Abstract
Developing efficient strategies for Markovnikov hydrosilylation of alkynes is still an important goal. The steric and electronic properties of hydrosilanes are key factors in controlling selectivity in these reactions. Here by using a trimethylsilyl-protected trihydroxysilane, we report a mild, efficient strategy for Markovnikov hydrosilylation of terminal alkynes with the simple catalyst [Ir(μ-Cl)(cod)]2. A variety of terminal alkynes are hydrosilylated efficiently with outstanding α-regioselectivity. This protocol is successfully utilized in the late-stage hydrosilylation of derivatives of various bio-relevant molecules. The residual silyl group, -Si(OSiMe3)3, can participate in organic transformations directly, or be converted into other useful silyl groups.
Similar content being viewed by others
Introduction
Vinyl-substituted silicon compounds, or vinylsilanes, are remarkable building blocks in organic synthesis, polymer chemistry and materials science. Hydrosilylation of alkynes is arguably the most straightforward and atom-economical route to vinylsilanes1,2. One main challenge in such processes is the regio- and stereoselectivity control. Compared with various well established catalytic systems for hydrosilylation of terminal alkynes towards β-(E)- and β-(Z)-vinylsilanes, the ways to geminal α-vinylsilanes are still exiguous. In recent years, this problem has been partially addressed with the developments of several efficient protocols3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29. For instance, Ru-based complexes have been developed as the predominant catalysts for this field since the pioneering works from Trost and Yamamoto, et al.3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18. Co-catalyzed α-hydrosilylation of alkynes showed superiority in modification of aryl alkynes19,20,21,22,23. Highly regioselective silylcupration of terminal alkynes reported by Loh is another efficient approach to α-vinylsilanes24. However, further commercial and industrial use of these methods is limited by their own properties, such as the unstability of Ru catalysts, requirement of complex ligands and low efficiency towards alkyl alkynes in Co catalysis30, usage of expensive PhMe2SiBpin as silyl source and high catalyst loading in Cu catalysis. New strategies for Markovnikov hydrosilylation of alkynes that require simple catalysts or ligands, low-cost silanes, but have outstanding tolerance towards various functional groups and suit for large-scale operations, are still in urgent desire.
The selectivity of alkyne hydrosilylation processes is known to depend on several factors, such as the metal catalysts and ligands, steric or electronic properties of hydrosilanes and alkynes, etc. For example, studies on the Wu-Trost mechanism revealed the significance of ligands (cyclopentadiene and L in a1, Fig. 1a) in controlling the regiospecificity of the product31,32,33,34,35. Huang and Lu developed rational ligands for the cobalt-catalyzed α hydrosilylation of terminal alkynes, respectively (Fig. 1b)19,20,21,22. By contrast, exploitation of suitable hydrosilanes to achieve α regioselectivity under simple catalysis has been rarely studied36. Hydrosilanes with bulkier substituents are presumed to have lower reactivity, which might allow the coordination of the metal center with the alkyne to go first, leading the process to follow the Wu-Trost mechanism and bring α-vinylsilane adducts (Fig. 1c).
Markovnikov hydrosilylation of terminal alkynes. a Ruthenium-based catalytic system for α hydrosilylation of terminal alkynes. b Copper- and cobalt-based catalytic systems for α hydrosilylation of terminal alkynes. c This work: iridium-based catalytic system for α hydrosilylation of terminal alkynes
We share our findings here. By using a trimethylsilyl-protected trihydroxysilane, a mild and efficient strategy for α-hydrosilylating terminal alkynes is realized with a simple iridium catalyst, which is compatible with a variety of functional groups, and performs efficiently in modifying derivatives of bio-relevant molecules. The bulky silyl group could be easily converted into other useful silyl groups, or participate in subsequent derivatizations directly. It’s noteworthy that though iridium complexes have been broadly investigated for hydrosilylation of unsaturated C–C bonds37, anti-Markovnikov selectivity is usually observed in their use toward terminal alkynes38,39,40,41,42,43,44,45,46,47,48,49. As one example of Markovnikov hydrosilylation of internal thioalkynes has been reported by Sun and Wu50, this is to our knowledge the first case in which α-vinylsilanes are mainly provided from terminal alkynes under iridium catalysis.
