Abstract
Crab culture has gained prominence in the last decade due to the large global market demand for live crabs and crab products. Portunus sanguinolentus is one of the economically important crab species in the Indo-Pacific region, with distinct differences in growth and size between male and female crabs, thus, leading to huge difference in their market values. The culture of P. sanguinolentus is still in its infancy, with crab supplies heavily dependent on wild catch. In order to unravel the molecular differences between male and female crabs, we generated a comprehensive transcriptomic dataset for P. sanguinolentus by sequencing the gonads of both sexes using the Illumina Hiseq 2500 system. Transcriptomes were assembled using Trinity de novo assembly followed by annotation. This transcriptomic data set for P. sanguinolentus would serve as an important reference data for genomic and genetic studies in this crab and related species.
Design Type(s) | transcription profiling design |
Measurement Type(s) | transcription profiling assay |
Technology Type(s) | RNA sequencing |
Factor Type(s) | |
Sample Characteristic(s) | Portunus sanguinolentus • testis • ovary |
Machine-accessible metadata file describing the reported data (ISA-Tab format)
Similar content being viewed by others
Background & Summary
Transcriptome sequencing encompasses several applications including revealing single nucleotide polymorphism (SNP), quantitative trait loci (QTL) and analysis of genes actively expressed in specific tissues or under specific conditions, especially in non-model organisms whose full genome data are unavailable. Successful transcriptome analysis of many economically important aquaculture crab species including Scylla paramamosain1,2, Scylla olivacea3, Portunus trituberculatus,4–6, Eriocheir sinensis7,8 and Sinopotamon henanensis9 have been carried out.
Transcriptome data generated from various organs or tissues, including testis5,10, ovary2,11, accessory sex gland12, hepatopancreas13 and eyestalk4 are usually mined to study the genetic differences between sexes, and to uncover potential sex-related mechanisms. Gonads are the most widely used organs for transcriptome sequencing in reproductive research due to their direct involvement in sexual differentiation, gonadal development and maturation3,5,12. Testes are involved in the regulation of androgenic gland hormones and spermatozoa production13 in males, whereas ovaries are involved in the production of oocytes and secretion of maturation-related hormones in females14.
The crab culture industry has gained importance in the last decade due to high global market demand for live crabs and crab products. Currently, crab culture mainly relies on wild-caught seeds. However, this unregulated crab fishing practice, coupled with various anthropogenic factors, such as overfishing and environmental deterioration, results in a rapid decline in numbers of many economically important crab species15,16. Portunus sanguinolentus is generally known as the three-spot swimming crab due to the presence of three red to maroon prominent spots on the posterior part of its carapace17. It is widely distributed in the Indo-Pacific region, from the east coast of South Africa to Hawaiian waters18 and mainly inhabits sandy oceanic habitats to a depth of 30 meters19,20. While P. sanguinolentus is becoming the main candidate for marine aquaculture due to its high market demand21, very limited genetic studies have been carried out on this crab species and its transcriptome information is still unavailable. A recent gonadal transcriptome study conducted during mating embrace on a closely-related portunid species, P. trituberculatus revealed several reproduction and gonadal development related genes such as ADRA1B, BAP1, ARL3 and TRPA1 (ref. 6) that might be useful for understanding the molecular mechanism of reproductive development in crabs. However, in genus Portunus, the transcriptome resources are only mined in P. trituberculatus, which severely limited the genetic studies on other species. So far, no transcriptome resources are available in P. sanguinolentus. Thus, gonadal transcriptomic profiles of P. sanguinolentus would greatly contribute to molecular marker discovery, help to uncover the regulatory roles of the gonads in portunids, and use as reference for studies in other Portunus species.
In the present study, the gonadal transcriptome of P. sanguinolentus was sequenced using the Illumina HiSeq 2500 platform. A total of 174,935,588 raw reads and 119,718 unigenes were obtained, with 38,909, 24,641, 31,849, 29,103, 14,937, and 18,406 unigenes annotated to the NR, NT, Swiss-Prot, KEGG, COG, and GO databases, respectively. In addition, a total of 93,196 microsatellites were detected in the unigenes, while 97,364 and 151,626 SNPs were identified in testis and ovary, respectively. This data descriptor provides the first transcriptomic information for P. sanguinolentus that is useful for future genomic and genetic studies of this crab and related species. This data would also serve as an important reference for studies on sex differentiation and gonadal maturation mechanisms in marine crabs.
