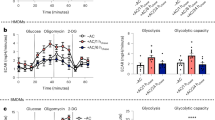
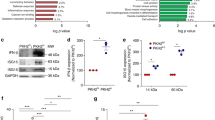
Abstract
The clearance of apoptotic cells by macrophages (efferocytosis) prevents necrosis and inflammation and activates pro-resolving pathways, including continual efferocytosis. A key resolution process in vivo is efferocytosis-induced macrophage proliferation (EIMP), in which apoptotic cell-derived nucleotides trigger Myc-mediated proliferation of pro-resolving macrophages. Here we show that EIMP requires a second input that is integrated with cellular metabolism, notably efferocytosis-induced lactate production. Lactate signalling via GPR132 promotes Myc protein stabilization and subsequent macrophage proliferation. This mechanism is validated in vivo using a mouse model of dexamethasone-induced thymocyte apoptosis, which elevates apoptotic cell burden and requires efferocytosis to prevent inflammation and necrosis. Thus, EIMP, a key process in tissue resolution, requires inputs from two independent processes: a signalling pathway induced by apoptotic cell-derived nucleotides and a cellular metabolism pathway involving lactate production. These findings illustrate how seemingly distinct pathways in efferocytosing macrophages are integrated to carry out a key process in tissue resolution.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All data supporting the present study are available within the paper and supplementary information files. Source data are provided with this paper.
References
Morioka, S., Maueröder, C. & Ravichandran, K. S. Living on the edge: efferocytosis at the interface of homeostasis and pathology. Immunity 50, 1149–1162 (2019).
Doran, A. C., Yurdagul, A. & Tabas, I. Efferocytosis in health and disease. Nat. Rev. Immunol. 20, 254–267 (2020).
Arandjelovic, S. & Ravichandran, K. S. Phagocytosis of apoptotic cells in homeostasis. Nat. Immunol. 16, 907–917 (2015).
Boada-Romero, E., Martinez, J., Heckmann, B. L. & Green, D. R. The clearance of dead cells by efferocytosis. Nat. Rev. Mol. Cell Biol. 21, 398–414 (2020).
Dalli, J. & Serhan, C. N. Pro-resolving mediators in regulating and conferring macrophage function. Front. Immunol. 8, 1400 (2017).
Colegio, O. R. et al. Functional polarization of tumour-associated macrophages by tumour-derived lactic acid. Nature 513, 559–563 (2014).
Cai, H. et al. Moderate l-lactate administration suppresses adipose tissue macrophage M1 polarization to alleviate obesity-associated insulin resistance. J. Biol. Chem. 298, 101768 (2022).
Morioka, S. et al. Efferocytosis induces a novel SLC program to promote glucose uptake and lactate release. Nature 563, 714–718 (2018).
Schilperoort, M., Ngai, D., Katerelos, M., Power, D. A. & Tabas, I. PFKFB2-mediated glycolysis promotes lactate-driven continual efferocytosis by macrophages. Nat. Metab. 5, 431–444 (2023).
Gerlach, B. D. et al. Efferocytosis induces macrophage proliferation to help resolve tissue injury. Cell Metab. 33, 2445–2463.e2448 (2021).
Yurdagul, A. Jr et al. Macrophage metabolism of apoptotic cell-derived arginine promotes continual efferocytosis and resolution of injury. Cell Metab. 31, 518–533.e10 (2020).
Zhang, S. et al. Efferocytosis fuels requirements of fatty acid oxidation and the electron transport chain to polarize macrophages for tissue repair. Cell Metab. 29, 443–456.e445 (2019).
Farrell, A. S. & Sears, R. C. MYC degradation. Cold Spring Harb. Perspect. Med. 4, a014365 (2014).
Marshall, G. M. et al. SIRT1 promotes N-Myc oncogenesis through a positive feedback loop involving the effects of MKP3 and ERK on N-Myc protein stability. PLoS Genet. 7, e1002135 (2011).
Menssen, A. et al. The c-MYC oncoprotein, the NAMPT enzyme, the SIRT1-inhibitor DBC1, and the SIRT1 deacetylase form a positive feedback loop. Proc. Natl Acad. Sci. USA 109, E187–E196 (2012).