Results
Reaction condition optimization
Heteroatoms and functional groups are commonly encountered in natural and pharmaceutical molecules. Developing hydrosilylation systems showing excellent tolerance toward them would be of great value. Meanwhile, the pre-existing heteroatoms or functional groups could usually provide inductive effect51,52, or coordinate with the metal center53,54,55,56,57, both of which might contribute to enhancing the selectivity in hydrofunctionalization reactions. With above considerations, phenyl propargyl ether was selected as the model alkyne substrate to initiate our study (Table 1). The bulky (TMSO)3SiH, which is easily prepared from HSiCl3 and (TMS)2O, was chosen as the silane partner to verify our anticipation. It is noteworthy that though in contrast with the wide utilization of Et3SiH, (EtO)3SiH and other common hydrosilanes in hydrosilylation of unsaturated molecules, examples that use (TMSO)3SiH as hydrosilane source are still limited58,59,60,61,62,63,64, it showed its unique function in promoting the reaction selectivity63 or inducing different selectivities64. Several simple iridium catalysts were tested firstly under a mild condition (entry 1–4). The desired α-vinylsilane product was obtained in 72% yield with excellent α regioselectivity under the catalysis by [Ir(μ-Cl)(cod)]2 (entry 1). Decreased yield and selectivity was observed with the utilization of (EtO)3SiH (entry 5). Using Et3SiH as the silyl source provided β-addition products as the majority in poor yield (entry 6). The yield was promoted to 88% with the addition of a catalytic amount of 1,5-cyclooctadiene (entry 7). Decreased yield and regioselectivity were observed when this process was performed under air and moisture (entry 8 and 9). In addition, other kinds of metal catalysts, including [Rh(μ-Cl)(cod)]2, Ni(cod)2, [Pd(allyl)Cl]2, were tested instead of [Ir(μ-Cl)(cod)]2 under the optimized condition, but giving no desired product.
Substrate scope
With this simple and mild optimum condition in hand, we then evaluated its generality towards alkynes (Fig. 2). Both of propargyl alcohol and a variety of its derivatives were highly regioselectively hydrosilylated under this catalytic system, affording related products in good to excellent yields (1–11). Hydrosilylation of electron-deficient propargylic acid and methyl propiolate proceeded with high efficiency and regioselectivity (12 and 13). Non-protected and protected homopropargyl alcohols could likewise participate in this hydrosilylation process smoothly (14–16). The regioselectivity deteriorated slightly in the hydrosilylation of alkynyl alcohol or acid substrates with extended main chains (17–19). Replacement of hydroxyl group with aryl group led to decreased regioselectivity as well (20). Various nitrogen-containing terminal alkynes were then tested, all of which could be transformed into corresponding vinylsilanes with high α regioselectivity (21–26). To further probe the efficiency of this protocol, reactions involving internal alkynes were carried out. Unsymmetrical 2-hexyn-1-ol was converted to a 2.2:1 mixture of α- and β-(E)-vinylsilanes in a total yield of 85% (27 and 28). Hydrosilylation of symmetric dialkyl alkyne provided syn adduct 29 in 87% yield with excellent stereoselectivity.
Scope of alkyne. i. Reaction conditions: The mixture of alkyne (0.50 mmol, 1.0 equiv), (TMSO)3SiH (0.75 mmol, 1.5 equiv), [Ir(μ-Cl)(cod)]2 (0.01 mmol, 2 mol %) and cod (0.02 mmol, 4 mol %) in MeCN (2.0 mL) was stirred for 6 h under room temperature; ii. Isolated total yield; iii. Determined by 1H NMR result of crude product; iv. 3.0 Equiv of silane was used; v. β-(E)/β-(Z) ratio is over 25/1; vi. Ratio of β-(E)/β-(Z)
Late-state hydrosilylation
Inspired by the excellent tolerance of this efficient strategy towards various functional groups, such as hydroxyl group, carboxyl group, alkenyl group, oxirane, ether, ester, amine and amide, etc., we were eager to learn about its performance in late-stage hydrosilylation of bio-relevant molecules (Fig. 3). Considering the widespread hydroxyl and amino groups in biomolecules, propargyl derivatives from them were selected as representatives in most cases (30–45). Propargyl ether analogues of menthol, carveol, prolinol, glucose and glucofuranose were successfully transformed into corresponding α-vinylsilanes in good to excellent yields (30–34). Other propargyl ethers derived from quinine, estrone, testosterone, cholesterol, and vitamin E could be effectively hydrosilylated as well, providing desired products with outstanding α regioselectivity (35–39). It is worthwhile to note that though terminal alkynyl derivatives from nitrogenous bases, theobromine, uridine, and phenylalanine showed poor solubility in acetonitrile, this protocol is well applicable in Markovnikov hydrosilylation of them as well (40–46).