Methods
RNA extraction, cDNA library and Illumina sequencing
Total RNA was extracted from the gonads of eight independent P. sanguinolentus (four males designated HXX and four females designated HXC, Table 1) using Trizol (Invitrogen, CA, USA) according to the manufacturer’s instructions. NanoDrop® spectrophotometers (Thermo Fisher, MA, USA), Qubit® RNA Assay Kit in Qubit® 3.0 Flurometer (Life Technologies, CA, USA) and RNA Nano 6000 Assay Kit of the Bioanalyzer 2100 system (Agilent Technologies, CA, USA) were used to determine the RNA purity, concentration and integrity values, respectively. Equal amounts of RNA (RNA quality score (RQS) 5.5–7.1 and OD260/280 1.8–2.0) from each individual in the same gender were pooled together as one group, and 1 μg of RNAs from each group used for the library construction. Sequencing libraries were generated using the VAHTS mRNA-seq v2 Library Prep Kit for Illumina® (Vazyme, NR601) following the manufacturer’s recommendations. Firstly, mRNA was purified from the total RNA using poly-A oligo-attached magnetic beads, followed by RNA fragmentation using divalent cations under elevated temperature in Vazyme Frag/Prime Buffer. The cleaved RNA fragments were used for first strand cDNA synthesis using reverse transcriptase and random primers. Second strand cDNA synthesis was subsequently performed using buffer, dNTPs, DNA polymerase I and RNase H. Then, the cDNA fragments were end repaired by the addition of a single ‘A’ base at the 3′-end of each strand, ligated with the special sequencing adapters (Vazyme, N803). The products were purified and size selected using VAHTSTM DNA Clean Beads (Vazyme, N411) in order to obtain the appropriate size (350–450 bp) for sequencing.
The preliminary concentrations of the cDNA libraries were determined using the Qubit® RNA Assay Kit on the Qubit® 3.0 Flurometer, while the insert size was assessed using the Agilent Bioanalyzer 2100 system. Samples with the appropriate insert size were then accurately quantified using qPCR on the Step One Plus Real-Time PCR system (ABI, USA). Next, clustering of the index-coded library samples was performed on a cBot Cluster Generation System (Illumina, USA) according to the manufacturer’s instructions, followed by sequencing on an Illumina Hiseq 2500 platform with 150 bp paired-end module carried out by Vazyme BioTechnologies CO. Ltd (Nanjing, JiangSu, China). The Illumina GA processing pipeline was used for image analysis and base calling.
De novo assembly and functional annotation
In order to obtain high quality sequences for de novo assembly analysis, raw reads were filtered by removing: (1) reads containing adapters; (2) reads containing ploy-N (i.e., unrecognized bases) or reads with a ratio greater than 5%; and (3) low-quality reads (number of bases with Q≤10 more than 50% of the entire reads). The clean reads of the two libraries were then assembled into contigs using software Trinity (version: release-20130225, settings: --min_contig_length 150 --CPU 8 --min_kmer_cov 3 --min_glue 3 --bfly_opts '-V 5 --edge-thr=0.1 --stderr'). Three independent software modules including Inchworm, Chrysalis, and Butterfly within Trinity were applied sequentially to process large volumes of RNA-seq reads. First, the Inchworm module assembled the RNA-seq data into unique sequences of transcripts, generating full-length transcripts for a dominant isoform. However, only unique portions of alternatively spliced transcripts (Inchworm contigs) were reported. Next, the Inchworm contigs were assembled into clusters and complete de Bruijn graphs were constructed for each cluster using Chrysalis module. Each cluster represents the full transcriptional complexity for a given gene (or sets of genes that shared common sequences). Using the same module, the full read set among disjoint graphs were partitioned. Next, the Butterfly module then processed the individual graphs in parallel, tracing the paths that reads and pairs of reads take within the graph, ultimately reporting full length transcripts for alternatively spliced isoforms, and teasing apart transcripts that correspond to paralogous genes. The resulting sequences from Trinity are called unigenes. Since all samples were of the same species, unigenes from each sample's assembly were further processed with TGICL to cluster assembly sequences, remove redundancy (setting: -l 40 -c 10 -v 25 -O '-repeat_stringency 0.95 -minmatch 35 -minscore 35') and to acquire non-redundant unigenes. Assembly sequences longer than or equal to 200 bp were extracted as unigenes for subsequent analysis. Finally, annotations were assigned to each unigene based on the top hit in BLASTx search against the protein databases, with the non-redundant (NR) protein database at GenBank (http://www.ncbi.nlm.nih.gov) as the highest priority, followed by Swiss-Prot (http://www.expasy.ch/sprot), Kyoto Encyclopedia of Genes and Genomes (KEGG) (http://www.genome.jp/kegg) and Cluster of Orthologous Group (COG) in that priority order. The significant threshold of E-value was set at ≤10−5. To further annotate the unigenes, the Blast2GO22 program v2.5.0 was employed to obtain their Gene Ontology (GO) annotations based on molecular function, biological process and cellular component features.
