset map was prepared in Corel Draw software by redrawing an original map from George Schatz.
In early 2014, samples collected in Antoetra and Ranomafana tested positive for Bd. At Ranomafana six sites were sampled. One out of 45 samples collected at Valohoaka tested positive for Bd (Boophis reticulatus; 2.53 GE; Table 2, Supplementary Table 1). In Vatoharanana, which was positive in August 2013, none of 87 sampled individuals were found positive for Bd. All samples collected from the other sites in Ranomafana tested negative for Bd. In Antoetra, three sites were sampled. Bd was detected at one site, Soamazaka, where one out of nine samples tested positive for Bd (Mantella cowani; 2.88 GE; Table 2, Supplementary Table 1). Samples from Ambohitantely, An'Ala, Andasibe, Fierenana and Torotorofotsy all tested negative for Bd.
Bd prevalence tended to be greater in mid to high elevation sites (800+ meters above sea level35) (Figure 3A) and in the montane and subhumid bioclimatic regions (Figure 3B). In addition, there was a trend toward season having an effect on Bd's prevalence across the different locations, with higher values in the dry season (Figure 3C).
Bd lineage in Madagascar
Lineage-specific qPCR was undertaken to determine if the Bd present in Madagascar was more closely related to the BdGPL, BdCAPE or BdCH lineages. A subset of samples, that were Bd positive with the general ITS probe, were tested with lineage specific probes (Table 3). While not all samples showed positive amplification, the ones that amplified with the lineage specific probes all showed the presence of a BdGPL-like lineage (Table 3). This BdGPL-like lineage may be the true BdGPL, an endemic BdGPL-like, or a non-endemic BdGPL-like lineage. None of the tested samples showed positive amplification with the CAPE-specific or CH-specific probe (Table 3).
Non-amphibian Bd vectors: Crayfish
Crayfish in Antananarivo, Mandraka and Ranomafana were sampled for Bd in January-February 2014. No individuals of either the invasive Procambarus sp. or the native Astacoides spp. yielded positives for Bd.
Amphibian chytrid fungus infectivity
No data are so far available on the susceptibility of Malagasy frogs to chytridiomycosis. We report preliminary data on infectivity for a series of species of the families Ptychadenidae, Hyperoliidae and Mantellidae (details in Supplementary Methods). The exposure trials with an isolate of BdGPL confirmed that this Bd lineage has the potential to infect individuals of the species Boophis madagascariensis, B. viridis, Heterixalus betsileo, Mantidactylus betsileanus and Ptychadena mascareniensis (see Supplementary data).
Discussion
In this study, we document the presence of the amphibian chytrid fungus, Batrachochytrium dendrobatidis, in wild populations of amphibians in Madagascar. Previously published surveys (2005–2010) across the country13,14,15,16,32, in addition to data presented here, show the lack of Bd detection prior to 2010. Between 2010 and 2014, Bd has been recorded in five different areas of the country: Ankarafantsika (March 2012), Ankaratra (August 2012, August and December 2013), Antoetra, (October 2012, January 2014), Makay (December 2010, August 2011) and Ranomafana (August 2013, January 2014). So far, Bd positive samples in Madagascar are distributed over all four families of native Malagasy frogs: Hyperoliidae (Heterixalus), Microhylidae (Platypelis and Scaphiophryne33), Ptychadenidae (Ptychadena) and Mantellidae (Boophis, Gephyromantis, Mantella and Mantidactylus). The positive occurrences seem to be associated with mid to high elevation sites, which is consistent with the climatic suitability expected for Bd36 and is similar to other regions of the world where Bd prevalence and Bd-associated declines are greater in high elevation regions8,37,38,39.
Bd was first detected in Madagascar in 2010 in Makay at low prevalence and intensity. In 2011 Bd's presence was confirmed although with lower prevalence. Makay is a very remote massif containing relicts of humid forest within a predominantly dry region of western Madagascar and therefore it appears as a very unusual place for an initial introduction of Bd to occur. Nevertheless, several hypotheses can be proposed to explain the occurrence of Bd in this region. One hypothesis is that a Bd lineage that is endemic to Madagascar has been always present in Makay. Since no sampling was completed prior to 2010, this hypothesis cannot be excluded. Museum specimens collected previously from this region need to be tested in the future for the presence of Bd to better understand when Bd arrived in the Makay region, as was done in Central America to track Bd's movement through the region40. Alternatively, it is possible that Bd in Makay was introduced recently, potentially as a consequence of increased tourist activity.
At Ankarafantsika, Ankaratra and Antoetra there were surveys yielding negative results completed within 1–2 years of the first detection of Bd which may suggest a recent arrival of Bd at these locations. In Ranomafana, surveys were only conducted in 2006 and 2007 and then in 2013 and 2014; therefore, it is difficult to make any inference about the time of arrival of Bd in this region. Importantly, Ranomafana National Park is one of the areas that is most visited by tourists41 and scientific researchers and thus may carry a greater risk of pathogen introduction. Interestingly, three individuals out of 565 (one of Scaphiopyrne spinosa, Heterixalus betsileo and H. alboguttatus) imported to the US from Madagascar in February 2012 for the pet trade tested positive for Bd33. It is impossible to know whether these individuals were infected in the wild or as a result of contamination during shipment and transport; however, these individuals likely originated not far from Ranomafana where all three species are known to occur in sympatry42. Therefore, the likely origin of these specimens seems to parallel the positive occurrences seen in Ranomafana in 2013.