Late-stage hydrosilylation of bio-relevant compounds. i. Reaction conditions: The mixture of alkyne (0.50 mmol, 1.0 equiv.), (TMSO)3SiH (0.75 mmol, 1.5 equiv), [Ir(μ-Cl)(cod)]2 (0.01 mmol, 2 mol %) and cod (0.02 mmol, 4 mol %) in MeCN (2.0 mL) was stirred for 6 h under room temperature; ii. Isolated total yield; iii. Determined by 1H NMR result of crude product
Product derivatizations
The bulky hindrance of -Si(TMSO)3 group has little influence on its further transformations, such as desilylation and Tamao oxidation (Fig. 4a). To simplify purification, compound 48 was easily obtained from Markovnikov hydrosilylation of homopropargyl alcohol under this newly-established mild condition and subsequent Hiyama cross-coupling65 with 4-iodotoluene in one pot (Fig. 4b). In addition, –Si(OTMS)3 is useful in silicone materials because of the easy conversion of it into other kinds of silyl groups66,67. For instance, –OSiMe2(vinyl) was successfully anchored to replace the –OTMS group, which could be further decorated by other means (Fig. 4c)68.
Stereochemistry study
As mentioned above, under this iridium catalytic system, excellent syn addition stereoselectivity was observed in the trisubstituted vinylsilanes derived from internal alkynes (27–29 in Fig. 2). The result of deuterium labeling experiment toward terminal alkynes showed the same stereochemistry (Fig. 5).
Mechanistic considerations
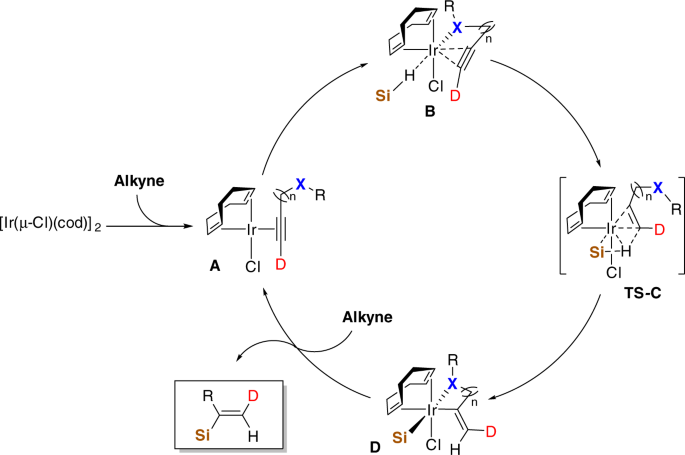
A plausible catalytic cycle is accordingly proposed as shown in Fig. 6. Instead of oxidation with (TMSO)3SiH, the iridium center will probably coordinate with alkyne to afford intermediate A in the initial step. Due to the steric repulsion between the bulky silyl group and the substituent on the alkyne, intermediate B is supposed to form and could be further stabilized through the chelation of the pre-existing heteroatoms or functional groups with iridium center. The following oxidative hydrometallation step via transition state C generates intermediate D, which finally affords α addition product and active catalytic species A for the next cycle through reductive silyl migration.