Li, L. et al. SIRT1 activation by a c-MYC oncogenic network promotes the maintenance and drug resistance of human FLT3-ITD acute myeloid leukemia stem cells. Cell Stem Cell 15, 431–446 (2014).
Nebbioso, A. et al. c-Myc modulation and acetylation is a key HDAC inhibitor target in cancer. Clin. Cancer Res. 23, 2542–2555 (2017).
Mao, B. et al. Sirt1 deacetylates c-Myc and promotes c-Myc/Max association. Int. J. Biochem. Cell Biol. 43, 1573–1581 (2011).
Zhang, Y. & Dong, F. Gfi1 upregulates c-Myc expression and promotes c-Myc-driven cell proliferation. Sci. Rep. 10, 17115 (2020).
Yang, L. et al. Lactate promotes synthetic phenotype in vascular smooth muscle cells. Circ. Res. 121, 1251–1262 (2017).
Watson, M. J. et al. Metabolic support of tumour-infiltrating regulatory T cells by lactic acid. Nature 591, 645–651 (2021).
Le, A. et al. Inhibition of lactate dehydrogenase A induces oxidative stress and inhibits tumor progression. Proc. Natl Acad. Sci. USA 107, 2037–2042 (2010).
McCubbrey, A. L. et al. MicroRNA-34a negatively regulates efferocytosis by tissue macrophages in part via SIRT1. J Immunol. 196, 1366–1375 (2016).
El Hayek, L. et al. Lactate mediates the effects of exercise on learning and memory through SIRT1-dependent activation of hippocampal brain-derived neurotrophic factor (BDNF). J. Neurosci. 39, 2369–2382 (2019).
Gertz, M. et al. Ex-527 inhibits Sirtuins by exploiting their unique NAD+-dependent deacetylation mechanism. Proc. Natl Acad. Sci. USA 110, E2772–E2781 (2013).
López-Villegas, D., Lenkinski, R. E., Wehrli, S. L., Ho, W. Z. & Douglas, S. D. Lactate production by human monocytes/macrophages determined by proton MR spectroscopy. Magn. Reson. Med. 34, 32–38 (1995).
Roussel, M. F. Signal transduction by the macrophage-colony-stimulating factor receptor (CSF-1R). J. Cell Sci. Suppl. 18, 105–108 (1994).
Stanley, E. R. & Chitu, V. CSF-1 receptor signaling in myeloid cells. Cold Spring Harb. Perspect. Biol. 6, a021857 (2014).
Cantó, C. et al. AMPK regulates energy expenditure by modulating NAD+ metabolism and SIRT1 activity. Nature 458, 1056–1060 (2009).
Costford, S. R. et al. Skeletal muscle NAMPT is induced by exercise in humans. Am. J. Physiol. Endocrinol. Metab. 298, E117–E126 (2010).
Cerda-Kohler, H. et al. Lactate administration activates the ERK1/2, mTORC1, and AMPK pathways differentially according to skeletal muscle type in mouse. Physiol. Rep. 6, e13800 (2018).
Jiang, S. et al. Mitochondria and AMP-activated protein kinase-dependent mechanism of efferocytosis. J. Biol. Chem. 288, 26013–26026 (2013).
Garcia, D. & Shaw, R. J. AMPK: mechanisms of cellular energy sensing and restoration of metabolic balance. Mol. Cell 66, 789–800 (2017).
Foster, J. R. et al. N-palmitoylglycine and other N-acylamides activate the lipid receptor G2A/GPR132. Pharmacol. Res. Perspect. 7, e00542 (2019).
Nii, T. et al. Imipridone ONC212 activates orphan G protein-coupled receptor GPR132 and integrated stress response in acute myeloid leukemia. Leukemia 33, 2805–2816 (2019).
Ampomah, P. B. et al. Macrophages use apoptotic cell-derived methionine and DNMT3A during efferocytosis to promote tissue resolution. Nat. Metab. 4, 444–457 (2022).