Identification of SSRs and SNPs
The MicroSAtellite (MISA) software (http://pgrc.ipk-gatersleben.de/misa/misa.html) was used to identify SSRs markers in all unigenes, with search criteria as follows: di-nucleotide repeats ≥6, tri-nucleotide to hexa-nucleotide repeats ≥5 and the largest interval between two SSRs was ≤100 bases.
All unigenes were used as reference sequences to detect potential single nucleotide polymorphisms (SNPs) using the SOAPsnp program (http://soap.genomics.org.cn/soapsnp.html)23. SNPs sites were predicted based on the different bases at one position in the assembled sequences from the same unigene.
Data Records
The transcriptome data are available in the NCBI Sequence Read Archive (SRA) database (Data citation 1). Assembly sequence file was uploaded to DDBJ/EMBL/GenBank (Data citation 2). The annotation data (NR, NT, Swiss-Prot, KEGG, COG, and GO database annotations) as well as microsatellites and SNPs were uploaded to figshare (Data Citation 3).
Technical Validation
A total of 174,935,588 raw reads were generated by Illumina sequencing. After removing the adapter primers as well as low-quality and very short (<50 nt) reads, 167,001,196 clean reads were obtained. Finally, a total of 119,718 unigenes were produced with an average length of 904 nt, which were uploaded to Transcriptome Shotgun Assembly (TSA) project deposited at DDBJ/EMBL/GenBank.
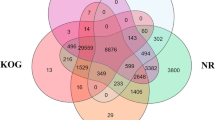
About 43.4% of the unigenes (47,536) were aligned to the protein databases NR at GenBank, Swiss-Prot, KEGG, and COG using BLASTx, while alignment to GO (E-value<10−5) was with Blast2GO, and nucleotide database NT (E-value<10−5) using BLASTn. A total of 38,909 unigenes were annotated to NR, 24,641 to NT, 31,849 to Swiss-Prot, 29,103 to KEGG, 14,937 to COG, and 18,406 to GO database (Table 2 and Data Citation 3).
All unigenes were subjected to functional annotation and classifications analyses. In the COG analysis, 12,617 (14.60%) unigenes were annotated and grouped into 25 COG classifications (Fig. 1 and COG annotation, Data citation 3). The largest cluster was “the general function prediction only (R)”, indicating that the functions of most of the bioinformatics predicted genes had not yet been confirmed by experimentation. The next cluster was “translation, ribosomal structure and biogenesis (J)”, followed by “replication, recombination and repair (L)”, and “cell cycle control, cell division, chromosome partitioning (D)”.
For the GO classification, Blast2GO program was first used to obtain the GO annotations followed by the use of WEGO software program to perform the GO functional classification to understand the distribution patterns and functions of the genes at the macro level. A total of 18,406 (15.37%) unigenes were annotated to 60 GO classification terms. In the “biological process” category, majority of the unigenes were represented in cellular process (12,963), single-organism process (10,591), metabolic process (9,889), biological regulation (7,265) and regulation of biological process (6,647). On the other hand, cell (10,673), cell part (10,660), organelle (7,413), membrane (5,837), and organelle part (4,415) were the most represented items in the “cellular component” category, while binding (9,392) and catalytic activity (7,736) were the highest in the molecular function category (Fig. 2, Table 3 and GO annotation, Data citation 3). Further analysis of the GO annotations at different levels revealed that most unigenes were enriched in membrane (Level 1, 1,888 unigenes), protein binding (Level 2, 1,732 unigenes), cytoplasm (Level 5, 1,497 unigenes), nucleus (Level 7, 1,480 unigenes) and integral component of membrane (Level 4, 1,456 unigenes).
Functional classification and pathway assignment based on KEGG analysis showed that metabolic pathways, regulation of actin cytoskeleton, amoebiasis, Vibrio cholerae infection and focal adhesion were the top five KEGG pathways (Table 3 and KEGG pathways, Data citation 3). The presence of the Vibrio cholera infection pathway among the top five KEGG pathways suggests that virus might have infected the wild crabs used in the study. While this is uncommon due to the ineluctable microbial presence in the open habitat of crabs, it is synonymous with the previous observation where microbial metabolism in diverse environment was among the top 5 KEGG pathways found in gonadal transcriptome profiles of the mud crab, S. paramamosain2,24.