Some experiments were carried out to probe the mechanistic details (Fig. 7). (TMSO)3SiH was found to be stable when mixing with [Ir(μ-Cl)(cod)]2 at a ratio of 2:1 in CD3CN (Fig. 7a). Subsequent addition of alkyne to this mixture provided α-vinylsilane 1 in quantitative yield. By contrast, consumption of (EtO)3SiH was observed after the addition of iridium complex to its solution in CD3CN, and no signal of desired hydrosilylation product showed up in the 1H NMR spectra with further alkyne addition (Fig. 7b). The proposal for formation of intermediate A could be in some degree support by these findings and further mechanistic studies (see Supplementary Figs. 7–14 and Supplementary Discussions). More detailed studies are currently ongoing in our lab.
Mechanistic studies. a Comparison of 1H NMR spectra of (TMSO)3SiH, mixture of (TMSO)3SiH with [Ir(μ-Cl)(cod)]2 (2:1), and mixture with further alkyne addition (2.0 eq.). b Comparison of 1H NMR spectra of (EtO)3SiH, mixture of (EtO)3SiH with [Ir(μ-Cl)(cod)]2 (2:1), and mixture with further alkyne addition (2.0 eq.)
Gram-scale reaction
The stability of the mixture involving [Ir(μ-Cl)(cod)]2 and (TMSO)3SiH promoted us to investigate the efficiency of this protocol towards large-scale reactions. With lower loading of catalyst and silane, α-hydrosilylation products 1 and 2 were respectively afforded on gram scale (Fig. 8).
Discussion
To sum up, we reported here a mild and efficient strategy for Markovnikov hydrosilylation of terminal alkynes with excellent regio- and stereoselectivity. With a silane with bulky substituents and a simple iridium catalyst, a variety of terminal alkynes, especially those with heteroatoms or functional groups, were α-hydrosilylated smoothly in up to nearly quantitative yield. It’s noteworthy that this is, to our knowledge, the first iridium-catalyzed hydrosilylation of terminal alkynes with outstanding α regioselectivity. Furthermore, this strategy showed high efficiency in late-stage hydrosilylation of bio-relevant derivatives. The steric hindrance of the silyl group not only has little influence on subsequent manipulations of the α-vinylsilane products, but also permits increased stability of the silane towards the iridium catalyst, which allows this protocol to be used in larger-scale reactions with lower loading of both catalyst and silane. A catalytic cycle that is similar to the Wu-Trost mechanism was proposed for this new method. Further mechanistic investigations and exploration of other metal-based catalytic systems that could use the intrinsic properties of silanes to achieve selectivity control are on the way.
Methods
General procedure for iridium-catalyzed Markovnikov hydrosilylation of terminal alkynes
In a glove box, to an oven-dried 5-mL vial was added the alkyne (0.50 mmol), the silane (0.75 mmol), [Ir(μ-Cl)(cod)]2 (0.01 mmol, 2 mol %), cod (0.02 mmol, 4 mol %) and MeCN (2.0 mL). The vial was capped and removed from the glove box. The reaction mixture was stirred at room temperature for 6 h, and then concentrated under reduced pressure. The residue was purified by silica gel flash column chromatography (eluent: 0–50% EtOAc in petroleum ether or 0–20% MeOH in DCM) to give the desired product.
Product derivatizations
Full procedures for synthetic transformations of compounds 47–53 are available in the Supplementary Methods and Supplementary Figures. 1–3.
Deuterium labeling experiment
Please see Supplementary Methods and Supplementary Figs. 4–6.
Mechanistic study experiments
Please see Supplementary Methods, Supplementary Figs. 7–14, and Supplementary Discussion.
NMR spectra
1H and 13C NMR Spectra of all hydrosilylation products and unknown substrate were provided. 13C DEPT135 NMR Spectra of products 3, 4, 5, 6, 7, 9, 10, 12, 13, 15, 19, 20, 21, 26, 30, 31, 32, 36, 37, 38, 39, 40, 41, 42, 44, 45 were afford. 2D NOESY Results of products 1 and 27 are also provided. Please see Supplementary Figs. 15–145.
Data availability
The authors declare that all the data supporting the findings of this study are available within the paper and its supplementary information files, and also are available from the corresponding author upon reasonable request.
References
Marciniec, B. Hydrosilylation: A Comprehensive Review on Recent Advances (Springer, 2009).