Kasikara, C. et al. Deficiency of macrophage PHACTR1 impairs efferocytosis and promotes atherosclerotic plaque necrosis. J. Clin. Invest. 131, e145275 (2021).
Park, D. et al. Continued clearance of apoptotic cells critically depends on the phagocyte Ucp2 protein. Nature 477, 220–224 (2011).
Yurdagul, A., Doran, A. C., Cai, B., Fredman, G. & Tabas, I. A. Mechanisms and consequences of defective efferocytosis in atherosclerosis. Front. Cardiovasc. Med. 4, 86 (2017).
Vadevoo, S. M. P. et al. The macrophage odorant receptor Olfr78 mediates the lactate-induced M2 phenotype of tumor-associated macrophages. Proc. Natl Acad. Sci. USA 118, e2102434118 (2021).
Sag, D., Carling, D., Stout, R. D. & Suttles, J. Adenosine 5′-monophosphate-activated protein kinase promotes macrophage polarization to an anti-inflammatory functional phenotype. J. Immunol. 181, 8633–8641 (2008).
Jha, M. K. et al. Macrophage monocarboxylate transporter 1 promotes peripheral nerve regeneration after injury in mice. J. Clin. Invest. 131, e141964 (2021).
Wang, Y. et al. Mitochondrial fission promotes the continued clearance of apoptotic cells by macrophages. Cell 171, 331–345.e322 (2017).
Li, X. et al. Lactate metabolism in human health and disease. Signal Transduct. Target. Ther. 7, 305 (2022).
Brooks, G. A. Lactate as a fulcrum of metabolism. Redox Biol. 35, 101454 (2020).
Sharma, M. et al. Regulatory T cells license macrophage pro-resolving functions during atherosclerosis regression. Circ. Res. 127, 335–353 (2020).
Newby, A. C. & Zaltsman, A. B. Fibrous cap formation or destruction–the critical importance of vascular smooth muscle cell proliferation, migration and matrix formation. Cardiovasc. Res. 41, 345–360 (1999).
Haas, R. et al. Lactate regulates metabolic and pro-inflammatory circuits in control of T cell migration and effector functions. PLoS Biol. 13, e1002202 (2015).
Skuljec, J. et al. Absence of regulatory T cells causes phenotypic and functional switch in murine peritoneal macrophages. Front. Immunol. 9, 2458 (2018).
Proto, J. D. et al. Regulatory T cells promote macrophage efferocytosis during inflammation resolution. Immunity 49, 666–677.e666 (2018).
Miao, P., Sheng, S., Sun, X., Liu, J. & Huang, G. Lactate dehydrogenase A in cancer: a promising target for diagnosis and therapy. IUBMB Life 65, 904–910 (2013).
Sinha, S. K. et al. Local M-CSF (macrophage colony-stimulating factor) expression regulates macrophage proliferation and apoptosis in atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 41, 220–233 (2021).
Shantha, G. P. et al. Association of blood lactate with carotid atherosclerosis: the Atherosclerosis Risk in Communities (ARIC) Carotid MRI Study. Atherosclerosis 228, 249–255 (2013).
Garcia-Alvarez, M., Marik, P. & Bellomo, R. Sepsis-associated hyperlactatemia. Crit. Care 18, 503 (2014).
Tabas, I. Consequences and therapeutic implications of macrophage apoptosis in atherosclerosis: the importance of lesion stage and phagocytic efficiency. Arterioscler. Thromb. Vasc. Biol. 25, 2255–2264 (2005).
Cui, X. et al. The G2A receptor deficiency aggravates atherosclerosis in rats by regulating macrophages and lipid metabolism. Front. Physiol. 12, 659211 (2021).
Koyama, T. Lactated Ringer’s solution for preventing myocardial reperfusion injury. Int. J. Cardiol. Heart Vasc. 15, 1–8 (2017).
Gleeson, M. et al. The anti-inflammatory effects of exercise: mechanisms and implications for the prevention and treatment of disease. Nat. Rev. Immunol. 11, 607–615 (2011).