The use of lineage specific qPCR shows that at least some of the Bd detected in Madagascar is a BdGPL-like lineage. The BdGPL lineage occurs on every continent43, is associated with all of the known epizootics that have occurred, is spatially emerging on a worldwide-scale and in experimental settings is more virulent than other lineages44. If it is further confirmed that the Bd in Madagascar is the true BdGPL then it was likely introduced and may be highly virulent.
If Bd was introduced in Madagascar it is important to understand the route and timing of introduction. In 2003 crayfish, Procambarus sp., were introduced with road construction equipment from outside Madagascar near the capital, Antananarivo45. These organisms could be a potential source for Bd introduction as crayfish can be an alternative host for the pathogen46. However, invasive as well as native crayfish were sampled in January and February of 2014 and no Bd was detected. This result, along with data showing that the distribution of the invasive crayfish does not overlap with the occurrences of Bd, suggests that crayfish are most likely not responsible for introducing Bd in Madagascar. Recently it has also been documented that an alien toad species (Duttaphrynus melanostictus) has locally invaded eastern Madagascar47. If the origin of these invaders was a Bd-positive area of the world, then these amphibians could be Bd-carriers and may have subsequently introduced Bd to Madagascar; however, so far this species has never been recorded as a carrier of the pathogen48. Other possible routes of introduction include bird feathers or moist soil49, accidental human-assisted transport, trade of wildlife, aquarium fish and plants50, or machinery of foreign companies transported from diseased regions of the world27.
Although it is not always the case, in a scenario of recent arrival of a virulent Bd lineage to Madagascar, one may expect negative host effects on the frog species, similar to those seen in other tropical regions5,10. During the conducted surveys, no individuals exhibited signs of clinical chytridiomycosis and up to now (January 2015) no mortality events associated to Bd occurrence have been reported in Madagascar. Based on the intensity of recent herpetological and biological fieldwork throughout Madagascar, it thus seems unlikely that amphibian mass-mortality events have occurred widely on the island. It is possible that Malagasy amphibians are in some way pre-adapted to be resistant and/or tolerant to the Bd lineage present in Madagascar. This concept of preadaptation of Malagasy frogs needs to be thoroughly investigated before any conclusions can be drawn. Research on the defensive function of the adaptive immunity51, innate immunity (AMPs)52 and cutaneous microbial communities53 can develop a better understanding of the resistance, tolerance and susceptibility of Malagasy amphibians to Bd.
Preliminary exposure trials showed that Malagasy amphibians can become infected with Bd (Supplementary Data); however, these trials do not allow for any inference about these species' susceptibility to chytridiomycosis. These data must be interpreted with caution as these trials were conducted for a short duration of time, with small sample sizes and individuals of each species were co-housed within their respective treatment.
Additional evidence for ex situ infectivity and susceptibility of Malagasy frogs come from a breeding facility in Tokyo where individuals of Plethodontohyla tuberata were found to be infected with Bd54 and from a chytridiomycosis outbreak in a zoo with high mortality of Dyscophus antongilii55. In spite of some Malagasy species being able to acquire infection, continued studies are needed to fully understand how Malagasy frogs would respond to the Bd lineage(s) identified in Madagascar as well as other genotypes that could arrive in the future.
The current lack of detection of negative effects in the wild populations may suggest that the strain of Bd in Madagascar is hypovirulent, as Bd strains are known to vary in virulence44. Furthermore, we cannot rule out the long-term presence of an endemic Madagascar specific genotype/lineage of Bd that has evaded detection due to timing of sampling and/or methodologies or that it has recently increased in prevalence as a result of shifting environmental factors. With the current data it is not possible to discriminate between the opposing hypotheses that the detected Bd is introduced or endemic. Importantly, these hypotheses are not mutually exclusive. It is indeed possible that the high prevalence documented in August 2013 at Ranomafana and Ankaratra was an emergence event of an introduced genotype/lineage while the detection of Bd at Ankaratra in 2012, Ankarafantsika, Antoetra and particularly in Makay at lower levels was the detection of an endemic strain or lineage. Alternatively, the highly divergent prevalence recorded in Ankaratra from different years could be the result of an hybridization event between endemic and introduced strains (similar to what is reported for BdBrazil x BdGPL56), while the higher prevalence at Ranomafana in 2013 (versus the lack of detection of Bd in 2007) could be a result of a native Bd that through recombination became more virulent44. Continued research is needed to discern these hypotheses.