About 38.5% of the unigenes exhibited a strong homology with the sequences available in the NR database (E-value <1.0e-45) (Fig. 3a), with 30,958 unigenes matched to the known sequence of 591 species. Most of the unigenes top hit species include the water flea Daphnia pulex (3477, 8.94%), red flour beetle Tribolium castaneum (2487, 6.39%), body louse Pediculus humanus corporis (1703, 4.38%), parasitoid wasp Nasonia vitripennis (1219, 3.13%) and purple sea urchin Strongylocentrotus purpuratus (1162, 2.99%) (Fig. 3c). Only 5.5% of the unigenes had similarity above 80% (Fig. 3b). (NR annotation, Data citation 3).
In analyzing the differences in gene expression between the two libraries (testis and ovary), the expression of 117,555 unigenes for testis and ovary from the transcriptome data were used. Of these unigenes, 72,517 unigenes were commonly expressed, while 12,503 unigenes were expressed only in ovary and 32,535 unigenes expressed only in testis.
The two libraries (testis and ovary) generated a total of 93,196 SSRs (Table 3), with the largest number of SSR motifs being di-nucleotide repeats (22,574) followed by tri-nucleotide repeats (14,675) (Fig. 4 and Microsatellites, Data citation 3). SNP loci were predicted based on the different bases at one position in the assembled sequences from the same unigenes. A total of 97,364 SNPs were detected in the unigenes from the testis library while 151,626 SNPs were found in the ovary library (Table 3). There were about 2.6 times more frequent occurrence of transition type SNPs than transversion in both testis and ovary, with similar numbers of A-G and C-T types being the most abundant transition SNPs in the two groups (Fig. 5 and SNPs, Data citation 3).
The data provided in these experimental datasets are the first report on the transcriptome resources for male and female P. sanguinolentus, which includes microsatellite sequences and SNP loci analyzed by Illumina high throughput sequencing technology. These findings are useful for identification of sex-related genes, as well as development of polymorphic genetic markers in P. sanguinolentus and other closely related species.
Additional information
How to cite this article: Zhang, Y. et al. Transcriptome sequencing and molecular markers discovery in the gonads of Portunus sanguinolentus. Sci. Data 5:180131 doi: 10.1038/sdata.2018.131 (2018).
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
References
Ma, H. et al. Transcriptome analysis of the mud crab (Scylla paramamosain) by 454 deep sequencing: assembly, annotation and marker discovery. PLoS ONE 9, e102668 (2014).
Gao, J. et al. Transcriptome analysis of the differences in gene expression between testis and ovary in green mud crab (Scylla paramamosain). BMC Genomics 15, 585 (2014).
Waiho, K. et al. Transcriptome analysis and differential gene expression on the testis of orange mud crab, Scylla olivacea during sexual maturation. PLoS ONE 12, e0171095 (2017).
Lv, J. et al. Transcriptomic variation of eyestalk reveals the genes and biological processes associated with molting in Portunus trituberculatus. PLoS ONE 12, e0175315 (2017).
Meng, X. L. et al. De novo Transcriptome analysis of Portunus trituberculatus ovary and testis by RNA-seq: Identification of genes involved in gonadal development. PLoS ONE 10, e0128659 (2015).
Wang, Z et al. De novo transcriptome sequencing and analysis of male and female swimming crab (Portunus trituberculatus) reproductive systems during mating embrace (stage II). BMC Genetics 19, 3 (2018).
Xu, Y. et al. Comparative transcriptome sequencing of the hepatopancreas reveals differentially expressed genes in the precocious juvenile Chinese mitten crab, Eriocheir sinensis (Crustacea: Decapoda). Aquac. Res. 48, 3645–3656 (2016).
Li, E. et al. Transcriptome sequencing revealed the genes and pathways involved in salinity stress of Chinese mitten crab. Eriocheir sinensis. Physiol. Genomics 46, 177–190 (2014).
Sun, M. et al. Transcriptome assembly and expression profiling of molecular responses to cadmium toxicity in hepatopancreas of the freshwater crab Sinopotamon henanense. Sci. Rep. 6, 19405 (2016).
Zhang, W. et al. A transcriptome analysis of mitten crab testes (Eriocheir sinensis). Genet. Mol. Biol. 34, 136–141 (2011).
Yang, Y. et al. Ovarian transcriptome analysis of Portunus trituberculatus provides insights into genes expressed during phase III and IV development. PLoS ONE 10, e0138862 (2015).
He, L. et al. Comparative transcriptome analysis of the accessory sex gland and testis from the Chinese mitten crab (Eriocheir sinensis). PLoS ONE 8, e53915 (2013).