Marciniec, B., Maciejewski, H. & Pawluć P. in Organosilicon Compounds: Experiment (Physico-Chemical Studies) and Applications (eds Lee, V. Y.) 169–217 (Elsevier, 2017).
Trost, B. M. & Ball, Z. T. Addition of metalloid hydrides to alkynes: hydrometallation with boron, silicon, and tin. Synthesis 6, 853–887 (2005).
Frihed, T. G. & Fürstner, A. Progress in the trans-reduction and trans-hydrometalation of internal alkynes. Applications to natural product synthesis. Bull. Chem. Soc. Jpn. 89, 135–160 (2016).
Zaranek, M., Marciniec, B. & Pawluć, P. Ruthenium-catalysed hydrosi-lylation of carbon–carbon multiple bonds. Org. Chem. Front 3, 1337–1344 (2016).
Na, Y. & Chang, S. Highly stereoselective and efficient hydrosilylation of terminal alkynes catalyzed by [RuCl2(p-cymene)]2. Org. Lett. 2, 1887–1889 (2000).
Trost, B. M. & Ball, Z. T. Markovnikov alkyne hydrosilylation catalyzed by ruthenium complexes. J. Am. Chem. Soc. 123, 12726–12727 (2001).
Kawanami, Y., Sonoda, Y., Mori, T. & Yamamoto, K. Ruthenium-catalyzed hydrosilylation of 1-alkynes with novel regioselectivity. Org. Lett. 4, 2825–2827 (2002).
Trost, B. M. & Ball, Z. T. Synthetic Stitching with Silicon: Geminal Alkylation-Hydroxylation of Alkynyl Carbonyl Compounds. J. Am. Chem. Soc. 126, 13942–13944 (2004).
Aricó, C. S. & Cox, L. R. Regio- and stereoselective hydrosilylation of terminal alkynes using Grubbs’ first-generation olefin-metathesis catalyst. Org. Biomol. Chem. 2, 2558–2562 (2004).
Trost, B. M., Ball, Z. T. & Laemmerhold, K. M. An alkyne hydrosilylation-oxidation strategy for the selective installation of oxygen functionality. J. Am. Chem. Soc. 127, 10028–10038 (2005).
Trost, B. M. & Ball, Z. T. Alkyne hydrosilylation catalyzed by a cationic ruthenium complex: efficient and general trans addition. J. Am. Chem. Soc. 127, 17644–17655 (2005).
Menozzi, C., Dalko, P. I. & Cossy, J. Hydrosilylation of terminal alkynes with alkylidene ruthenium complexes and silanes. J. Org. Chem. 70, 10717–10719 (2005).
Maifeld, S. V., Tran, M. N. & Lee, D. Hydrosilylation of alkynes catalyzed by ruthenium carbene complexes. Tetrahedron Lett. 46, 105–108 (2005).
Sore, H. F., Blackwell, D. T., MacDonald, S. J. F. & Spring, D. R. Diversity-oriented synthesis of disubstituted alkenes using masked silanols. Org. Lett. 12, 2806–2809 (2010).
Ding, S. et al. Ligand-controlled remarkable regio- and stereodivergence in intermolecular hydrosilylation of internal alkynes: experimental and theoretical studies. J. Am. Chem. Soc. 135, 13835–13842 (2013).
Rummelt, S. M., Radkowski, K., Roşca, D.-A. & Fürstner, A. Interligand interactions dictate the regioselectivity of trans-hydrometalations and related reactions catalyzed by [Cp*RuCl]. Hydrogen bonding to a chloride ligand as a steering principle in catalysis. J. Am. Chem. Soc. 137, 5506–5519 (2015).
Zhang, X., Ji, X., Xie, X. & Ding, S. Construction of highly sterically hindered 1,1-disilylated terminal alkenes. Chem. Commun. 54, 12958–12961 (2018).
Guo, J. & Lu, Z. Highly chemo-, regio-, and stereoselective cobalt-catalyzed Markovnikov hydrosilylation of alkynes. Angew. Chem., Int. Ed. 55, 10835–10838 (2016).
Zuo, Z., Yang, J. & Huang, Z. Cobalt-catalyzed alkyne hydrosilylation and sequential vinylsilane hydroboration with Markovnikov selectivity. Angew. Chem., Int. Ed. 55, 10839–10843 (2016).