Detection of Bd in Madagascar appears to vary with seasons. For example, in Ankaratra at the site called Tavolotara, Bd was first detected in August 2012. Subsequently in August 2013, this site was again positive, with 100% prevalence; however, in December of 2013, no individuals were found positive. August is dry and cooler whereas December/January corresponds to the wet, warmer season in Madagascar. A similar trend of higher prevalence in the dry season and lower prevalence in the wet season was observed in Ankaratra at the Ambohimirandrana site as well as in Ranomafana at Vatoharanana. The same species that were previously found positive for Bd were resampled in the subsequent surveys, therefore, these differences in prevalence are likely not associated with the identity of the species sampled. In other regions of the world, including central America and Australia, Bd has been found to show dramatic seasonal trends, with higher prevalence occurring in the dryer and cooler season because cooler temperatures are more suitable for Bd57. In addition to temperature suitability, it is also possible that the seasonal dynamics of host microbial communities53 as well as environmental microbial and planktonic communities58 may be playing a role in the Bd dynamics observed in Madagascar.
It is important to note that different sample storage, extraction and detection methods have been used throughout the sampling events in Madagascar (Supplementary Table 1). Under the National Monitoring Plan, the protocol proposed to test the collected samples was a simple salt extraction followed by the traditional PCR assay59. Although more sensitive diagnostic assays were available60, the intent was to have this project running continuously in Madagascar where a molecular lab equipped with a traditional PCR platform was available. The above stated methodologies were used in the first two sampling events of the NMP, although this testing (when possible) was complemented with a qPCR assay. We acknowledge that variation in methodologies may confound time and season with detection method and could in part compromise the conclusions of the recent detection of Bd and seasonality trends. This does not compromise one of our main conclusions - that Bd has been detected in wild amphibians in Madagascar. It does, however, stress the importance of standardizing protocols for future investigations61. We suggest the following methodologies: 1) swabbing should be done with fine tip swabs and samples should be stored dry in cool temperatures, as recommended in Hyatt et al.62, 2) for extraction, Prepman should be used (although when time and money are not a constraint, Qiagen DNeasy extraction kits should be used as this is the best balance of efficiency and removal of PCR inhibition63), 3) for the detection assay we suggest the use of qPCR with BSA which is currently the most sensitive detection assay for Bd60,62. In addition, it will be important to standardize the reference strain of Bd used in different laboratories or to use cloned DNA fragment standards to allow accurate comparisons.
In light of Bd's presence in Madagascar it is imperative to ensure continuous monitoring across the country, especially at the sites already monitored under the NMP, at sites that have tested positive for Bd and at mid-high altitude sites, where the pathogen is more likely to be present64. This practice will develop a better understanding of Bd trends and dynamics in Madagascar enabling an effective response to an emerging threat. To minimize the spread of Bd, all researchers must adopt strict hygiene protocols31,65. While we do not know the virulence and real impacts of the Bd present in Madagascar, it is crucial to apply a precautionary principle and ensure surveillance of the frog populations to facilitate early detection of declines due to the importance of Malagasy amphibian species to global amphibian diversity. A further research priority must be to isolate the Malagasy Bd lineage(s) so that experimental approaches can be used to determine its virulence and evolutionary history.
In coordination with the Malagasy authorities, researchers and conservationists must prepare stakeholders for an effective response to a chytridiomycosis outbreak by the development and implementation of disease mitigation strategies66. In 2010, the first in-situ amphibian breeding facility was established in Andasibe, Madagascar, which can serve as a model for other captive assurance breeding centres, such as the coming breeding centre in Ivoloina Zoological Park67. These facilities may become vital resources for housing and preserving species if Bd-associated declines are documented30. Furthermore, probiotic therapy for amphibians is a promising disease mitigation strategy and can provide a potential mechanism to combat Bd in Madagascar53. In vitro assays of Malagasy amphibian skin bacteria against Bd have shown that some of the collected bacteria can strongly inhibit Bd growth. Similarly, recent discoveries58 in other regions of the world suggest that understanding and characterizing the microorganism communities of freshwater systems where Bd is present (or absent) may be important since these organisms could be part of a natural integrated strategy to reduce the spread of Bd and its infection potential.
We have documented the presence of Bd in wild amphibian populations in Madagascar; however, evidence for clinical signs of chytridiomycosis is so far lacking. The rarity of pre-2013 positives and the low intensity values found, in contrast with the high prevalence and intensity values found recently suggests either an emergence event, or a high degree of seasonality leading us to misdiagnose infection status in previous years in Madagascar. Continued research to fully understand the distribution, origin, type and virulence of the Bd lineage(s) present in Madagascar is essential and increased capacity to develop and implement conservation strategies are imperative for the successful conservation of Malagasy amphibians.
Methods
Site information
See Supplementary Table 1 for a list of the sites across Madagascar that were sampled for Bd (Figure 1). Sampling effort has been distributed across all bioclimatic regions of Madagascar (Figure 2, Supplementary Table 1).