Waiho, K. et al. Gonadal development in males of the orange mud crab, Scylla olivacea (Herbst, 1796) (Decapoda, Brachyura, Portunidae). Crustaceana 90, 1–19 (2017).
Mente, E. et al. in Reproductive Biology of Crustaceans (USA: Science Publishers, 2008).
Waiho, K. et al. Size distribution, length–weight relationship and size at the onset of sexual maturity of the orange mud crab, Scylla olivacea, in Malaysian waters. Mar. Biol. Res. 12, 726–738 (2016).
Fazhan, H. et al. Sympatric occurrence and population dynamics of Scylla spp. in equatorial climate: Effects of rainfall, temperature and lunar phase. Estuar. Coast Shelf Sci. 198, 299–310 (2017).
Carpenter K. E. & Niem V. H. (eds) FAO species identification guide for fishing purpose. The living marine resources of the Western Central Pacific, vol. 2, Cephalopds, Crustaceans, Holothurian and Sharks. FAO Report 687, 1–139 (1998).
Apel, M. & Spiridonov, V. A. Taxonomy and zoogeography of the portunid crabs (Crustacea: Decapoda: Brachyura: Portunidae) of the Arabian Gulf and adjacent waters. Fauna of Arabia 17, 159–331 (1998).
Sumpton, W. D., Smith, G. S. & Potter, M. A. Notes on the biology of the portunid crabs, Portunus sanguinolentus (Herbst) in subtropical Queensland waters. Aust. J. Mar. Freshw. Res. 40, 711–717 (1989).
Carpenter, K. E. et al. FAO species identification field guide for fishery purpose. The living marine resources of Kuwait, Eastern Saudi Arabia, Bahrain, Qatar, and the United Arab Emirates. FAO Report 7, 1–293 (1997).
Williams, M. J. & Primavera, J. H. Choosing tropical portunid species for culture, domestication and stock enhancement in the Indo-Pacific. Asian Fish. Sci. 14, 121–142 (2001).
Conesa, A. et al. Blast2GO: a universal tool for annotation, visualization and analysis in functional genomics research. Bioinformatics 21, 3674–3676 (2005).
Li, R. et al. SOAP2: an improved ultrafast tool for short read alignment. Bioinformatics 25, 1966–1967 (2009).
Yang, X. et al. Comparative transcriptome analysis provides insights into differentially expressed genes and long non-coding RNAs between ovary and testis of the mud crab (Scylla paramamosain). Mar. Biotechnol. 20, 20–34 (2017).
Data Citations
NCBI Sequence Read Archive SRX3324256 (2017)
GenBank GFZC01000000 (2017)
Zhang, Y. et al. Figshare https://doi.org/10.6084/m9.figshare.c.3972747 (2018)
Acknowledgements
This study was supported by the National Program for Support of Top-Notch Young Professionals, the STU Scientific Research Foundation for Talents (No. NTF17006), the National Natural Science Foundation of China (No. 31772837), and the “Sail Plan” Program for the Introduction of Outstanding Talents of Guangdong Province, China.
Author information
Authors and Affiliations
Contributions
Experiments were conceived and designed by M.H., and performed by Z.Y., M.G. Data were analyzed by Z.Y., W.Q. Paper was written by Z.Y., and revised by M.H., L.F., Y.C. and W.S. A.J.J. was involved in language improvement of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
ISA-Tab metadata
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/ The Creative Commons Public Domain Dedication waiver http://creativecommons.org/publicdomain/zero/1.0/ applies to the metadata files made available in this article.
About this article
Cite this article
Zhang, Y., Miao, G., Wu, Q. et al. Transcriptome sequencing and molecular markers discovery in the gonads of Portunus sanguinolentus. Sci Data 5, 180131 (2018). https://doi.org/10.1038/sdata.2018.131
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/sdata.2018.131
This article is cited by
-
Microsatellite markers reveal genetic diversity and population structure of Portunus trituberculatus in the Bohai Sea, China
Scientific Reports (2023)
-
Development of 84 single nucleotide polymorphism (SNP) markers for the three-spot swimming crab (Portunus sanguinolentus) by using RAD approach
Conservation Genetics Resources (2022)
-
The Single-molecule long-read sequencing of Scylla paramamosain
Scientific Reports (2019)
-
Transcriptome sequencing, molecular markers, and transcription factor discovery of Platanus acerifolia in the presence of Corythucha ciliata
Scientific Data (2019)
-
De novo assembly of genome and development of polymorphic microsatellite loci in the blue swimming crab (Portunus pelagicus) using RAD approach
Molecular Biology Reports (2018)