Guo, J. & Lu, Z. Regio- and enantioselective cobalt-catalyzed sequential hydrosilylation/hydrogenation of terminal alkynes. Angew. Chem., Int. Ed. 56, 615–618 (2017).
Wen, H., Wan, X. & Huang, Z. Asymmetric synthesis of silicon-stereogenic vinylhydrosilanes by cobalt-catalyzed regio- and enantioselective alkyne hydrosilylation with dihydrosilanes. Angew. Chem., Int. Ed. 57, 6319–6323 (2018).
Wu, G., Chakraborty, U. & von Wangelin, A. J. Regiocontrol in the cobalt-catalyzed hydrosilylation of alkynes. Chem. Commun. 54, 12322–12325 (2018).
Wang, P., Yeo, X.-L. & Loh, T.-P. Copper-catalyzed highly regioselective silylcupration of terminal alkynes to form α-Vinylsilanes. J. Am. Chem. Soc. 133, 1254–1256 (2011).
Liu, Y., Yamazaki, S. & Yamabe, S. Regioselective hydrosilylations of propiolate esters with tris(trimethylsilyl)silane. J. Org. Chem. 70, 556–561 (2005).
Rivero-Crespo, M. A., Leyva-Pérez, A. & Corma, A. A Ligand-free Pt3 cluster catalyzes the Markovnikov hydrosilylation of alkynes with up to 106 turnover frequencies. Chem. Eur. J. 23, 1702–1708 (2017).
Rivera, G., Elizalde, O., Roa, G., Montiel, I. & Bernès, S. Fluorinated N-heterocyclic carbenes rhodium (I) complexes and their activity in hydrosilylation of propargylic alcohols. J. Organomet. Chem. 699, 82–86 (2012).
Iglesias, M. et al. A. Preferential α-hydrosilylation of terminal alkynes by bis-N-heterocyclic carbene rhodium(III). Catal. Adv. Synthe. Catal. 357, 350–354 (2015).
Nakamura, S., Uchiyama, M. & Ohwada, T. Chemoselective silylzincation of functionalized terminal alkynes using dianion-type zincate (SiBNOL-Zn-ate): regiocontrolled synthesis of vinylsilanes. J. Am. Chem. Soc. 126, 11146–11147 (2004).
Obligacion, J. V. & Chirik, P. J. Earth-abundant transition metal catalysts for alkene hydrosilylation and hydroboration. Nat. Rev. Chem. 2, 15–34 (2018).
Chung, L. W., Wu, Y.-D., Trost, B. M. & Ball, Z. T. A theoretical study on the mechanism, regiochemistry, and stereochemistry of hydrosilylation catalyzed by cationic ruthenium complexes. J. Am. Chem. Soc. 125, 11578–11582 (2003).
Yang, Y.-F., Chung, L. W., Zhang, X., Houk, K. N. & Wu, Y.-D. Ligand-controlled reactivity, selectivity, and mechanism of cationic ruthenium-catalyzed hydrosilylations of alkynes, ketones, and nitriles: a theoretical study. J. Org. Chem. 79, 8856–8864 (2014).
Cheng, G.-J., Zhang, X., Chung, L. W., Xu, L. & Wu, Y.-D. Computational organic chemistry: bridging theory and experiment in establishing the mechanisms of chemical reactions. J. Am. Chem. Soc. 137, 1706–1725 (2015).
Zhang, X., Chung, L. W. & Wu, Y.-D. New mechanistic insights on the selectivity of transition-metal-catalyzed organic reactions: the role of computational chemistry. Acc. Chem. Res. 49, 1302–1310 (2016).
Song, L.-J., Wang, T., Zhang, X., Chung, L. W. & Wu, Y.-D. A combined DFT/IM-MS study on the reaction mechanism of cationic Ru(II)-catalyzed hydroboration of alkynes. ACS Catal. 7, 1361–1368 (2017).
Ding, S. et al. Highly regio- and stereoselective hydrosilylation of internal thioalkynes under mild conditions. Angew. Chem., Int. Ed. 54, 5632–5635 (2015).