Casteels, K. M. et al. Sex difference in resistance to dexamethasone-induced apoptosis in NOD mice: treatment with 1,25(OH)2D3 restores defect. Diabetes 47, 1033–1037 (1998).
Acknowledgements
This work was supported by an American Heart Association Postdoctoral Fellowship (grant no. 900337 to M.S.); the Niels Stensen Fellowship (to M.S.) and NIH/NHLBI grant nos. R35-HL145228 and P01-HL087123 (to I.T.). We thank L. Becker (University of Chicago) for providing Ldhafl/fl and Ldhafl/fl; LysMCre+/− mouse femurs for BMDM differentiation to use in our in vitro studies. We thank X. Wang (Columbia University) for assisting with intravenous injections for BMT. We acknowledge C. Lu of the Columbia Center for Translational Immunology Core Facility for assisting in the immunofluorescent imaging experiments that were conducted in the Columbia Center for Translational Immunology Core Facility, funded by NIH grant nos. P30CA013696, S10RR027050 and S10OD020056.
Author information
Authors and Affiliations
Contributions
D.N. and I.T. conceived the project. D.N., M.S. and I.T. provided intellectual input to the development of the project. D.N. performed the in vitro experiments. D.N. and M.S. performed the in vivo dexamethasone-thymus experiment.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Metabolism thanks Roel De Maeyer, Nicholas Leeper and Feilong Wang for their contribution to the peer review of this work. Primary Handling Editor: Alfredo Giménez-Cassina, in collaboration with the Nature Metabolism team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Related to Figs. 1 and 2: Controls for BMDM experiments.
a, BMDMs were incubated with or without ACs for 45 minutes before washing and chasing for 1 hour in low-serum DMEM ± 50 µM FX11. The media were assayed for lactate concentration (n = 3). b, BMDMs were transfected with scrambled (Scr) or Ldha siRNA for 72 hours and then assayed for Ldha mRNA by RT-qPCR (n = 3). c) BMDMs pre-treated with 50 µM FX11 for 1 hour were incubated for 45 minutes with PKH26-labelled ACs and quantified for % PKH26+ macrophages (n = 3). d, BMDMs were incubated with ACs for 45 minutes, chased for 3 hours ± 50 µM FX11 and ± 2/5/10 mM LA, and immunoblotted for Myc (n = 3). e, BMDMs were incubated with or without ACs for 45 minutes, chased for 3 hours ± 50 µM FX11 and ± 10 mM sodium lactate (NaLa), and immunoblotted for Myc (n = 3). f, BMDMs transfected with 50 nM scrambled or Myc siRNA for 72 hours were incubated with or without ACs for 45 minutes, chased for 3 hours, and immunoblotted for Myc (n = 3). g, BMDMs were treated ± 10 mM LA for 3 hours and immunoblotted for Myc (n = 3). h, BMDMs were treated for 24 hours ± 10 mM LA before performing a cell count (n = 3). i, BMDMs were incubated with or without ACs for 45 minutes, chased for 3 hours ± 10 mM LA, and immunoblotted for Myc (n = 3). j, BMDMs were incubated with ACs for 45 minutes, chased for 3 hours ± 50 µM FX11 and ± 10 or 25 mM LA, and immunoblotted for Myc. Similar results were obtained in a repeat experiment. k, BMDMs were incubated with or without ACs for 45 minutes, chased for 3 hours ± 10 µM MG132, and immunoblotted for Myc (n = 3). Bars represent means ± s.e.m. Statistics were performed by two-tailed student’s t-test in panels b-c, h, or one-way ANOVA in panels a, d, e, i, and k. n.s. = non-significant (P > 0.05).