Frog capture and sampling methods
NMP surveys were coordinated by the CEC: three focal species were selected and sampled at each of the eight monitored sites31. Surveys were completed in 2011–2013 by selected conservation organisations31. The last year of sampling for the first 3-years period of the NMP was recently concluded. For independent surveys undertaken outside the NMP, frogs were captured opportunistically during day and night searches. For all surveys, each individual was swabbed on its ventral surface of its abdomen, hind limbs and feet 5–10 times with a sterile fine-tip swab. Swabs collected under the NMP and independent surveys completed in 2007–2008 were immediately stored in unique vials with 96% ethanol. For independent surveys completed in 2013–2014 swabs were stored dry in 1.5 ml vials on ice until access to a freezer was available. In all conducted surveys, individuals were collected with newly gloved hands and placed in separate bags until processing to prevent cross contamination. Frogs were immediately released at the site of capture after sampling. In order to avert potential cross-contamination, hygiene procedures, including washing boots and all reusable sampling equipment with a 10% bleach solution were performed between sites.
DNA extraction and PCR detection analysis
Different DNA extraction methods have been used for the collected swab samples, including PrepMan Ultra Reagent Protocol as described by Boyle et al.60, standard salt extraction as described in Weldon et al.31 or a modification of this method as described in Bandi et al.68, Mobio PowerSoil DNA Isolation Kit (MoBio, Carlsbad, CA) as described by Costello et al.69 and 5 Prime Archive Pure Kit (Supplementary Table 1). For tissue samples, Qiagen DNeasy Blood and Tissue Kits (Qiagen, Valencia, CA) were used according to the manufacturer's protocol for animal tissues. DNA extracts were stored at −20, −25 or −80°C until downstream processing.
Tissue and swabs samples were assessed for the presence of Bd using both traditional polymerase chain reaction (PCR) and quantitative real-time polymerase chain reaction (qPCR). Samples from Andasibe (2011), Ankarafantsika (2011), Ankaratra (Oct 2010), Ankaratra (May 2012), Antoetra (2011–2012), Itremo (2008), Mandena (2011), Masoala (2012) and Toamasina (2011) were all tested using traditional PCR. All other samples were tested with qPCR or with both PCR and qPCR (Supplementary Table 1). Traditional PCR was performed according to Annis et al.59 while qPCR analyses were performed in accordance to the protocol outlined by Boyle et al.60. All extracts from PrepMan and salt extraction procedures were diluted 1:10 prior to PCR/qPCR analysis while all other extracts remained undiluted for testing. For qPCR, standards of known zoospore concentrations (made from a GPL isolate-Bd JEL 423 provided by Joyce Longcore- University of Maine) and negative controls were included in each qPCR plate. Each sample was run in duplicate qPCR reactions and single replicate positives were rerun. Samples were considered positive when amplification occurred in two qPCR reactions and the GEs quantity was greater than 0.1 GE (genome equivalents: reported as mean value for each sample). We used these values as an index of the intensity of an individual's infection. For a subset of samples from each lab completing qPCR analyses exogenous internal positive controls were included as described by Hyatt et al.62 to test for PCR inhibition. We found no evidence of PCR inhibition.
It is important to note, for the qPCR results, that different strains of Bd have different copy numbers of the ITS1-5.8S DNA fragment70, in part due to variable chromosomal copy numbers among strains and within strains44,71. Therefore, comparisons of the Bd infection intensities performed by different teams should be made with caution. For the same reasons, infection intensity should be interpreted with caution, as it is not yet known what type of Bd is present in Madagascar. Importantly, these issues are less of a concern for presence-absence data.
Lineage-specific qPCR
Singleplex quantitative PCR reactions utilizing Taqman MGB probes were used to discriminate single nucleotide polymorphisms in the Bd mitochondrial genome that are diagnostic for three major lineages of Bd: BdGPL, BdCAPE and BdCH44,72,73. qPCR conditions were adapted from the conditions described by Boyle et al.60, differing at the annealing step where the temperature was raised from 60°C to 62°C. These methods were performed on the samples collected in 2010 and 2011 at Makay, a set of samples from 5 locations sampled in 2012 under the NMP and a subset of samples from August 2013 collected by Bletz and colleagues in 2013 and 2014 (Supplementary Table 1).
Crayfish sampling
In 2013, crayfish were found to be an alternative host for Bd46. The recent introduction of an invasive crayfish in Madagascar raised concerns about the role of the species as a possible source of Bd introduction and therefore, we decided to include crayfish sampling in the following year to investigate this hypothesis. In January 2014, crayfish in Antananarivo (n = 55), Mandraka (n = 10) and Ranomafana (n = 11) were surveyed for Bd. In Antananarivo and Mandraka, individuals of the introduced invasive species Procambarus spp. were collected while in Ranomafana individuals of the native species Astacoides sp. were collected. Crayfish were euthanized via freezing and the gastrointestinal (GI) tract was dissected from each individual. The dissected GI tracts were stored in EtOH until laboratory processing. The intestinal tissue was cleaned using sterilized scissors and forceps to remove waste and debris. DNA was extracted from intestinal tissue using Qiagen DNeasy Blood and Tissue Kit, according to the manufacturer's protocol for animal tissues and qPCR as described above was used to test for the presence of Bd.