Marciniec, B. & Kownacki, I. in Iridium Complexes in Organic Synthesis (eds Oro, L. A. & Claver, C.) 345–367 (Wiley-VCH, 2009).
Fernandez, M. J. & Oro, L. A. Iridium-catalyzed hydrosilylation of hex-1-yne: the unusual formation of 1-triethylsilylhex-1-yne. J. Mol. Catal. 45, 7–15 (1988).
Tanke, R. S. & Crabtree, R. H. Iridium(I), (III), and -(V) complexes of an O-donor ligand in alkyne hydrosilylation. J. Chem. Soc., Chem. Commun. 1056–1057 (1990).
Tanke, R. S. & Crabtree, R. H. Unusual activity and selectivity in alkyne hydrosilylation with an iridium catalyst stabilized by an O-donor Ligand. J. Am. Chem. Soc. 112, 7984–7989 (1990).
Esteruelas, M. A., Oliván, M. & Oro, L. A. Reactions of the square-planar compounds Ir(C2Ph)L2(PCy3) (L2 = 2 CO, TFB) with HSiR3 (R = Et, Ph) and Hx+1SiPh3-x (x = 1, 2): stoichiometric and catalytic formation of Si-C bonds. Organometallics 15, 814–822 (1996).
Esteruelas, M. A., Lahoz, F. J., Onate, E., Oro, L. A. & Rodríguez, L. Reactions of Ir(acac)(cyclooctene)(PCy3) with H2, HCCR, HSiR3, and HSnPh3: the acetylacetonato ligand as a stabilizer for iridium(I), iridium(III), and iridium(V) derivatives. Organometallics 15, 823–834 (1996).
Field, L. D. & Ward, A. J. Catalytic hydrosilylation of acetylenes mediated by phosphine complexes of cobalt(I), rhodium(I), and iridium(I). J. Organomet. Chem. 681, 91–97 (2003).
Miyake, Y., Isomura, E. & Iyoda, M. Selective hydrosilylation of 1-alkynes using iridium catalyst with biphosphinine ligand. Chem. Lett. 35, 836–837 (2006).
Sridevi, V. S., Fan, W. Y. & Leong, W. K. Stereoselective hydrosilylation of terminal alkynes catalyzed by [Cp*IrCl2]2: a computational and experimental study. Organometallics 26, 1157–1160 (2007).
Zanardi, A., Peris, E. & Mata, J. A. Alkenyl-functionalized NHC iridium-based catalysts for hydrosilylation. New J. Chem. 32, 120–126 (2008).
Iglesias, M. et al. A synthon for a 14-electron Ir(III) species: catalyst for highly selective β-(Z) hydrosilylation of terminal alkynes. Chem. Commun. 48, 9480–9482 (2012).
Pérez-Torrente, J. J. et al. Hydrosilylation of terminal alkynes catalyzed by a ONO-pincer iridium(III) hydride compound: mechanistic insights into the hydrosilylation and dehydrogenative silylation catalysis. Organometallics 35, 2410–2422 (2016).
Corre, Y. et al. Regioselective hydrosilylation of terminal alkynes using pentamethylcyclopentadienyl iridium(III) metallacycle catalysts. J. Mol. Catal. A-Chem. 423, 256–263 (2016).
Song, L.-J. et al. Ir-catalyzed regio- and stereoselective hydrosilylation of internal thioalkynes: a combined experimental and computational study. J. Org. Chem. 81, 6157–6164 (2016).
Xi, Y. & Hartwig, J. F. Diverse asymmetric hydrofunctionalization of aliphatic internal alkenes through catalytic regioselective hydroboration. J. Am. Chem. Soc. 138, 6703–6706 (2016).
Xi, Y., Butcher, T. W., Zhang, J. & Hartwig, J. F. Regioselective, asymmetric formal hydroamination of unactivated internal alkenes. Angew. Chem., Int. Ed. 55, 776–780 (2016).
Ickes, A. R., Ensign, S. C., Gupta, A. K. & Hull, K. L. Regio- and chemoselective intermolecular hydroamination of allyl imines for the synthesis of 1,2-diamines. J. Am. Chem. Soc. 136, 11256–11259 (2014).