Extended Data Fig. 2 Related to Figs. 3 and 4: Controls for BMDM experiments.
a, BMDMs were transfected with 50 nM scrambled or Sirt1 siRNA for 72 hours, incubated with PKH26-labelled ACs for 45 minutes, and quantified for the percent PKH26+ macrophages (n = 3). b, BMDMs were incubated with or without ACs for 45 minutes, chased for 3 hours ± 10 µM EX527 and ± 10 mM LA, and immunoblotted for Myc (n = 3). c, BMDMs were treated with 50 ng/mL CSF-1 for 3, 6, 12, or 24 hours and immunoblotted for Myc. The image presented is one representative replicate (n = 3). d, BMDMs were treated with or without 50 ng/mL CSF-1 + 50 µM FX11 or 10 µM EX527, and immunoblotted for Myc (n = 3). e, BMDMs were treated for 24 hours with 50 ng/mL CSF-1 + 50 μM FX11 or 10 μM EX527, and quantified for cell number (n = 3). f, BMDMs were transfected with 50 nM scrambled or Myc siRNA for 72 hours, incubated ± ACs for 45 minutes or with CSF-1, and then assayed 24 hours later for cell number and Myc protein for CSF1-treated cells (n = 3). g, BMDMs pre-treated for 1 hour with 10 µM CompC were incubated with PKH26-labelled ACs and quantified for the percent PKH26+ (n = 3). h, BMDMs were chased for 1 hour + 10 µM CompC with or without 500 µM NMN and then assayed for NAD+ to NADH ratio (n = 3). i, BMDMs were treated ± 500 µM NMN for 3 hours and then immunoblotted for Myc (n = 3). j, BMDMs transfected with scrambled or Sirt1 siRNA were chased for 3 hours ± 500 µM NMN and then immunoblotted for Myc (n = 3). Bars represent means ± s.e.m. Statistics were performed by two-tailed student’s t-test in panels a and g, or one-way ANOVA in panels b-f, h, and j. n.s. = non-significant (P > 0.05).
Extended Data Fig. 3 Related to Fig. 5: Controls for BMDM experiments.
a, BMDMs were transfected with 50 nM scrambled or Slc16a1 siRNA for 72 hours and then assayed for Slc16a1 mRNA by RT-qPCR (n = 3). b, BMDMs transfected with 50 nM scrambled or Slc16a1 siRNA for 72 hours were incubated with ACs for 45 minutes, chased for 3 hours in 1% FBS DMEM before collecting supernatants and cell lysates to measure intracellular and extracellular lactate concentrations (n = 3). c, BMDMs were transfected with 50 nM scrambled or Gpr132 siRNA for 72 hours and then assayed for Gpr132 mRNA by RT-qPCR (n = 3). d, BMDMs transfected with 50 nM scrambled or Gpr132 siRNA for 72 hours were incubated with or without ACs for 45 minutes, chased for 3 hours ± 50 µM FX11 and ± 10 mM LA, and immunoblotted for Myc. The displayed immunoblot is a representative replicate (n = 3). e, BMDMs were incubated with or without ACs for 45 minutes, chased for 3 hours ± 50 μM FX11, and assayed for Gpr132 mRNA (n = 3). Bars represent means ± s.e.m. Statistics were performed by two-tailed student’s t-test in panels a-c, one-way ANOVA in panel d, or two-way ANOVA in panel e. n.s. = non-significant (P > 0.05).
Extended Data Fig. 4 Related to Fig. 6: Controls and blood counts for the dexamethasone-thymus experiment.
a-f, Counts of blood WBCs, neutrophils, lymphocytes, monocytes, eosinophils, and basophils (n = 5). g, Plasma was measured for lactate concentration (n = 5). h, Mac2+ cells in immunostained thymus sections were counted per field of view (FOV) using images taken with a 20x objective (n = 8,6). Bars represent means ± s.e.m. Statistics were performed by two-tailed student’s t-test in panels a-e and g-h, and Mann–Whitney test for panel f. n.s. = non-significant (P > 0.05).
Supplementary information
Supplementary Table 1
qPCR primer sequences.
Source data
Source Data Fig. 1–6 and Extended Data Figs. 1–3
Unprocessed western blots for Figs. 1–6 and Extended Data Figs. 1–3.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ngai, D., Schilperoort, M. & Tabas, I. Efferocytosis-induced lactate enables the proliferation of pro-resolving macrophages to mediate tissue repair. Nat Metab 5, 2206–2219 (2023). https://doi.org/10.1038/s42255-023-00921-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s42255-023-00921-9