Exposure trial methods
Preliminary exposure trials were carried out on six species of Malagasy frogs (Boophis madagascariensis, B. viridis, Heterixalus betsileo, Guibemantis liber, Mantidactylus betsileanus and Ptychadena mascareniensis), at the North-West University (NWU) in Potchefstroom, South Africa. All experimental methods were carried out in accordance with the approved guidelines and protocols under the ethics permit no. NWU-00013-10-S4 issued by the NWU Research Ethics Committee. After a 12-day acclimation period, individuals were assigned either to a control treatment or exposed to a 5-day regiment of BdGPL (strain MG04 isolated from Amietia fuscigula, Western Cape, South Africa). Frogs were swabbed before treatment and after six, 15 and 20 days and samples analysed with qPCR. See Supplementary Materials for more details on methods.
Statistical Analysis
Given the ad-hoc nature of the sampling design, no formal statistical analyses were performed, which avoids giving false confidence to preliminary findings.
References
Hoffmann, M. et al. The impact of conservation on the status of the world's vertebrates. Science 330, 1503–9 (2010).
Duffus, A. & Cunningham, A. Major disease threats to European amphibians. Herpetol. J. 20, 117–127 (2010).
Fisher, M. C. et al. Emerging fungal threats to animal, plant and ecosystem health. Nature 484, 186–94 (2012).
Skerratt, L. F. et al. Spread of chytridiomycosis has caused the rapid global decline and extinction of frogs. Ecohealth 4, 125–134 (2007).
Berger, L. et al. Chytridiomycosis causes amphibian mortality associated with population declines in the rain forests of Australia and Central America. Proc. Natl. Acad. Sci. U. S. A. 95, 9031–6 (1998).
Lips, K. R. et al. Emerging infectious disease and the loss of biodiversity in a Neotropical amphibian community. Proc. Natl. Acad. Sci. U. S. A. 103, 3165–70 (2006).
Vredenburg, V. T., Knapp, R. A., Tunstall, T. S. & Briggs, C. J. Dynamics of an emerging disease drive large-scale amphibian population extinctions. Proc. Natl. Acad. Sci. U. S. A. 107, 9689–94 (2010).
Rosa, G. M. et al. Evidence of chytrid-mediated population declines in common midwife toad in Serra da Estrela, Portugal. Anim. Conserv. 16, 306–315 (2013).
La Marca, E. et al. Catastrophic population declines and extinctions in Neotropical harlequin frogs (Bufonidae: Atelopus). Biotropica 37, 190–201 (2005).
Crawford, A. J., Lips, K. R. & Bermingham, E. Epidemic disease decimates amphibian abundance, species diversity and evolutionary history in the highlands of central Panama. Proc. Natl. Acad. Sci. U. S. A. 107, 13777–82 (2010).
Wake, D. B. & Vredenburg, V. T. Are we in the midst of the sixth mass extinction? A view from the world of amphibians. Proc. Natl. Acad. Sci. U. S. A. 105, 11466–11473 (2008).
Dahl, C. et al. Batrachochytrium dendrobatidis not found in rainforest frogs along an altitudinal gradient of Papua New Guinea. Herpetol. J. 22, 183–186 (2012).
Weldon, C., du Preez, L. & Vences, M. Lack of detection of the amphibian chytrid fungus (Bactrachochytrium dendrobatidis) in Madagascar in. A Conservation strategy for the amphibians of Madagascar (ed Andreone, F.) 95–106 (Monographie del Museo Regionale di Scienze Naturali di Torino, 2008).
Crottini, A., Barbuto, M., Casiraghi, M. & Andreone, F. Amphibian survey at Itremo-Ambatofinandrahana, central Madagascar, with confirmed absence of chytrid fungus and recommendations for future monitoring activities. North West. J. Zool. 7, 346–351 (2011).
Vredenburg, V. T. et al. A molecular survey across Madagascar does not yield positive records of the amphibian chytrid fungus Batrachochytrium dendrobatidis. Herpetol Notes 5, 507–517 (2012).
Crottini, A. et al. Amphibian survey and current absence of Batrachochytrium dendrobatidis (Bd) in Ivoloina Park, Toamasina (eastern Madagascar). African J. Herpetol. 63, 1–9 (2014).
Lötters, S. et al. Meeting the challenge of conserving Madagascar's megadiverse amphibians: addition of a risk-assessment for the chytrid fungus. PLoS Biol. 6, e118 (2008).
Crottini, A. et al. Vertebrate time-tree elucidates the biogeographic pattern of a major biotic change around the K-T boundary in Madagascar. Proc. Natl. Acad. Sci. U. S. A. 109, 5358–63 (2012).
Perl, R. G. B. et al. DNA barcoding Madagascar's amphibian fauna. Amphibia-Reptilia 35, 197–206 (2014).
Frost, D. R. Amphibian Species of the World: an Online Reference. Am. Museum Nat. Hist. (2014). at <http://research.amnh.org/herpetology/amphibia/index.html> Date of access: 01/05/2014).
Rosa, G. M. et al. The amphibians of the relict Betampona low-elevation rainforest, eastern Madagascar: an application of the integrative taxonomy approach to biodiversity assessments. Biodivers. Conserv. 21, 1531–1559 (2012).
Strauß, A., Randrianiaina, R. D., Vences, M. & Glos, J. Species distribution and assembly patterns of frog larvae in rainforest streams of Madagascar. Hydrobiologia 702, 27–43 (2012).