Ensign, S. C., Vanable, E. P., Kortman, G. D., Weir, L. J. & Hull, K. L. Anti-Markovnikov hydroamination of homoallylic amines. J. Am. Chem. Soc. 137, 13748–13751 (2015).
Kennemeur, J. L., Kortman, G. D. & Hull, K. L. Rhodium-catalyzed regiodivergent hydrothiolation of allyl amines and imines. J. Am. Chem. Soc. 136, 11914–11919 (2016).
Bai, X.-Y., Wang, Z.-X. & Li, B.-J. Iridium-catalyzed enantioselective hydroalkynylation of enamides for the synthesis of homopropargyl amides. Angew. Chem., Int. Ed. 55, 9007–9011 (2016).
Wang, Z.-X., Bai, X.-Y., Yao, H.-C. & Li, B.-J. Synthesis of amides with remote stereocenters by catalytic asymmetric γ‑alkynylation of α,β-unsaturated amides. J. Am. Chem. Soc. 138, 14872–14875 (2016).
Perry, R. J., Karageorgis, M. & Hensleer, J. Hydrosilylation reactions of 13-diynes and bis(silyl hydrides): model studies and polymerizations. Macromolecules 40, 3929–3938 (2007).
Dierick, S., Vercruysse, E., Berthon-Gelloz, G. & Markó, I. E. User-friendly platinum catalysts for the highly stereoselective hydrosilylation of alkynes and alkenes. Chem. Eur. J. 21, 17073–17078 (2015).
Roşca, D.-A. et al. Ruthenium-catalyzed alkyne trans-hydrometalation: mechanistic insights and preparative implications. J. Am. Chem. Soc. 139, 2443–2455 (2017).
Morales-Cerón, J. P. et al. Rhodium(I) complexes with ligands based on N-heterocyclic carbine and hemilabile pyridine donors as highly E stereoselective alkyne hydrosilylation catalysts. Organometallics 36, 2460–2469 (2017).
Julian, A. et al. Mechanistic insights on the reduction of CO2 to silylformates catalyzed by Ir-NSiN species. Chem. Eur. J. 23, 11898–11907 (2017).
Ding, S. et al. Highly regio- and stereoselective hydrosilylation of internal thioalkynes under mild conditions. Angew. Chem., Int. Ed. 54, 5632–5635 (2015).
Kitano, T., Komuro, T., Ono, R. & Tobita, H. Tandem hydrosilylation/o-C-H silylation of arylalkynes catalyzed by ruthenium bis(silyl) aminophosphine complexes. Organometallics 36, 2710–2713 (2017).
Hatanaka, Y. & Hiyama, T. Cross-coupling of organosilanes with organic halides mediated by palladium catalyst and tris(diethylamino)sulfonium difluorotrimethylsilicate. J. Org. Chem. 53, 918–920 (1988).
Zhang, Z., Feng, S. & Zhang, J. Facile and efficient synthesis of carbosiloxane dendrimers via orthogonal click chemistry between thiol and ene. Macromol. Rapid Commun. 37, 318–322 (2016).
Doherty, M. D. et al. Small molecule cyclic amide and urea based thickeners for organic and sc-CO2/organic solutions. Energy Fuels 30, 5601–5610 (2016).
Xie., X. et al. Iridium-catalyzed hydrosilylation of unactivated alkenes: scope and application to late-stage functionalization. J. Org. Chem. 84, 1085–1093 (2019).
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (No. 21702015, 21871023), State Key Laboratory of Organic−Inorganic Composites (Beijing University of Chemical Technology, oic-201801011), the Fundamental Research Funds for the Central Universities (buctrc201719).
Author information
Authors and Affiliations
Contributions
X.X., X.Z., W.G., C.M. and X.W. performed the experiments and prepared the Supplementary Information. S.D. conceived and directed the project. S.D. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Xie, X., Zhang, X., Gao, W. et al. Iridium-catalyzed Markovnikov hydrosilylation of terminal alkynes achieved by using a trimethylsilyl-protected trihydroxysilane. Commun Chem 2, 101 (2019). https://doi.org/10.1038/s42004-019-0206-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42004-019-0206-4
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.