Andreone, F., Rosa, G. M. & Raselimanana, A. P. Les amphibiens de l'Ouest et du Sud de Madagascar. (University of Chicago Press, Antananarivo, 2014).
Raxworthy, C. & Nussbaum, R. Extinction and extinction vulnerability of amphibians and reptiles in Madagascar. Amphib. Reptil. Conserv. 2, 15–23 (2000).
Vallan, D. Influence of forest fragmentation on amphibian diversity in the nature reserve of Ambohitantely, highland Madagascar. Biol. Conserv. 96, 31–43 (2000).
Andreone, F. et al. Species review of amphibian extinction risks in Madagascar: conclusions from the global amphibian assessment. Conserv. Biol. 19, 1790–1802 (2005).
Andreone, F. et al. The challenge of conserving amphibian megadiversity in Madagascar. PLoS Biol. 6, e118 (2008).
Andreone, F. & Randriamahazo, H. Sahonagasy Action Plan. (Museo Regionale di Scienze Naturali, Conservation International, International Union for Conservation of Nature, Torino, 2008).
Kremen, C. et al. Aligning conservation priorities across taxa in Madagascar with high-resolution planning tools. Science 320, 222–6 (2008).
Edmonds, D. et al. Building capacity to implement conservation breeding programs for frogs in Madagascar: Results from year one of Mitsinjo's amphibian husbandry research and captive breeding facility. Amphib. Reptil. Conserv. 5, 57–69 (2012).
Weldon, C. et al. Pre-emptive national monitoring plan for detecting the amphibian chytrid fungus in Madagascar. Ecohealth 10, 234–40 (2013).
Andreone, F. et al. Age structure, population estimate and Bd-status of two Critically Endangered frogs from the Ankaratra Massif (Madagascar), Boophis willliamsi and Mantidactylus pauliani (Amphibia: Mantellidae). Scr. Herpetol. dedicati a Benedetto Lanza. (2014).
Kolby, J. E. Presence of the amphibian chytrid fungus Batrachochytrium dendrobatidis in native amphibians exported from Madagascar. PLoS One 9, e89660 (2014).
Rabemananjara, F. C. E. & Raharivololoniaina, L. Madagascar and chytrid news: needed an urgent action and close collaboration between stakeholders. FrogLog 97, 33 (2011).
Puy, D. J. D. U., Moat, J., Royal, T., Gardens, B. & United, G. D. O. M. A refined classification of the primary vegetation of Madagascar vased on the underlying geology: using GIS to map its distribution and to assess its conservation status. Biogeogr. Madagascar 1996, 205–218 (1996).
Rödder, D. et al. Global amphibian extinction risk assessment for the panzootic chytrid fungus. Diversity 1, 52–66 (2009).
McDonald, K. R. & Alford, R. A. A review of declining frogs in northern Queensland in Declines and Disappearances of Australian frogs. (ed. Campbell, A.) 14–22 (Environment Australia, Canberra, 1999).
Gründler, M. C. et al. Interaction between breeding habitat and elevation affects prevalence but not infection intensity of Batrachochytrium dendrobatidis in Brazilian anuran assemblages. Dis. Aquat. Organ. 97, 173–84 (2012).
Walker, S. F. et al. Factors driving pathogenicity vs. prevalence of amphibian panzootic chytridiomycosis in Iberia. Ecol. Lett. 13, 372–382 (2010).
Cheng, T. L., Rovito, S. M., Wake, D. B. & Vredenburg, V. T. Coincident mass extirpation of Neotropical amphibians with the emergence of the infectious fungal pathogen Batrachochytrium dendrobatidis. Proc. Natl. Acad. Sci. U. S. A. 108, 9502–7 (2011).
Sarrasin, B. Ecotourism, poverty and resources management in Ranomafana, Madagascar. Tour. Geogr. 15, 3–24 (2013).
Glaw, F. & Vences, M. A field guide to the amphibians and reptiles of Madagascar. (Vences & Glaw, Köln, 2009).
Fisher, M. C., Garner, T. W. J. & Walker, S. F. Global emergence of Batrachochytrium dendrobatidis and amphibian chytridiomycosis in space, time and host. Annu. Rev. Microbiol. 63, 291–310 (2009).
Farrer, R. A. et al. Multiple emergences of genetically diverse amphibian-infecting chytrids include a globalized hypervirulent recombinant lineage. Proc. Natl. Acad. Sci. U. S. A. 108, 18732–18736 (2011).
Jones, J. et al. The perfect invader: a parthenogenic crayfish poses a new threat to Madagascar's freshwater biodiversity. Biol. Invasions 11, 1475–1482 (2009).
McMahon, T. et al. Chytrid fungus Batrachochytrium dendrobatidis has nonamphibian hosts and releases chemicals that cause pathology in the absence of infection. Proc. Natl. Acad. Sci. U. S. A. 110, 210–215 (2013).
Kolby, J. E. et al. Stop Madagascar's toad invasion now. Nature Corresp. 509, 563 (2014).
Bai, C., Liu, X., Fisher, M. C., Garner, T. W. J. & Li, Y. Global and endemic Asian lineages of the emerging pathogenic fungus Batrachochytrium dendrobatidis widely infect amphibians in China. Divers. Distrib. 18, 307–318 (2012).
Johnson, M. L. & Speare, R. Possible modes of dissemination of the amphibian chytrid Batrachochytrium dendrobatidis in the environment. Dis. Aquat. Organ. 65, 181–6 (2005).
Cunningham, A. A., Daszak, P. & Rodriguez, J. P. Pathogen pollution: Defining a parasitological threat to biodiversity conservation. J. Parasitol. 89, S78–S83 (2003).
Savage, A. E. & Zamudio, K. R. MHC genotypes associate with resistance to a frog-killing fungus. Proc. Natl. Acad. Sci. U. S. A. 108, 16705–10 (2011).
Rollins-Smith, L. A. The role of amphibian antimicrobial peptides in protection of amphibians from pathogens linked to global amphibian declines. Biochim. Biophys. Acta 1788, 1593–9 (2009).
Bletz, M. C. et al. Mitigating amphibian chytridiomycosis with bioaugmentation: characteristics of effective probiotics and strategies for their selection and use. Ecol. Lett. 16, 807–20 (2013).
Une, Y., Kadekaru, S., Tamukai, K., Goka, K. & Kuroki, T. First report of spontaneous chytridiomycosis in frogs in Asia. Dis. Aquat. Organ. 82, 157–60 (2008).
Oevermann, A., Schildger, B., Feldman, S. & Robert, N. Chytridiomykose bei Tomatenfröschen (Dyscophus antongilii) in der Schweiz. Tierärztl. Umsch. 60, 211–217 (2005).
Schloegel, L. M. et al. Novel, panzootic and hybrid genotypes of amphibian chytridiomycosis associated with the bullfrog trade. Mol. Ecol. 21, 5162–5177 (2012).
Kriger, K. M. & Hero, J.-M. Large-scale seasonal variation in the prevalence and severity of chytridiomycosis. J. Zool. 271, 352–359 (2006).
Schmeller, D. S. et al. Microscopic aquatic predators strongly affect infection dynamics of a globally emerged pathogen. Curr. Biol. 24, 176–80 (2014).
Annis, S. L., Dastoor, F. P., Ziel, H., Daszak, P. & Longcore, J. E. A DNA-based assay identifies Batrachochytrium dendrobatidis in amphibians. J. Wildl. Dis. 40, 420–8 (2004).
Boyle, D. G., Boyle, D. B., Olsen, V., Morgan, J. A. T. & Hyatt, A. D. Rapid quantitative detection of chytridiomycosis (Batrachochytrium dendrobatidis) in amphibian samples using real-time Taqman PCR assay. Dis. Aquat. Organ. 60, 141–8 (2004).
Fisher, M. C., Schmeller, D. S., Bosch, J., Aanensen, D. M. & Garner, T. W. J. RACE: Risk Assessment of Chytridiomycosis to European Amphibian Biodiversity. FrogLog 101, 45–47 (2012).
Hyatt, A. D. et al. Diagnostic assays and sampling protocols for the detection of Batrachochytrium dendrobatidis. Dis. Aquat. Organ. 73, 175–92 (2007).
Bletz, M. C., Rebollar, E. A. & Harris, R. N. Differential efficiency among DNA extraction methods influences detection of the amphibian pathogen Batrachochytrium dendrobatidis. Dis Aquat Org 113, 1–8; 10.3354/dao02822 (2015).
Grogan, L. F. et al. Surveillance for emerging biodiversity diseases of wildlife. PLoS Pathog. 10, e1004015 (2014).
Rabibisoa, N. H. & Raharivololoniaina, L. Plan strategique de prevention et de lutte contre l'introduction du chytride a Madagascar. (Conservation International & Amphibian Specialist Group, Antananarivo., 2012).
Woodhams, D. et al. Mitigating amphibian disease: strategies to maintain wild populations and control chytridiomycosis. Front. Zool. 8, 8 (2011).
Dawson, J. et al. Working together to develop Madagascar's amphibian captive breeding capacity. FrogLog, 22, 61–62 (2014).
Bandi, C. et al. Flavobacteria as intracellular symbionts in cockroaches. Proc. Biol. Sci. 257, 43–8 (1994).
Costello, E. K. et al. Bacterial community variation in human body habitats across space and time. Science 326, 1694–7 (2009).
Longo, A. V. et al. ITS1 copy number varies among Batrachochytriumdendrobatidis strains: implications for qPCR estimates of infection intensity from field-collected amphibian skin swabs. PLoS One 8, e59499 (2013).
Rosenblum, E. B. et al. Complex history of the amphibian-killing chytrid fungus revealed with genome resequencing data. Proc. Natl. Acad. Sci. U. S. A. 110, 9385–90 (2013).
Farrer, R. A. et al. Chromosomal copy number variation, selection and uneven rates of recombination reveal cryptic genome diversity linked to pathogenicity. PLoS Genet. 9, e1003703 (2013).
Ng, N. & Fisher, M. C. Variation in virulence, infectivity and competition between hypervirulent and hypovirulent lineages of Batrachochytrium dendrobatidis. MSc Thesis, Imperial College London (2011).
Acknowledgements
We are grateful to all Malagasy colleagues and authorities who helped in establishing a solid network of collaboration, facilitate chytrid research within the country and were committed to the creation and implementation of the Chytrid National Monitoring Program. We thank the Direction des Eaux et Forêts, Ministry of the Environment and Madagascar National Parks for supplying research and export permits. We are grateful to numerous students, assistants and colleagues who assisted during fieldwork. In particular we thank P. Bora, A. Thorel, J. Dawson, J. Lewis, L. Woolaver, R. Dolch, J. Noël, B. Iambana, K. Freeman, A. Bollen, M. Moore, L. du Preez, J. B. Ramanamanjato, J. C. Rakotoarisoa, A. Rakotoarison, T. F. Rakotonanahary, H. Razafindraibe, E. Wendenbaum, S. Ndriantsoa, V. Rabemananjara, C. Hutter, J. M. Ramanantsoa, P.O. Cochard and N. Rakotondrzafy. We are grateful to the team of Naturevolution for organizing the 2010 field work in the Makay Massif. We are thankful for the laboratory assistance of A. Pessier at the San Diego Zoo, Amphibian Disease Laboratory and the staff of FEM2 Ambiente srl of the Università degli studi di Milano-Bicocca. For outstanding support in terms of laboratory work and critical discussions we thank T.W.J. Garner. Much of this work was carried out under collaboration accords of TU Braunschweig, Germany and the American Museum of Natural History, USA, with the Université d'Antananarivo, Departement de Biologie Animale and of the Museo Regionale di Scienze Naturali Torino, Italy, with the Parc Botanique et Zoologique de Tsimbazaza. Funding was provided by the Saint Louis Zoo Field Conservation program (grants: FC #11-5) to F.A., A.C., F.R., D.E. and C.W.; the Rufford Small Grants for Nature Conservation (grant: 12931-1) to F.A., A.C., F.R. and C.W.; The Mohamed bin Zayed Species Conservation Fund (grants: 10051700; 1025982 and 12253505) to F.A., A.C., F.R., N.R. and C.W.; Andrew Sabin Family Foundation grant to F.A.; the European Association of Zoos and Aquaria grant to F.A. and C.W.; a grant of the Paris Zoo, France and of the Parc de Thoiry, France to F.A. and A.C.; a small grant from Brother Industries (Brother earth, 2010–2012) to N.R.; Volkswagen Foundation and the Deutsche Forschungsgemeinschaft (grant VE247/9-1) to M.V., The Mohamed bin Zayed Species Conservation Fund (grant 13255440) to R.N.H. and M.C.B., the Chester Zoo Conservation and Research Grant to R.N.H. and M.C.B., National Science Foundation grant 1136602 to R.N.H., Naturevolution Association Environnementale (grant: MakayNature) to E.C. and C.R. and the National Science Foundation (grant: DEB 0641023) to C.R. and a grant from the ERANET BiodivERsA project RACE (Risk Assessment of Chytridiomycosis to European amphibian biodiversity 2008-29014-62678-16) to E.A.C., M.C.F. and D.S.S. G.M.R. holds a doctoral scholarship from the Fundação para a Ciência e a Tecnologia (FCT) (SFRH/BD/69194/2010); A.C. was supported by a postdoctoral grant from FCT (SFRH/BPD/96982/2013) under the Programa Operacional Potencial Humano – Quadro de Referência Estratégico Nacional funds from the European Social Fund and Portuguese Ministério da Educação e Ciência. We acknowledge the project ‘Genomics and Evolutionary Biology' co-financed by North Portugal Regional Operational Programme 2007/2013 (ON.2 - O Novo Norte), under the National Strategic Reference Framework (NSRF), through the European Regional Development Fund (ERDF). MCF was funded by the UK Natural Environmental Research Council (NERC NE/K014455/1).
Author information
Authors and Affiliations
Contributions
M.C.B., G.M.R., A.C., M.V., M.F., R.N.H. and F.A. conceived and designed the study. M.C.B., G.M.R., A.C., E.A.C., N.R., F.R., M.V., C.W., D.E., C.R., R.N.H. and F.A. performed sampling and M.C.B., G.M.R., A.C., E.A.C., D.S.S., C.W. and M.C.F. performed Bd screening. C.W. and L.R. performed exposure experiments. M.C.B., G.M.R. and A.C. analysed the data and wrote the paper. All authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Supplementary Information
Supplementary Information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article's Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder in order to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Bletz, M., Rosa, G., Andreone, F. et al. Widespread presence of the pathogenic fungus Batrachochytrium dendrobatidis in wild amphibian communities in Madagascar. Sci Rep 5, 8633 (2015). https://doi.org/10.1038/srep08633
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep08633
This article is cited by
-
Chytrid fungi and global amphibian declines
Nature Reviews Microbiology (2020)
-
High interannual variability of a climate-driven amphibian community in a seasonal rainforest
Biodiversity and Conservation (2020)
-
Island provides a pathogen refuge within climatically suitable area
Biodiversity and Conservation (2015)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.